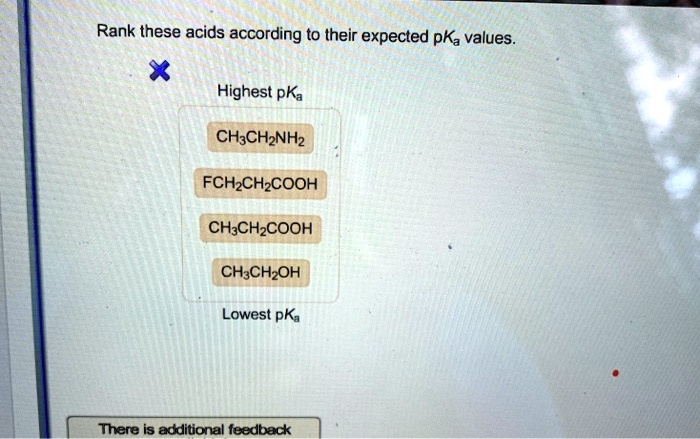
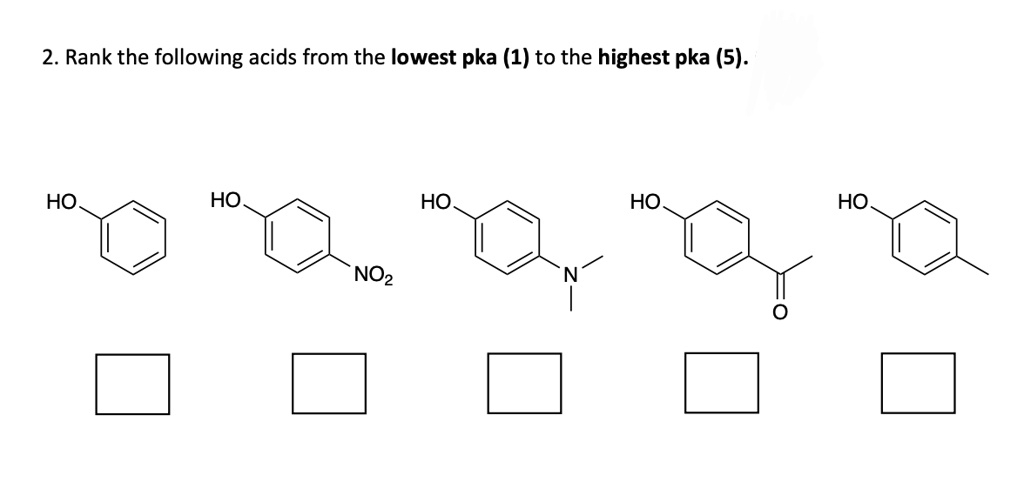
Rank The Following Acids From Lowest Pka To Highest Pka.

Hey there, curious minds! Ever found yourself staring at a bottle of something fizzy and wondering, "What's making this tingle?" Or maybe you've accidentally splashed some lemon juice on your skin and felt that little sting. Well, today, we're diving into the wonderfully wacky world of acids, specifically their acidity strength. And don't worry, this isn't going to be a dry, lab-coat-wearing lecture. Think of it more like a friendly chat over a cup of tea, but with a splash of science!
We're going to talk about something called the pKa. Now, that might sound a bit intimidating, like a secret handshake for chemists. But really, it's just a handy way to measure how easily an acid is willing to share its "proton." You can think of a proton as a tiny, eager little buddy that an acid molecule is holding onto. The lower the pKa, the more willing that acid is to let its proton buddy go. It's like a shy friend at a party who's already looking for an excuse to leave, versus someone who's glued to the dance floor.
Why should you care about this? Well, understanding pKa helps us figure out why some things are sour, why some cleaning products are super strong, and even how our own bodies manage their delicate chemical balance. It's like knowing the secret ingredient that makes your favorite recipe taste just right, but for the chemical world!
Let's Get Our Acids Lined Up!
Imagine you have a lineup of friends, and you're asking them to lend you a dollar. Some will hand it over instantly, no questions asked. Others might hem and haw, ask what it's for, or need a really good reason. That's kind of how acids and their pKa values work. We've got a few common acids here, and we're going to rank them from the one that's super eager to share its proton (lowest pKa) to the one that's a bit more possessive (highest pKa).
1. Hydrochloric Acid (HCl): The "Here, Take It!" Friend
When we talk about the strongest acids, Hydrochloric Acid often comes to mind. Its pKa is a whopping negative number, around -6.3. This means it's practically begging to get rid of its proton. Think of it like your friend who always has a spare dollar for you, no matter what. You just have to think about needing one, and poof, it's there!

