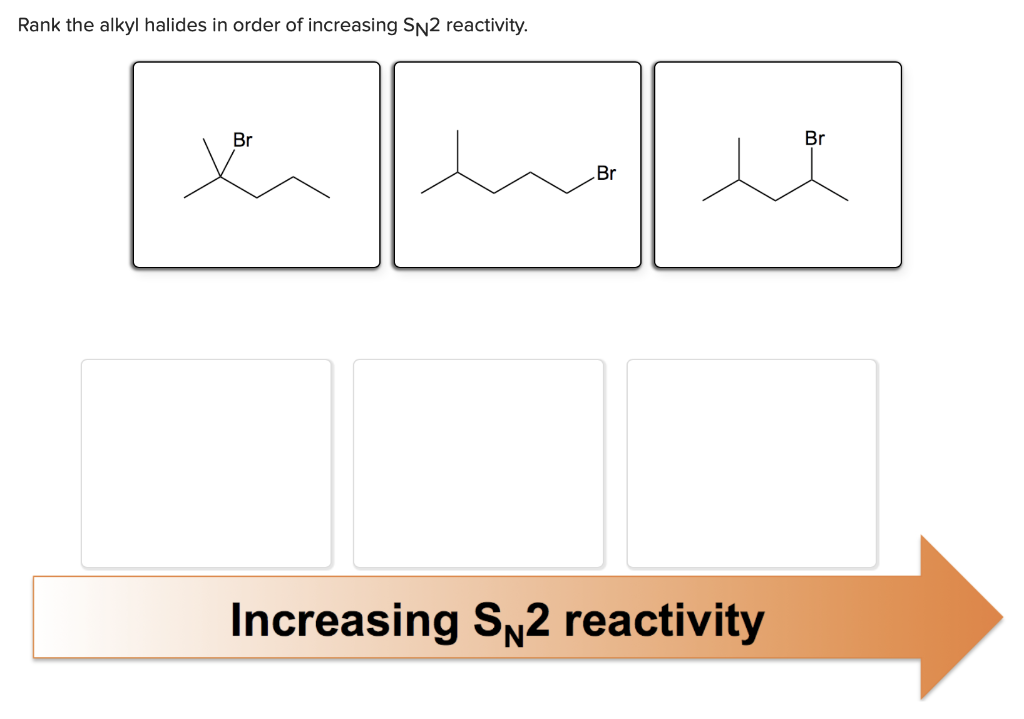
Rank The Alkyl Halides In Order Of Increasing Sn2 Reactivity
Hey there, future organic chemistry superstar! Let's chat about something super cool: SN2 reactions. You know, those sneaky little reactions where a nucleophile comes in, kicks out a leaving group, and basically does a swift little dance. Today, we're going to tackle the big question: Which alkyl halides are the most eager beavers when it comes to SN2? Think of it like a race, and we're figuring out who's got the nitro boost!
So, what even is an SN2 reaction? Well, the "SN" stands for Substitution Nucleophilic, and the "2" means it's a bimolecular reaction. That means two molecules are involved in the rate-determining step – the nucleophile and the substrate (our alkyl halide). The nucleophile, like a superhero with a negative charge or a lone pair of electrons, spots the electrophilic carbon (the one attached to the halogen, which is always a bit of a drama queen, pulling electron density away). Then, BAM! The nucleophile attacks the back of the carbon, and the halogen, our leaving group, peaces out. It’s a one-step, backside attack kind of deal. Pretty neat, right?
Now, the reactivity order. This is where things get interesting. It's not just random; there's some logic to it! We're going to rank these guys from the slowest movers to the speed demons of the SN2 world. So, grab your imaginary pit crew and let's get this race started!
The Slowest of the Slow: Tertiary Alkyl Halides
Alright, let's start with the guys who are, let's say, a little… reluctant to get involved in SN2. We're talking about tertiary alkyl halides. What makes them tertiary? It means the carbon atom that's holding onto the halogen is also attached to three other carbon atoms. Think of it like this: our central carbon is wearing a big, bulky sweater with three sleeves. And on each sleeve, there's another carbon attached. Super crowded!
Why is this crowding such a big deal for SN2? Remember that SN2 reaction is a backside attack. The nucleophile has to get in there and poke the carbon from the opposite side of where the leaving group is. But with a tertiary alkyl halide, it's like trying to sneak into a VIP party through the back door when the bouncers (those bulky alkyl groups) are having a serious chat. They're just in the way!
These bulky groups create what we call steric hindrance. It's like trying to navigate a crowded dance floor – the more people there are, the harder it is to move around. The nucleophile just can't get close enough to the electrophilic carbon to launch its attack. So, tertiary alkyl halides are practically out of the running for SN2 reactions. They'd rather do other things, like maybe take a nap or contemplate the meaning of life. They’re the kings of saying, "Nope, not today!"
Imagine you're trying to pass a note to someone across a table, but there are three really big people sitting between you. It's a tough gig, right? That's what it's like for a nucleophile trying to attack a tertiary carbon. They're basically saying, "Can't squeeze in, sorry!"
