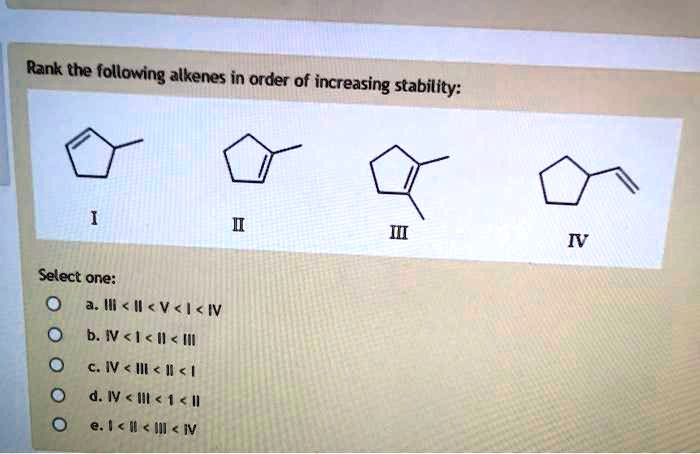
Rank The Following Alkenes In Order Of Increasing Stability
Hey there, science curious folks! Ever wonder why some things in life just… hang around longer than others? Like your favorite comfy socks that seem to magically reappear in the laundry, or that one really stubborn stain that just won't quit? Well, it turns out the universe has its own version of this, and it applies to the little building blocks of many things around us – even things you might not think about, like the invisible world of molecules.
Today, we're going to chat about something called "alkenes." Don't let the fancy name scare you! Think of them as molecules that have a special kind of double bond, like a little handshake between two atoms that's a bit more committed than a single handshake. These double bonds are pretty neat, and they have a big impact on how stable these molecules are. Stability, in this case, basically means how content and unbothered a molecule is. A more stable molecule is less likely to go on an adventure and change into something else. It's like a well-rested cat versus a cat that's just had a triple espresso – one is chilling, the other is ready to climb the curtains.
So, why should you even care about the stability of these tiny alkenes? It’s actually pretty cool! Understanding this helps chemists, who are basically like the chefs of the molecular world, figure out how to make new medicines, better plastics, or even the flavors in your favorite snacks. It’s all about knowing which ingredients are going to play nicely together and which ones might get a bit… feisty.
Let's imagine we've got a little lineup of alkenes, and we're going to rank them from the least happy campers to the ultimate chill masters. Think of it like a "Most Likely to Stick Around" competition. The less stable ones are the ones that are a bit fidgety, always looking for a chance to change. The more stable ones are the ones that are just perfectly content, like they’ve found their spot on the sunny couch and aren’t moving.
The Least Stable Guys: The Twos and Threes
Alright, let’s start with our least stable friends. These are generally the alkenes with the fewest "extra hands" to help them out. Imagine you've got a party, and some guests are just standing around, a bit awkward. These are our less stable alkenes.
At the very bottom, we’ve got things that look like a single little connection, but with that special double bond. Think of it as the simplest possible handshake. These guys are a bit exposed, a bit vulnerable. They're not unhappy, per se, but they're definitely looking around for a better deal, a more stable situation. They’re like the person who agrees to help move furniture on a Saturday morning without realizing how heavy the sofa is.
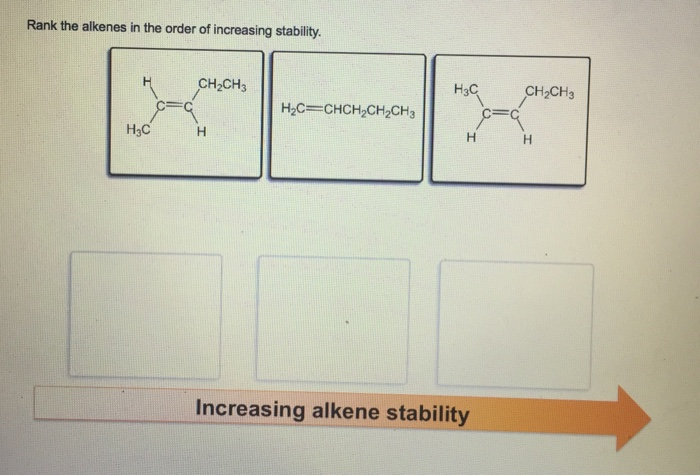
Then we have the ones with just a little bit of company. Imagine you’ve got a couple of friends nearby. This extra company, these little groups of atoms called "alkyl groups" (don't worry too much about the name, just think of them as helpful buddies), actually makes the alkene a tiny bit more stable. It’s like having a friend to share the awkward silence with at the party. They offer a little bit of support, a little bit of protection from the outside world.
