Rank The Atoms Below In Order Of Increasing Electronegativity.

Ever wondered what makes some elements a little clingy when it comes to electrons, while others are more laid-back? Well, buckle up, because we're diving into the fascinating world of electronegativity! It might sound like a fancy science term, but trust us, it's one of the coolest concepts in chemistry, and understanding it can unlock a whole new level of appreciation for the building blocks of our universe. Think of it as the "electron-grabbing power" of an atom – who's the ultimate tug-of-war champion in the atomic arena? It's a concept that’s not just popular among chemists and scientists, but it’s also surprisingly relevant to everyday life, from the way water sticks to your glass to how medicines are designed. So, let's get ready to have some fun and demystify this electrifying idea!
The "Electron-Grabbing" Game
So, what exactly is this electronegativity we're talking about? In simple terms, it's a measure of how strongly an atom in a chemical compound attracts the shared electrons towards itself. Imagine two atoms forming a bond, sharing their precious electrons like a pair of kids sharing a toy. Electronegativity tells us which kid is more likely to snatch the toy and keep it closer!
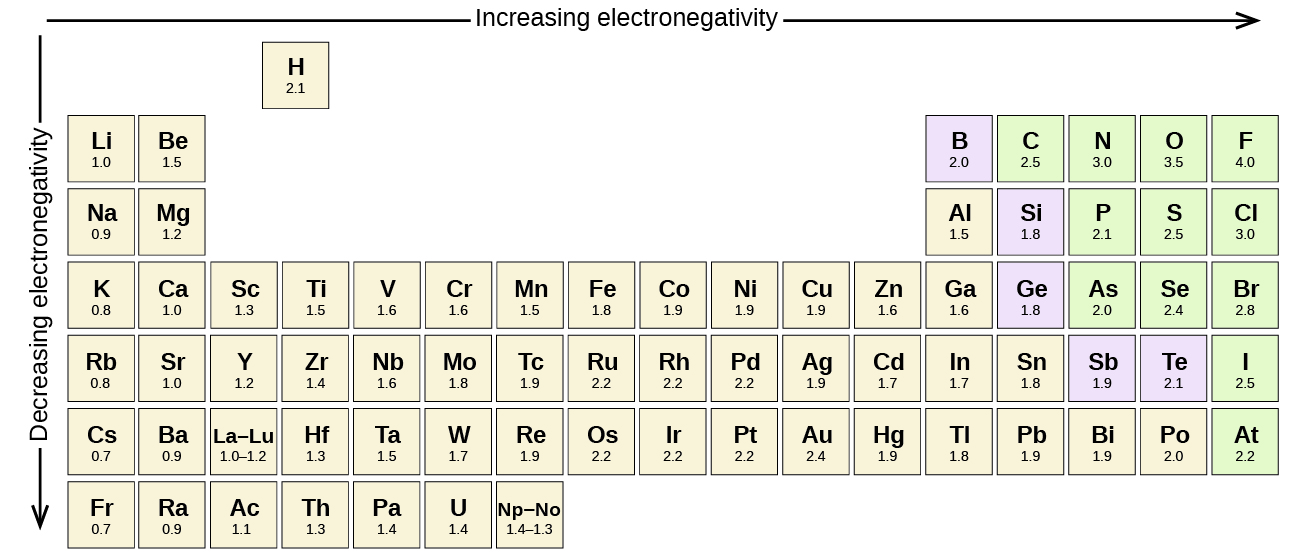
This "electron-grabbing" power isn't some random quirk; it follows a predictable pattern across the periodic table. Generally, as you move across a row (period) from left to right, electronegativity increases. This is because the number of protons in the nucleus increases, creating a stronger positive pull on the electrons. Conversely, as you move down a column (group), electronegativity decreases. This happens because the outer electrons are further away from the nucleus and are shielded by inner electron shells, weakening the pull.
Why is this so useful? Well, understanding electronegativity helps us predict how atoms will behave when they come together to form molecules. It's the key to understanding the type of chemical bonds that form – whether they'll be ionic (where electrons are completely transferred) or covalent (where electrons are shared). This, in turn, dictates the properties of the resulting substances. Will a substance dissolve easily in water? Will it conduct electricity? Will it have a high boiling point? The answers often lie in the differences in electronegativity between the atoms involved.
Think of electronegativity as a superpower that atoms possess – some have a mighty grip, while others have a gentle hold. Understanding this superpower helps us predict how they'll interact!
Electronegativity Series in Increasing Order
The Importance of Knowing Who's Who
The benefits of grasping electronegativity are vast and touch upon many areas:
- Predicting Chemical Bonds: As mentioned, it's the bedrock of predicting whether a bond will be ionic, polar covalent, or nonpolar covalent. This is fundamental to understanding how molecules are formed.
- Understanding Molecular Polarity: When atoms with different electronegativities bond, they create a separation of charge within the molecule, making it polar. Think of water (H₂O) – its polarity is a direct result of the high electronegativity of oxygen compared to hydrogen. This polarity is crucial for so many biological processes, like acting as a universal solvent.
- Explaining Chemical Reactivity: The distribution of electrons, influenced by electronegativity, directly affects how readily a molecule will participate in a chemical reaction.
- Designing New Materials: From pharmaceuticals to advanced materials, chemists use electronegativity to design molecules with specific properties. For example, in drug design, understanding how a drug molecule interacts with biological targets often involves considering its polarity and electron distribution.
- Appreciating the Natural World: So many natural phenomena, from the way plants absorb water to the electrical signals in our own bodies, are influenced by the electronegative properties of the elements involved.
So, are you ready to put your knowledge to the test and see how these specific atoms stack up? We're going to rank them in order of increasing electronegativity. This means we'll start with the atom that has the weakest "electron-grabbing" power and work our way up to the one with the strongest grip. It's like a friendly competition to see who's the real electron magnet!
The Ranking Challenge
Let's take a look at the atoms you've provided and see where they fall on the electronegativity spectrum. We'll be using the Pauling scale, which is the most commonly used scale for measuring electronegativity. Remember, higher numbers mean a stronger pull on electrons.

