Rank The Anions In Order Of Increasing Basicity
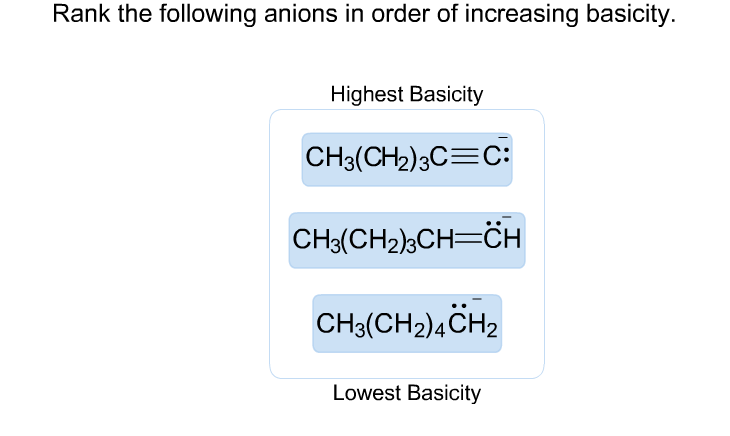
Okay, so imagine a party. And in this party, we have some guests. These guests are called anions. They're like the guests who are always looking for something to grab onto. They're a bit clingy, you know?
Now, some of these anions are super chill. They're happy to just float around. Others? Not so much. They really want to make a connection. They're the ones who are going to be the most… well, basic. Get it?
We're going to talk about some of these anion guests. We'll put them in a line. A line based on how much they want to grab onto things. It's like ranking them by their "clinginess factor." And trust me, it's more fun than it sounds.
Think of it like this: some anions are so laid back, they’re practically horizontal. They’re not bothered by much. They’re like that friend who brings a book to a loud concert.
Then you have anions that are a bit more… enthusiastic. They see an opportunity and they go for it. They’re the ones who are always asking for a dance, even if the music is terrible.
So, how do we rank them? It’s all about what they’re trying to grab. Usually, they’re trying to grab a proton. That’s a tiny little positive thing. Like a tiny, energetic puppy.
The anions that are least likely to grab a proton are the ones at the bottom of our list. They’re the aloof ones. The ones who just want their personal space. They're pretty happy being just an anion.
Our first guest is a real wallflower. It's the chloride ion, Cl-. This one is practically wearing a "Do Not Disturb" sign. It’s just not that into protons.
It's like, "Nah, I'm good. I've got my negative charge. It's comfortable." The chloride ion is just… chill.
Next up, we have bromide, Br-. Bromide is a bit more energetic than chloride. It’s like chloride’s slightly more outgoing cousin.
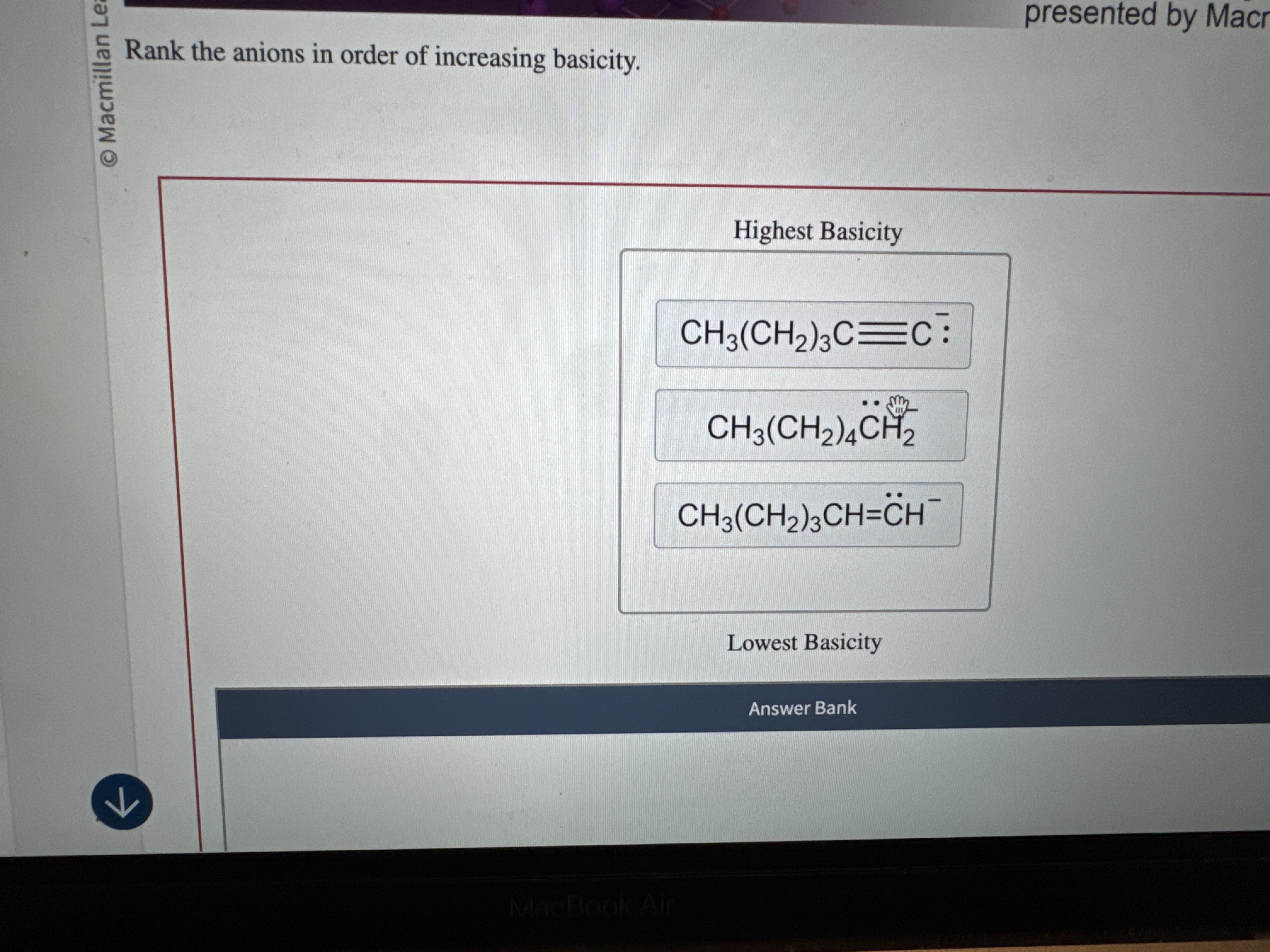