The Middle Ground: The Four and Fives
As we move up the ladder, things get a bit cozier. The alkenes here have more of those helpful "alkyl groups" hanging around. Think of it like more friends joining the party. The more friends you have, the more robust the social group becomes, right? It’s harder for one person to feel isolated or look for trouble when there's a good crowd.
These middle-ground alkenes are pretty content. They're not actively seeking out a change, but they're not exactly "rock solid" either. They're like someone who's found a good seat at the party, maybe has a decent conversation going, but is still open to mingling if a really interesting person walks by.
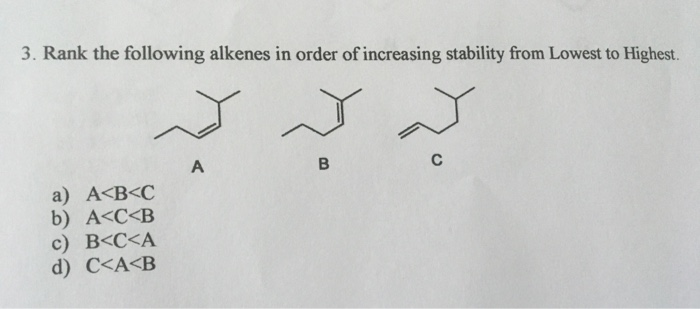
The more of these helpful buddies an alkene has, the more it spreads out the "electron density" of that double bond. Think of it like a group of people holding hands in a circle. The more people, the more everyone is connected and supported. This makes the double bond less eager to react with anything new. It's already got its hands full, in a good way!
The Ultimate Chill Masters: The Sixes and Beyond
Now, let's talk about the VIPs of alkene stability. These are the ones that are practically melting into their comfy chairs. Why are they so chill? Because they have the most of those helpful alkyl groups surrounding their double bond. Imagine a massive, super-fun party where everyone knows everyone, there's great music, and plenty of snacks. Nobody's going anywhere!
These alkenes are the most stable because those extra alkyl groups act like little shields. They protect the double bond from getting into mischief. It’s like having a whole entourage following you around – not many people are going to try and pick a fight with you then, are they?
The more these alkyl groups are attached, the more they "donate" electron density to the double bond, making it even more content and less reactive. It's like having a constant stream of positive affirmations and good vibes flowing into the molecule. They're just happy to exist as they are, and they don't need to change anything to feel better.
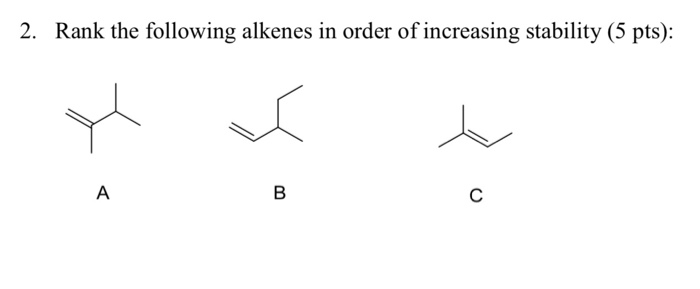
Putting It All Together: The Grand Ranking!
So, if we were to line them up from least stable to most stable, it would look something like this:
Least Stable: The alkene with the fewest helpful buddies (think of it as having just one or two little attachments).
Getting Cozier: The alkene with a few more buddies (two or three attachments).
