Rank The Elements From Highest To Lowest Ionization Energy
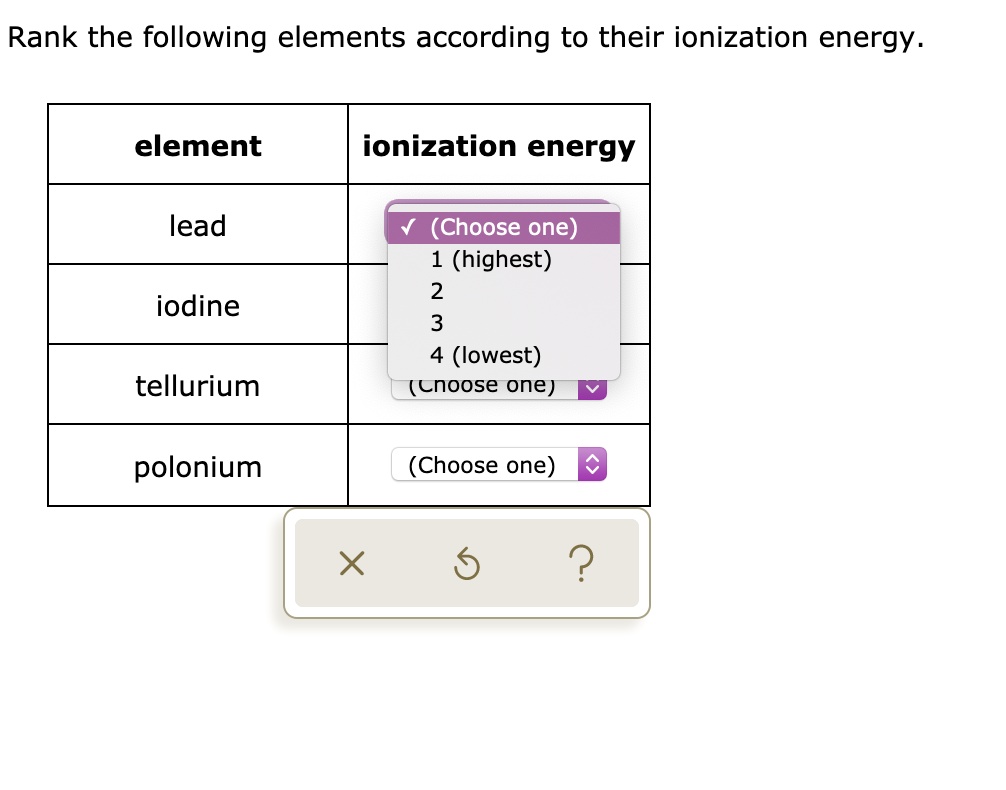
Alright, so you wanna talk about ionization energy? Awesome! It's basically how much effort it takes to steal an electron from an atom. Think of it like trying to snatch the last cookie from a cookie jar. Some atoms are super stingy with their electrons, while others are practically throwing them around.
Why is this fun? Because elements are like tiny little characters with personalities! And their ionization energy tells us a lot about their vibe. We're gonna rank 'em, from the electron-hoarders to the electron-givers. It's a wild ride through the periodic table.
The Electron-Snatching Superstars
So, who's the ultimate electron-snatcher? Drumroll please... it's the noble gases! These guys are like the introverts of the atomic world. They've got their electron shells all snug and complete, and they're NOT looking to share. Trying to rip an electron from them? You're gonna have a bad time. It takes a ton of energy. Imagine asking a grumpy cat for its favorite toy. Nope. Not happening.
Helium, Neon, Argon... they're all in this club. They're so stable, they rarely bother with other elements. They're the ultimate prize if you're looking for something that doesn't react much. Think of them as the cool, aloof celebrities of chemistry. They've got it all, and they're not giving it up.
The Halogen Hustle
Right behind the noble gases, we've got the halogens. Fluorine, Chlorine, Bromine... these guys are super eager to grab an electron. They're just one electron away from that cozy, complete shell like the noble gases. So, they're always on the hunt. They're the energetic teenagers of the periodic table, always looking for that one missing piece to complete their ensemble.
Fluorine is the absolute champion here. It's the most electronegative element, meaning it's the biggest electron hog. Seriously, it's like a black hole for electrons. It will yank them right out of other atoms. It’s kinda terrifying, but also incredibly cool. If you’re building something that needs to be super reactive, fluorine is your go-to.
The Alkali Metal Giveaway
Now, let's flip the script. Who are the most generous with their electrons? The alkali metals! Lithium, Sodium, Potassium... these guys are practically begging you to take their outermost electron. They've got just one electron hanging out in their outer shell, and it's not feeling very secure. It's like that one friend who always offers to buy the first round of drinks. They're just happy to get rid of it!
Sodium, the stuff in your table salt, is a classic example. It's so eager to lose that electron that it reacts violently with water. Like, boom! Firework show! It’s not exactly a chill element, but it’s definitely generous. This low ionization energy is what makes them so reactive. They're the life of the atomic party, always ready to mingle and share.