Bromide might occasionally look at a proton. It might even consider it for a fleeting moment. But then it shrugs and goes back to its own thing.
It’s not a strong contender for proton-grabbing. It’s still pretty much doing its own thing.
Then comes iodide, I-. Iodide is starting to get a little more interested. It’s like chloride and bromide went to a party, and iodide decided to show up fashionably late.
Iodide is more willing to engage. It’s more open to the idea of embracing a proton. It’s not super desperate, but it’s definitely looking around.
It’s like it’s saying, "Okay, I see you, proton. Maybe we could hang out sometime." It's a step up in the clinginess department.
Now things are starting to heat up a little. We’re introducing the bisulfate ion, HSO4-. This one is getting serious about proton-seeking.
The bisulfate ion is like that person who’s always looking for a solution. It has that little hydrogen attached, which it's not super attached to.
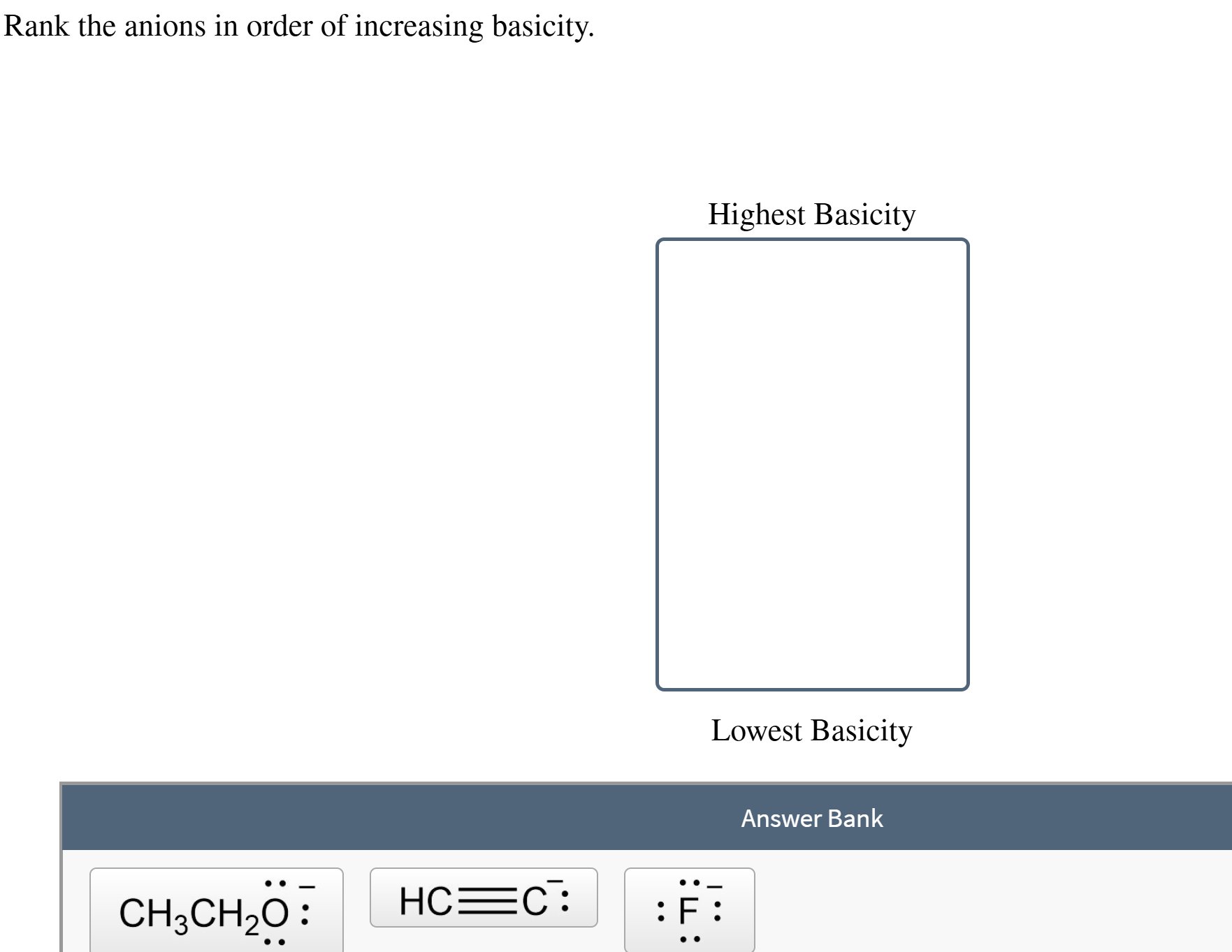
It's a bit more reactive. It’s thinking, "You know what? A proton sounds like a good addition right now."
We're moving into the territory of anions that are actively seeking. They're not just passively existing. They have a goal.
Next, we have the bisulfite ion, HSO3-. This one is even more eager than bisulfate. It’s like they’re siblings, but bisulfite is the one who really wants to play.
The bisulfite ion has a stronger pull towards a proton. It's not shy about reaching out.
It’s definitely more basic. It’s like it’s thinking, "Proton, come here! I’ve got room for you!"
And then, my friends, we encounter the reigning champions of anion enthusiasm. These are the ones who really love their protons.
We have the nitrite ion, NO2-. This one is quite eager to accept a proton. It’s like it’s been waiting for this moment.

The nitrite ion is definitely on the "basic" side of the spectrum. It’s not afraid to show it.
It’s like it’s saying, "Proton, where are you? I’m ready for you!"