Here are the atoms we’ll be ranking:
- Lithium (Li)
- Carbon (C)
- Nitrogen (N)
- Oxygen (O)
Let's break them down and figure out their positions. We need to consider their location on the periodic table and the general trends we discussed.

The Gradual Climb
We start with Lithium (Li). Lithium is in the first group (alkali metals) and the second period. Alkali metals are known for having very low electronegativity because they tend to lose their single valence electron very easily to achieve a stable electron configuration. They're not interested in grabbing electrons; they're all about giving them away!
Next up, we have Carbon (C). Carbon is in the second period, but it's in group 14. It's located to the right of Lithium. As we move across a period, electronegativity generally increases. Carbon is a crucial element in organic chemistry and often forms covalent bonds, meaning it shares electrons. However, its electron-grabbing power is moderate compared to some other elements.
Now, let's consider Nitrogen (N). Nitrogen is in the second period and group 15. It's located to the right of Carbon. Following the trend, we expect its electronegativity to be higher than Carbon's. Nitrogen is a key component of proteins and DNA, and its ability to form strong bonds is partly due to its moderate-to-high electronegativity.
.PNG)
Finally, we have Oxygen (O). Oxygen is in the second period and group 16, directly to the right of Nitrogen. Oxygen is one of the most electronegative elements, second only to fluorine. Its high electronegativity is why it readily forms bonds with many other elements, and it plays a vital role in respiration and combustion. It's a real electron hog!
The Grand Finale: The Ranking!
Putting it all together, here are the atoms ranked in order of increasing electronegativity:
- Lithium (Li) - The electron donor, with the lowest electronegativity.
- Carbon (C) - Moderately electronegative, a master of sharing.
- Nitrogen (N) - Significantly more electronegative than Carbon, a stronger pull.
- Oxygen (O) - One of the most electronegative elements, a powerful electron attractor.
There you have it! From the electron-shedding Lithium to the electron-snatching Oxygen, we've seen how electronegativity changes across the periodic table. It's a simple concept with profound implications, making chemistry not just a subject to study, but a language to understand the world around us. So, the next time you see a molecule, you can start to guess who's got the stronger grip on those electrons!