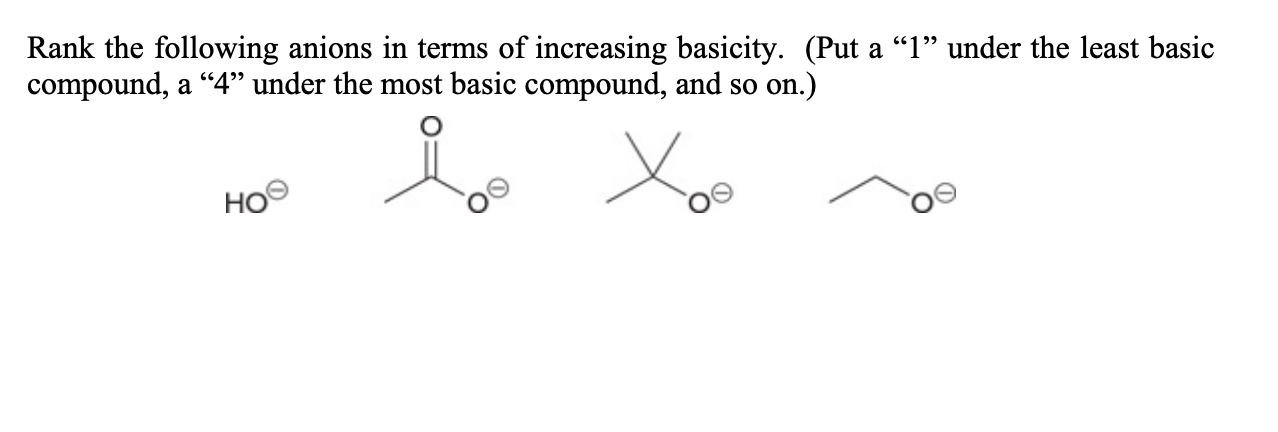
Rank The Following Anions In Terms Of Increasing Basicity
Hey there, coffee buddy! So, we're diving into the wild and wacky world of anions today, huh? Don't worry, it's not as scary as it sounds. Think of it like ranking your friends based on how likely they are to crash your party. Some are super chill, others... well, they bring the whole band. And today, we're ranking some anions based on their, get this, basicity. Sounds fancy, right? But it's really just about how much they want to grab onto a proton. You know, that little positively charged guy running around. Some anions are practically begging for one, while others are like, "Nah, I'm good."
So, what exactly is basicity when we're talking about these negatively charged dudes? It’s all about their hunger for a proton, H+. A stronger base is one that's really good at accepting that proton. Imagine a hungry hippo. That's a strong base. A really weak base? That's like your super-polite friend who asks, "May I perhaps have a sip of your water?" It's all about their willingness to get cozy with a proton.
And here's the super-secret handshake for figuring this stuff out: the weaker the conjugate acid, the stronger the base. Mind. Blown. It’s like, if something is really bad at letting go of a proton (meaning its conjugate acid is weak), then its anion form must be really good at snatching one up. It’s a whole relationship thing, you see? Like a clingy ex versus a chill roommate.
Alright, let’s get down to business. We’ve got a list of anions to rank, and it’s gonna be a fun ride. Think of it as a chemical popularity contest. Who’s the most desperate for a proton? Who’s just kinda… there?
Let's Get This Ranking Party Started!
Okay, drumroll please! We’re ranking these anions from least basic to most basic. So, the shyest ones at the beginning, and the proton-hoarders at the end. Get your metaphorical scorecards ready!
The "I'm Too Cool For Protons" Club (Weakest Bases)
First up, we have the anions that are basically saying, "Protons? Ew, no thanks." They're super stable on their own, like that one friend who’s always got their life together and doesn't need anyone's validation. They’ve got their negative charge spread out all over the place, making them feel all chill and unbothered. They’re the epitome of chemical indifference. Can you even imagine?
Think about it: if an anion is super stable, why would it bother reacting with a proton? It's got no incentive! It's like offering a Michelin-star chef a hot dog. They're probably gonna pass. And that’s exactly what these guys do. They’re the ones who make their conjugate acids super strong. They're like, "I can totally let go of this proton and be fine, thank you very much."
So, who are these aloof characters? We’re talking about anions that come from very, very strong acids. Like, acids so strong they practically ionize just by looking at water. Their conjugate bases are like the distant cousins you only see at Thanksgiving – barely acknowledge their existence, let alone interact.
The "Maybe Later" Crew (Moderately Weak Bases)
Next, we have the anions that are a little more open to the idea of proton cuddles, but they’re not exactly jumping at the chance. They're still pretty stable, but maybe they’ve had a rough day and wouldn’t mind a little positive reinforcement. They’re like your friend who’s always almost ready to go out but needs a few more minutes to get their hair just right. They’re not completely against the idea, but there’s a bit of a process.