The Next in Line: Secondary Alkyl Halides
Moving up the speed ladder, we encounter secondary alkyl halides. These guys are a bit more chill. Here, the carbon attached to the halogen is connected to two other carbon atoms and one hydrogen atom. So, it’s like our central carbon is wearing a sweater with two sleeves and one smaller arm.
There's still some steric hindrance here, but it's significantly less than with tertiary alkyl halides. It's like the dance floor is a bit less crowded. The nucleophile can still manage a backside attack, but it might have to do a little sidestep or two. It's not as easy as it could be, but it's definitely more possible than with the tertiary guys.
Secondary alkyl halides can undergo SN2 reactions, but their reactivity is moderate. They're like the middle-of-the-pack runners in our race. They’re not the fastest, but they’re not the slowest either. They’re just… there. Getting the job done, eventually.
Think of it like trying to pass that note again. Now there are only two big people between you and your friend. Still a bit tricky, but you might be able to find a gap and slide it over. It's all about managing the space!
The Speedsters Emerge: Primary Alkyl Halides
Now we're getting to the good stuff! Enter the primary alkyl halides. These are the ones where the carbon holding the halogen is attached to only one other carbon atom and two hydrogen atoms. This carbon is wearing a sweater with only one sleeve and two smaller arms. Much less clutter!
With primary alkyl halides, the backside attack by the nucleophile is much, much easier. The steric hindrance is minimal. It's like the dance floor has cleared up, and the nucleophile can waltz right in for that backside attack without breaking a sweat. This makes them significantly more reactive in SN2 reactions compared to their secondary and tertiary cousins.
Primary alkyl halides are the workhorses of SN2 chemistry. They’re consistently ready to go, accepting nucleophiles with open arms (well, with an open backside, technically). They’re fast, efficient, and reliable. They’re the sprinters of our race, leaving the others in the dust.
Imagine that note-passing scenario again. Now there's only one person between you and your friend. Easy peasy! You can just lean over and pass the note with no problem. That’s the kind of accessibility primary alkyl halides offer to nucleophiles.
The Ultimate Winner (Almost): Methyl Halides
And now, for the grand champion, the absolute king of the SN2 hill (or at least the closest we get to it in simple alkyl halides): methyl halides! In this case, the carbon attached to the halogen is only attached to three hydrogen atoms. That's right, just hydrogens! No bulky carbon groups to get in the way at all.
This is the most unhindered situation possible. The nucleophile can approach the electrophilic carbon with absolutely no resistance. It's like a wide-open highway for the nucleophile! The backside attack is incredibly easy and fast. Methyl halides are the fastest SN2 reactants among the simple alkyl halides.
These guys are the Usain Bolts of the SN2 world. They’re incredibly reactive and will practically jump at the chance to participate in an SN2 reaction. If you want a reaction to happen quickly and efficiently, a methyl halide is your best bet.
So, the note-passing analogy? It’s like you and your friend are sitting right next to each other. No one in between. You can just whisper the note. That’s how easy it is for a nucleophile to attack a methyl halide. Pure, unadulterated speed!
The Leaving Group Factor: Not Just About Sterics!
Okay, so we've talked a lot about the carbon chain and the crowding. But there's another crucial player in the SN2 game: the leaving group! The halogen itself. Not all halogens are created equal when it comes to deciding to leave. Think of the leaving group as the person who’s going to make the exit. Some people are happy to leave a party, while others are glued to the dance floor.
Generally, the better the leaving group, the faster the SN2 reaction. What makes a good leaving group? Stability! When the halogen leaves, it takes a pair of electrons with it. If the leaving group is stable on its own (usually because it’s a weak base), it's more likely to ditch the party. We're talking about halides like iodide (I⁻), bromide (Br⁻), and chloride (Cl⁻).
Iodide (I⁻) is the best leaving group among these. Why? Because iodine is a big atom, and it can spread out that negative charge over a larger volume, making it more stable. Think of it as a big blanket to keep the charge cozy. Bromide (Br⁻) is next, then chloride (Cl⁻).
Fluoride (F⁻), on the other hand, is a terrible leaving group. Fluorine is small, and it holds onto that negative charge very tightly. It’s like trying to get a tiny, grumpy baby to give up its favorite toy – not happening easily!
So, even if you have a super unhindered carbon, if your leaving group is weak (like fluoride), the SN2 reaction will be sluggish. It's a team effort, you see! The carbon's openness and the leaving group's willingness to leave both matter.

Putting It All Together: The Grand SN2 Reactivity Ranking!
Alright, let’s summarize our findings and create the ultimate SN2 reactivity ranking, from slowest to fastest. Remember, this is for simple alkyl halides, where we're mostly considering the steric hindrance around the carbon and the nature of the leaving group.