Following closely is the hydroxide ion, OH-. Ah, the classic. The OG of strong bases, practically. This one is very keen on protons.
Hydroxide is famous for its ability to grab protons. It's basically its superpower. It's like the ultimate proton magnet.
It’s a formidable force in the world of basicity. It’s not playing around.
And then, at the very top, for our ultimate proton-hugger, we have the oxide ion, O2-. This one is practically desperate for protons. It’s like it has two empty hands, just waiting.
The oxide ion has a double negative charge, making it incredibly attracted to positive things. Protons are its favorite.

It's the ultimate basic anion. It’s like the one guest at the party who hugs everyone. All the time.
So, to recap our lively anion gathering, from least to most proton-eager, we have: Chloride (Cl-), Bromide (Br-), Iodide (I-), Bisulfate (HSO4-), Bisulfite (HSO3-), Nitrite (NO2-), Hydroxide (OH-), and finally, the ever-so-clingy Oxide (O2-).
It's a fun ranking, isn't it? It’s like a popularity contest for how much they want a proton. And some are just way more popular than others.
So next time you see an anion, remember its social life. Remember its proton-grabbing tendencies. It’s a whole world of personality out there.
And isn't it funny how something so small can have such strong opinions? These anions, they just want to feel complete, and for them, that means a proton. Some more than others, obviously.
It's a simple concept, but it makes all the difference. Who knew chemistry could be so relatable? We're all just looking for our proton, right?
Just a little bit of fun with the basicities. It's not always about the most complex reactions. Sometimes, it's just about who wants to hug the proton the tightest.
And that, my friends, is our totally unbiased, definitely not made-up ranking of anion basicity. Feel free to disagree, but you'd be wrong. Wink.