Hydrochloric acid is the stuff that gives stomach acid its punch. It’s what helps us break down our food. Imagine your stomach is a busy restaurant, and HCl is the super-efficient waiter who clears the plates (food) away lightning fast. It’s so strong that even a diluted solution can cause damage if you're not careful – kind of like giving that dollar-lending friend too much attention and they end up broke!
2. Sulfuric Acid (H₂SO₄): The Generous, But Slightly More Thoughtful Friend
Next up, we have Sulfuric Acid. Its first proton has a pKa of around -3. Now, it's still super eager to share, but maybe just a tiny bit less frantic than HCl. It's like that friend who's always ready to lend a dollar, but might pause for half a second to ask, "You good?" before handing it over.
Sulfuric acid is another industrial powerhouse. It’s used in making fertilizers, batteries, and even in oil refining. It’s the unsung hero in many processes that give us the stuff we use every day. It’s a strong acid, and while its pKa is slightly higher than HCl, it's still incredibly potent and needs respect. Think of it as a very dependable loan shark – they'll give you the money, but you gotta be careful!
3. Phosphoric Acid (H₃PO₄): The "Let Me Check My Pockets" Friend
Phosphoric acid is a bit more interesting because it has multiple protons it can share, and each one comes off with a different level of enthusiasm. The first proton has a pKa of around 2.15. This is a significant jump from our previous acids. Now, our friend is less like an instant dispenser of cash and more like someone who pats their pockets, checks their wallet, and then carefully counts out the dollar.
You find phosphoric acid in a lot of places, including some sodas! That slight tang you taste in a cola? That's partly thanks to phosphoric acid. It adds a bit of zest without being overwhelmingly harsh. It’s also used in rust removal and as a food additive to control acidity. It's still a good sharer, but it takes a little more nudging than HCl or sulfuric acid.
4. Acetic Acid (CH₃COOH): The "Are You Sure You Need It?" Friend
Ah, acetic acid! This is the star ingredient in vinegar, with a pKa of around 4.76. Now we're talking about an acid that's much more reserved. Imagine asking this friend for a dollar, and they sigh, look at you with a mixture of concern and mild exasperation, and say, "Okay, but what is this really for?"
Acetic acid is what gives vinegar its distinctive smell and taste. It's used in pickling, cleaning, and in salad dressings. It's mild enough that we can consume it (in diluted form, of course!) and even enjoy it. It’s a much more approachable acid, willing to share its proton, but only when there’s a good reason, and perhaps after a bit of negotiation.
5. Carbonic Acid (H₂CO₃): The "Let's Think About This" Friend
Finally, let's look at carbonic acid, with a pKa of around 6.35. This is the acid that forms when carbon dioxide dissolves in water. Remember that tingle from our fizzy drinks earlier? That's carbonic acid at work. It's a very weak acid compared to the ones we've discussed so far.

Think of carbonic acid as that friend who’s super laid-back. When you ask for a dollar, they might say, "Uh, maybe? Let me see if I even have cash on me. And are you sure you can't just, like, ask someone else?" It readily forms and readily breaks down. It’s in our blood, helping to maintain a crucial pH balance, and it’s responsible for that delightful effervescence in your soda. It’s a gentle sharer, and its ability to readily transform makes it a key player in many biological and environmental systems.
So, What's the Big Deal?
As you can see, our acids go from being super generous and eager to share their proton (low pKa) to being much more hesitant (high pKa). This difference in willingness to donate a proton is what dictates how strong an acid is. Strong acids react more readily and can do things like dissolve metals or cause significant burns. Weaker acids are more manageable and are found in many of the foods and drinks we consume daily.
Understanding pKa helps scientists and engineers predict how chemicals will behave. It's essential for everything from developing medicines to designing industrial processes. For us everyday folks, it gives us a cool little insight into the chemistry of the world around us. So, the next time you taste something sour or see something bubbling, you'll have a little secret knowledge about the proton-sharing habits of those sneaky acids!