Here it is, the moment of truth:
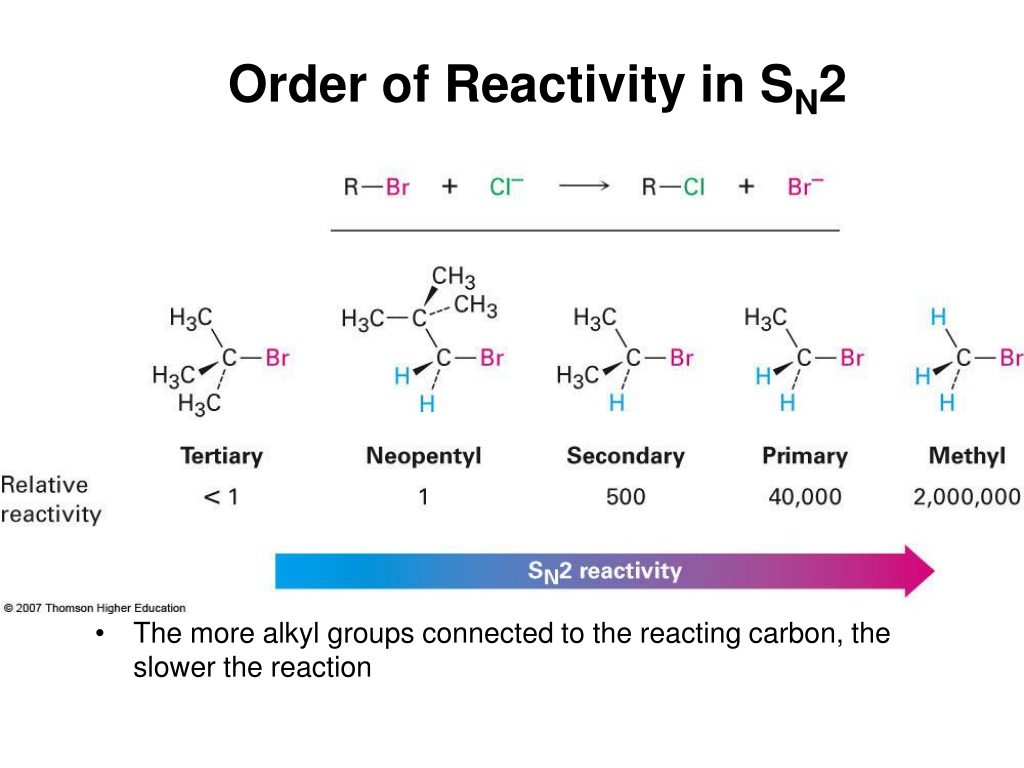
Tertiary Alkyl Halides (R₃C-X) < Secondary Alkyl Halides (R₂CH-X) < Primary Alkyl Halides (RCH₂-X) < Methyl Halides (CH₃-X)
And within each category, the reactivity also depends on the leaving group, with iodide being the best and fluoride being the worst.
So, if you have a tertiary iodide, it'll be much slower than a primary iodide. But a tertiary fluoride? Forget about it for SN2! A methyl iodide will be the speediest of the bunch.
Let's break it down with some specific examples. Imagine you have these four compounds:
- (CH₃)₃C-I (tert-butyl iodide)
- (CH₃)₂CH-I (isopropyl iodide)
- CH₃CH₂-I (ethyl iodide)
- CH₃-I (methyl iodide)
In an SN2 reaction, the order of reactivity would be:
(CH₃)₃C-I (slowest) < (CH₃)₂CH-I < CH₃CH₂-I < CH₃-I (fastest)
Now, what if we kept the structure the same but changed the halogen? Let’s compare ethyl iodide, ethyl bromide, and ethyl chloride:
- CH₃CH₂-I (ethyl iodide)
- CH₃CH₂-Br (ethyl bromide)
- CH₃CH₂-Cl (ethyl chloride)
The reactivity order for SN2 would be:
CH₃CH₂-Cl (slowest) < CH₃CH₂-Br < CH₃CH₂-I (fastest)
Pretty logical when you see it laid out, right? It all comes down to how easily the nucleophile can get to the carbon and how willing the leaving group is to make its exit.
The Takeaway: It's All About Ease and Eagerness!
So, there you have it! The grand SN2 reactivity ranking. Remember, it's a combination of having a clear path (less steric hindrance) and a willing partner (a good leaving group) that makes for a speedy SN2 reaction. The simpler the carbon skeleton around the reactive center, the better. And the more stable the leaving group, the happier it is to go.
Don't let these concepts seem overwhelming. Think of it like learning a new dance. At first, you might be a bit clumsy, bumping into people. But with practice, you learn the steps, you see the open spaces on the dance floor, and soon you’re moving with grace and speed. These rules are just the choreography for the SN2 dance!
Keep practicing, keep experimenting (in your mind, of course, unless you're in a lab!), and you'll master the art of SN2 reactions in no time. You're doing great, and every question you ask, every concept you learn, brings you closer to becoming an organic chemistry whiz. Keep that curiosity alive, and remember, the world of chemistry is full of fascinating reactions waiting for you to discover them. Now go forth and conquer those SN2 reactions with a smile!
