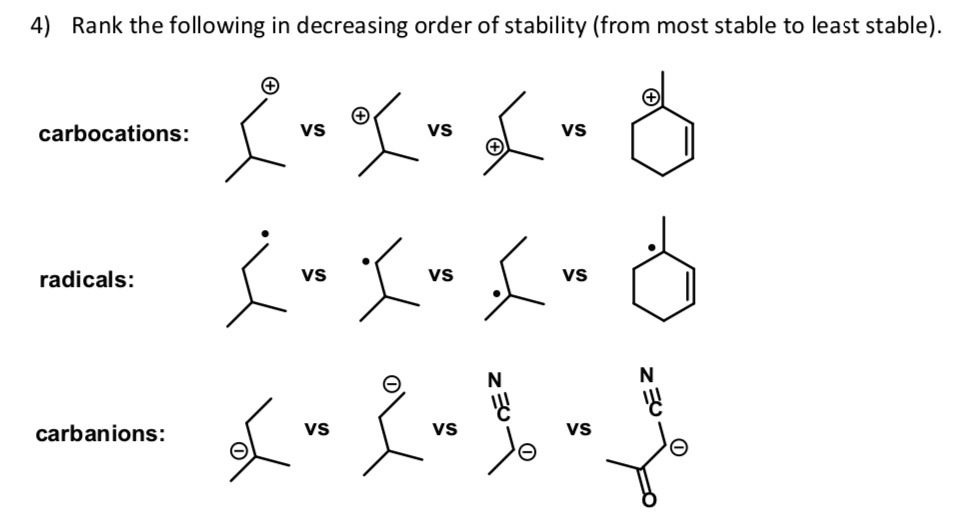
Rank The Carbocations In Order Of Decreasing Stability
Ever wondered what makes some molecules more eager to react than others? It's a bit like trying to understand why some people are more outgoing and others more reserved. In the fascinating world of chemistry, we have little critters called carbocations, and they're essentially molecules with a bit of a positive charge. Now, not all carbocations are created equal. Some are quite happy and stable, while others are a bit more… fidgety. Learning to rank them by their stability is like learning to read the mood of these chemical beings!
Why should you care about ranking carbocations? Well, it's not just about memorizing lists. Understanding carbocation stability is a fundamental building block in organic chemistry. It helps us predict how chemical reactions will proceed, which is incredibly useful if you're pursuing a career in medicine, materials science, or even just trying to understand how that new medication or plastic is made. Think of it as a detective's tool, helping you figure out the "whodunit" of a chemical transformation. The benefits extend to designing new reactions, optimizing existing ones for efficiency, and even understanding the degradation pathways of materials.
In the realm of education, this concept is a cornerstone. Students learning organic chemistry will encounter carbocation stability constantly. It's the key to understanding reaction mechanisms for everything from simple additions to complex rearrangements. In daily life, while you might not be explicitly ranking carbocations, the principles underpin technologies you interact with every day. The synthesis of polymers in plastics, the creation of pharmaceuticals that improve our health, and even the way fuels burn efficiently all rely on the predictable behavior of molecules, a behavior often dictated by the stability of intermediate species like carbocations.
So, how do we actually rank them? It boils down to a few simple rules, and the most important factor is the presence of electron-donating groups. Think of these groups as little "huggers" that can share their electron density with the positively charged carbon, essentially calming it down and making it more stable. The more groups we can attach, the happier the carbocation. This leads to a general hierarchy: tertiary carbocations (where the positive carbon is attached to three other carbons) are the most stable, followed by secondary (attached to two other carbons), and then primary (attached to one other carbon). Methyl carbocations (where the positive carbon is attached to nothing else) are the least stable.
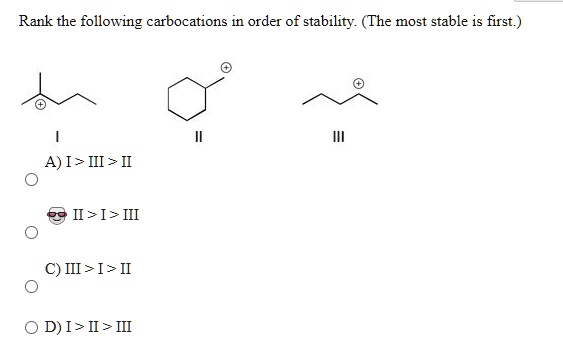
Another important factor is resonance. If the positive charge can be spread out over multiple atoms through delocalization of electrons, the carbocation becomes significantly more stable. Think of it as sharing the burden of the positive charge, making it less intense for any single atom. So, allylic and benzylic carbocations, which can undergo resonance, are often more stable than even tertiary carbocations.
Ready to explore this yourself? It's simpler than you think! In your studies, pay close attention to diagrams illustrating reaction mechanisms. You'll see these carbocations appearing as fleeting intermediates. Try to identify the type of carbocation formed (primary, secondary, tertiary) and look for opportunities for resonance. You can even find online resources and quizzes that test your ability to rank carbocations. It's a bit like a puzzle, and the more you practice, the more intuitive it becomes. So, the next time you encounter a chemical reaction, remember to look for those positively charged little guys and see how stable they are – it's a fun way to peek behind the curtain of chemical transformations!