These guys are derived from acids that are, well, strong. But maybe not super strong. They’re the middle-grounders. They’ll accept a proton, but they’re not going to go out of their way for it. They’re like, "Sure, if it’s convenient, I guess." Their conjugate acids are moderately strong. They're not as determined to hold onto that proton as the super-stable ones, but they're not exactly eager to hand it over either.
They’ve got some electron density, but it’s not like they’re just bursting with negativity. It’s contained, you know? Like a well-behaved dog that only barks when necessary. They’re part of the conversation, but they’re not dominating it. They’re the sensible ones, the ones who weigh their options before committing.
The "It's Getting Kinda Cozy In Here" Bunch (Stronger Bases)
Now we’re getting somewhere! This group of anions is starting to feel the proton pull. They’re not as stable as the ones before, and they’ve got a bit more of a negative charge that’s just begging to be neutralized. They’re like your friend who’s always up for a spontaneous adventure, but you still have to text them twice. They’re definitely interested, but there’s a slight delay.
These anions come from acids that are weaker. Like, significantly weaker. The acids themselves aren’t as eager to give up their protons, which means their conjugate bases are more than happy to snatch them back. It’s a beautiful symbiotic relationship, isn’t it? The acid is a bit stingy, and the base is a bit grabby. A perfect balance, really.
They’re starting to become a bit more reactive. They’re not just passively existing anymore; they're actively seeking out positive ions. Their electron density is more concentrated, making them more prone to interactions. Think of them as the friends who are always the first ones to suggest getting pizza. They’re proactive!
The "Where's My Proton? I Need It NOW!" Fanatics (Strongest Bases)
And at the very end of our ranking, we have the absolute proton-fiends! These anions are practically screaming, "Give me a proton! I’m practically vibrating with negative energy!" They are unstable on their own and desperate for that positive charge to calm them down. They are the life of the chemical party, always looking for a proton to dance with.
These are the anions that come from very weak acids. Acids that are so weak, they barely exist as acids in the first place. It’s like their conjugate acid is the friend who always borrows money and never pays it back. The anion is the one holding the bag, so to speak, and it’s looking for someone to balance things out. Their conjugate acids are incredibly weak, meaning they have absolutely no intention of holding onto a proton.
These guys are highly reactive. They are the definition of a strong base. They will grab onto any proton they can find. They’re like the friends who are always texting you memes at 2 AM. You might love them, you might find them a little much, but they are definitely there. They are the ones who make things happen, for better or worse!
The Actual, You Know, Rank-y Thing
Okay, okay, enough with the analogies. Let's see the actual anions in order. We’re going from least basic (the super chill ones) to most basic (the proton addicts). Remember, it's all about how badly they want that proton!
So, imagine we're throwing some anions into a proton party. Who gets the first dance?
At the very, very bottom of the basicity ladder, we have the conjugate bases of the strongest acids. Think about acids like perchloric acid (HClO₄). That anion, perchlorate (ClO₄⁻), is ridiculously stable. Its conjugate acid, HClO₄, is like the ultimate proton-donor. It’s so happy to get rid of its proton, the perchlorate ion is left feeling pretty neutral about taking one back. It's just... not that into protons. It's like saying, "I've seen it all, protons are old news." Its charge is super spread out due to resonance, making it a very weak base. Like, really weak.
Then we have anions from other super strong acids, like sulfuric acid (H₂SO₄). Its first conjugate base is bisulfate (HSO₄⁻). Bisulfate is still a pretty weak base. It's a little more inclined to grab a proton than perchlorate, but not by much. Its conjugate acid, H₂SO₄, is still a strong acid, so it’s still pretty good at holding onto its proton. HSO₄⁻ is like, "Yeah, I could maybe take a proton if you really insist, but don't expect me to be thrilled about it." It’s got a bit of negativity, but it’s not exactly desperate.
Moving up, we encounter anions derived from strong acids, but maybe not as strong as perchloric or sulfuric. Think about hydrochloric acid (HCl). Its anion is chloride (Cl⁻). Chloride is also a very weak base. It’s similar to perchlorate and bisulfate in its lack of enthusiasm for protons. It's like the reliable but not overly exciting friend. Its conjugate acid, HCl, is strong, so Cl⁻ is stable and doesn't really want a proton. It’s seen it all before, and it’s perfectly happy with its current charge.
Following along those lines, we have nitrate (NO₃⁻), the conjugate base of nitric acid (HNO₃). Similar to chloride, nitrate is a very weak base. Its conjugate acid, HNO₃, is strong, meaning NO₃⁻ is stable and doesn’t have a strong desire for protons. It's all about that resonance stabilization, keeping the negative charge spread out and making it less attractive to incoming positive charges.