The Alkaline Earth Metal Buddies
Close cousins to the alkali metals are the alkaline earth metals. Beryllium, Magnesium, Calcium... they're also pretty chill about giving up electrons. They’ve got two in their outer shell, and they’re usually happy to let one or both go to achieve a more stable configuration. They're not quite as desperate as the alkali metals, but they're definitely not hoarders.
Calcium, the stuff in your bones, is a good example. It's essential for so many biological processes because it's willing to participate. It's like the supportive friend who's always there to lend a hand (or an electron). Their ionization energies are a bit higher than the alkali metals, but still pretty low on the grand scale of things.
The Middle Ground Explorers
As we move towards the center of the periodic table, things get a little more... nuanced. The transition metals are a mixed bag. Some are more stingy, some are more generous, and it can change depending on what they're doing. They're like the teenagers who are still figuring themselves out. They've got multiple electron shells to play with, so things can get a bit complicated.
Think of Iron, Copper, Gold. They have interesting ionization energy patterns. Gold, for instance, is pretty unreactive, partly due to its relatively high ionization energy. It's like the "too cool for school" element. But then you have metals like Iron, which are much more reactive and prone to oxidation. They're the adaptable ones, the chameleons of the atomic world.

The Nonmetal Neighbors
The nonmetals, other than the halogens and noble gases, fall somewhere in between. Elements like Carbon, Nitrogen, Oxygen. They're not as aggressive electron-snatchers as the halogens, but they're definitely not giving them away like the alkali metals. They're the workhorses, the ones that form the backbone of so many compounds.
Carbon, the basis of all life, has a moderate ionization energy. It's perfectly happy to share electrons, forming covalent bonds. It's the ultimate team player. Oxygen, while more electronegative than carbon, still participates in a wide range of bonding. They’re the dependable folks, ready to form bonds and build complex structures.
The Bottom of the Barrel
And who’s at the very bottom, the least likely to let go of an electron? The lanthanides and actinides. These are the f-block elements, tucked away at the bottom. They have a lot of electrons, and the ones on the outside are pretty well shielded by all the inner ones. It takes a serious amount of energy to dislodge them.
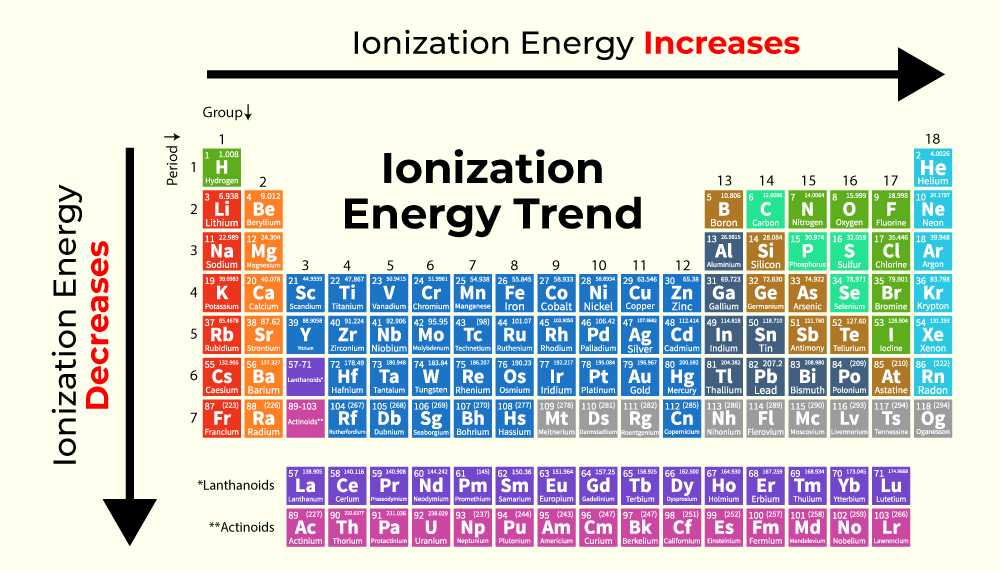
These elements are often radioactive and pretty obscure. They're like the reclusive billionaires of the periodic table. They've got all the stuff, and they're not about to hand it over easily. They’re the ultimate electron hoarders, requiring immense power to even consider a transfer. It’s a fascinating corner of the periodic table, full of mystery and heavy elements.
So, What's the Point?
Why should you care about this electron-snatching saga? Because ionization energy is the secret sauce behind chemical reactions! It tells us why certain elements bond, how they react, and what kind of compounds they'll form. It's the fundamental driver of chemical behavior.
It’s like understanding a person’s motivation. Are they eager to share? Are they defensive? Are they just looking for stability? This is what ionization energy tells us about atoms. It’s a fun way to see the periodic table not just as a chart, but as a collection of characters with unique traits and personalities. So next time you see an element, think about its ionization energy. It's a tiny detail that unlocks a world of chemical fun!