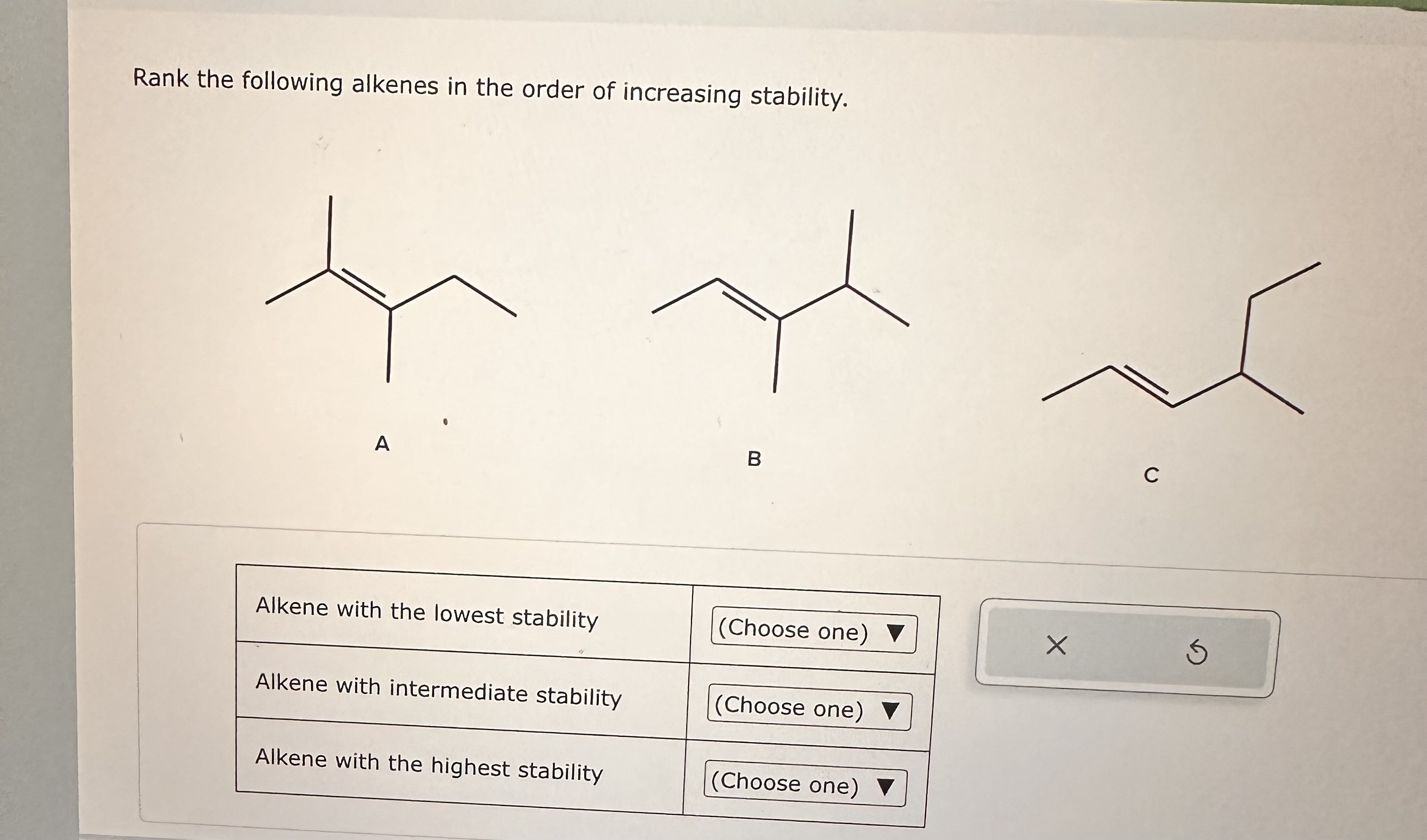
Pretty Content: The alkene with a good number of buddies, feeling supported (four or five attachments).
Most Stable: The alkene with a whole crew of buddies, practically unshakeable (multiple attachments all around).
It’s really all about how many of those little "alkyl group" friends are hanging out with the double bond. The more friends, the happier, and the more stable the alkene is. It’s like the universal law of parties: more friends, more fun, more stability!
Knowing this might seem a bit abstract, but it’s the foundation for so much. When scientists are designing a new drug, they need to know how stable the molecules are so they don't break down before they can do their job. When they're creating new materials, they want them to be stable enough to last. So, the next time you hear about molecules and their stability, you can nod knowingly, thinking about those little parties and the importance of having good friends – even if those friends are made of atoms!