Now, let's dip our toes into the slightly more interesting territory. We start seeing anions that are a bit more noticeable in their basicity. Think about phosphoric acid (H₃PO₄). Its second conjugate base is hydrogen phosphate (HPO₄²⁻). Now, HPO₄²⁻ is a bit more basic than the ones we just discussed. Its conjugate acid, H₂PO₄⁻, is a weaker acid than HCl or HNO₃. This means HPO₄²⁻ has a stronger pull towards protons. It’s like, "Okay, fine, I’ll take one. It’s not the worst thing in the world." It's starting to feel the pull more strongly.
Still in the realm of moderate bases, we have sulfide (S²⁻). Now, sulfide is derived from the second dissociation of hydrogen sulfide (H₂S), which is a weak acid. So, S²⁻ is a pretty decent base. It’s definitely more inclined to grab protons than, say, chloride. It’s like, "Alright, I’m feeling a bit negative, and a proton sounds like just the thing to balance me out!" It's got a concentrated negative charge, making it more available for protonation.
Getting warmer! Let's look at anions from weak acids. Consider acetic acid (CH₃COOH). Its anion is acetate (CH₃COO⁻). Acetate is a considerably stronger base than our earlier friends. Its conjugate acid, acetic acid, is a weak acid. This means acetate really wants to get its hands on a proton to become that weak acid again. It’s like, "Proton? Yes, please! I've been waiting for you!" You can really feel its desire for that positive charge.
And then there's carbonate (CO₃²⁻), the conjugate base of carbonic acid (H₂CO₃), which is a weak acid. Carbonate is a stronger base than acetate. It’s got a real hankering for protons. It’s like, "Hurry up, proton, don’t be shy!" Its charge is less delocalized compared to the really weak bases, making it more readily available to accept a proton.
We're nearing the top of the heap now, where the real proton enthusiasts reside. Think about hydroxide (OH⁻). This is the conjugate base of water (H₂O), which is a ridiculously weak acid. So, OH⁻ is a pretty strong base. It's like, "Proton, my dear, come here! Let's make some beautiful, neutral water molecules!" It’s a classic strong base for a reason – it's really good at picking up protons.
And who comes next? Well, there are a couple of anions that really embody the "proton fanatic" spirit. For example, consider the anions derived from polyprotic weak acids. Like phosphate (PO₄³⁻), the conjugate base of phosphoric acid after it’s lost all three protons. PO₄³⁻ is a very strong base because its conjugate acid, HPO₄²⁻, is a weak acid. It's just begging for a proton to neutralize its triple negative charge. It's like a kid asking for candy. "Please, please, please!"
Finally, at the absolute peak of basicity, are the conjugate bases of incredibly weak acids, like ammonia (NH₃). Its conjugate base is amide (NH₂⁻). This anion is extremely basic. It is so unstable on its own that it will snatch a proton from almost anything it can. It’s like, "Proton? WHERE ARE YOU? I NEED YOU LIKE I NEED AIR!" It’s a powerhouse of basicity, ready to react with anything to get that proton.
And let's not forget the really, really, really strong bases. Think about anions derived from very, very weak Brønsted-Lowry acids, like hydrides (H⁻) or even some organometallic species. For instance, the anion of hydrogen (H⁻) is incredibly basic. Its conjugate acid would be H₂, which is not an acid at all in the traditional sense. It's like, "Proton? My life's purpose is to find a proton!" These are the ultimate proton-seekers.
The Grand Finale: Ranked!
So, to recap, from least basic to most basic, here's the order:
1. Perchlorate (ClO₄⁻) - From perchloric acid. Super stable, barely wants a proton.
2. Bisulfate (HSO₄⁻) - From sulfuric acid. Still pretty chill.
3. Chloride (Cl⁻) - From hydrochloric acid. Another one that’s like, "Meh, protons."
4. Nitrate (NO₃⁻) - From nitric acid. Same vibe as chloride, thanks to resonance.
5. Hydrogen Phosphate (HPO₄²⁻) - From phosphoric acid. Starting to get a little more interested.

6. Sulfide (S²⁻) - From hydrogen sulfide. Definitely more of a proton seeker.
7. Acetate (CH₃COO⁻) - From acetic acid. Now we're talking! It actively wants a proton.
8. Carbonate (CO₃²⁻) - From carbonic acid. A real proton enthusiast.
9. Hydroxide (OH⁻) - From water. A classic strong base, always ready to react.
10. Phosphate (PO₄³⁻) - From phosphoric acid. Begging for protons!
11. Amide (NH₂⁻) - From ammonia. An extreme proton fanatic.
12. Hydride (H⁻) - From hydrogen gas. The ultimate proton magnet!
So there you have it! It’s all about how stable they are and how much they’re willing to play proton-grab with a proton. Pretty neat, right? It’s like a chemical game of musical chairs, but with protons.
Next time you’re sipping your coffee, you can ponder the basicity of whatever sugar you’re adding. Just kidding! (Mostly.) But seriously, understanding these anion vibes really helps you understand how different chemicals will behave. It’s all about their desire for protons. Who knew chemistry could be so dramatic?
