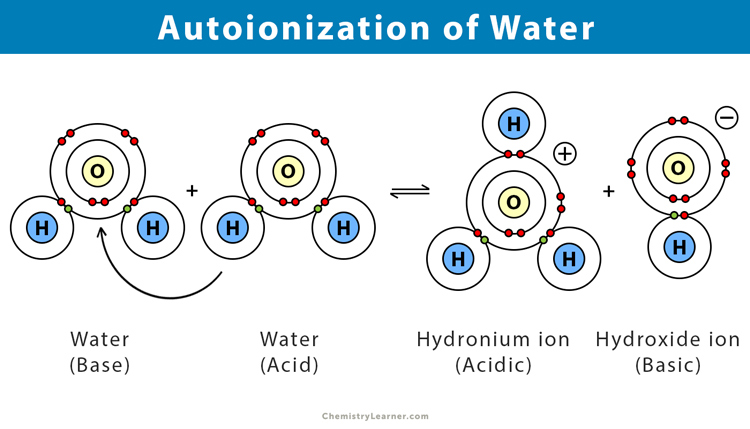
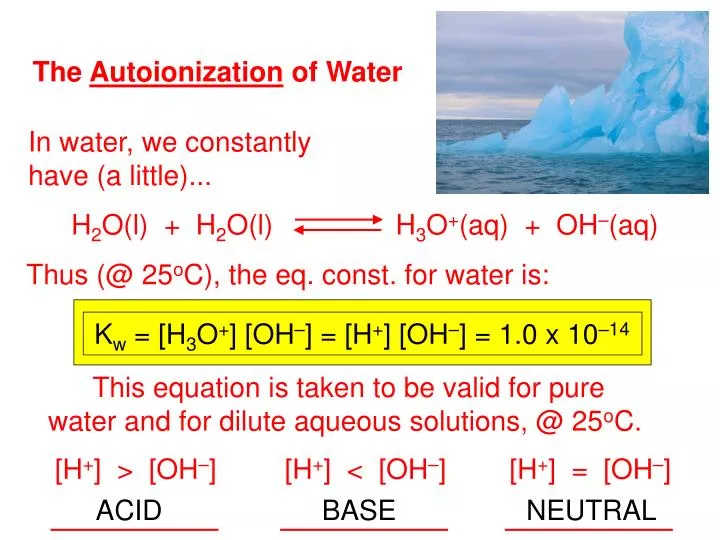
Pure Water Autoionizes As Shown In The Equation Above

So, let's talk about something really simple. Water. You know, that stuff you drink, shower in, and sometimes spill on your keyboard. It seems pretty straightforward, right? Just H₂O, happy and molecular.
But here’s where things get a little… well, dramatic. Turns out, pure water has a secret life. It’s not just chilling out as whole H₂O molecules. Oh no. Water is a bit of a drama queen.
It’s been scientifically proven, with fancy equations and everything, that water sometimes breaks apart. It doesn’t stay perfectly intact. It’s like it gets tired of being a whole molecule and decides to have a little… identity crisis.
The equation, which I’m not going to bore you with the details of, shows this happening. It’s like water is having a little internal party. A very, very small party, mind you. But a party nonetheless.
We call this autoionization. Sounds super scientific, doesn’t it? Like something a mad scientist in a lab coat would shout. But it’s just water being water, apparently. Getting a bit… split-personality.
Imagine H₂O molecules bumping into each other. It’s not like a gentle nudge. Sometimes, it’s a bit of a shove. One water molecule basically gives another one a bit of a steal.
It’s like one molecule is saying, “Hey, can I borrow an electron?” And the other is like, “Sure, here you go!” Except, it’s not really borrowing. It’s more like a permanent transfer. A very, very polite, molecular heist.
And what happens when you take away an electron from H₂O? Or give one away? Things get charged. Suddenly, you’ve got something that isn’t quite H₂O anymore. It’s like a water molecule that’s lost its cool.
You end up with bits that are positively charged, and bits that are negatively charged. Like tiny water-based batteries. Always on the verge of… something.

The positively charged bit is what we call a hydronium ion. Think of it as a water molecule that’s picked up an extra H+. It’s got a bit of an attitude now. It’s like the overachiever of the water world.
And the negatively charged bit? That’s a hydroxide ion. This one is like the leftover piece. The one that lost something. It’s got a bit of a moody vibe. A bit of a “what just happened to me?”
So, in pure water, you’ve got these perfectly normal H₂O molecules floating around, being… well, water. And then, every now and then, a couple of them do this whole autoionization thing. They break apart into hydronium and hydroxide.
It’s not a lot, mind you. It’s a teeny tiny amount. Like finding a unicorn at your local grocery store. It happens, but you wouldn't bet your house on it. The vast majority of water stays as H₂O.
But it's the principle of the thing, isn't it? The fact that it can and does happen. Even in the most pristine, pure water. It’s like water is whispering secrets to itself.
This little dance of ions is crucial for so many things. It’s why water can dissolve stuff. It’s why it can conduct electricity (a little bit, anyway). It’s the backbone of chemistry.
It’s almost like water is saying, “Don’t underestimate me. I might look simple, but I’ve got hidden depths. And by ‘depths,’ I mean a tendency to spontaneously disassemble.”
And this happens all the time, everywhere. In your tap water, in your fancy bottled water, even in the water your body is made of. It’s a constant, quiet rearrangement. A molecular ballet.
Sometimes I think about this when I’m doing the dishes. I’m splashing around in this stuff, and meanwhile, the water molecules are having their own little existential crises. Breaking off, reforming. It’s a wild world in there.
And the funny thing is, we just take it for granted. We think of water as this inert substance. But it’s actually incredibly dynamic. It’s always on the move, chemically speaking.
It’s a bit like those perfectly polished apples you see in fruit bowls. They look so still and perfect. But underneath, there’s all this biological activity happening. Water is the same.
This autoionization thing is like water’s way of saying, “I’m not just a bystander. I’m an active participant.” It’s a little bit rebellious. A little bit unexpected.
And honestly? I kind of love it. It’s an unpopular opinion, maybe, but I think it’s cool. Water isn't just passive. It’s got this hidden energy. This willingness to change.
It makes me look at a glass of water differently. It’s not just a drink. It’s a miniature chemical reaction happening right before my eyes. A tiny, silent, ongoing transformation.

Think about it. This simple molecule, H₂O, is constantly breaking down and reforming. It’s a testament to the fact that even the most common things can have hidden complexities.
It’s like finding out your quiet neighbour is secretly a world-class juggler. You just never knew it because they never felt the need to show off. Water is the quiet juggler of the molecular world.
So next time you take a sip of water, or wash your hands, spare a thought for the autoionization. The hydronium and hydroxide ions having their little tête-à-tête. It's happening. It's real.
And it’s just another one of those wonderfully weird things about our universe that makes you smile. The universe is full of surprises, and sometimes, those surprises are found in the most ordinary places. Like a glass of water.
It’s a tiny, almost imperceptible dance, but it’s a fundamental part of what makes water so special. The ability to break apart, to reform, to be a little bit… self-involved.
So, here's to water. The molecule that’s more than it seems. The one with a hidden talent for drama. The one that autoionizes, just because it can. And we’re all the better for it.
It’s like a tiny reminder that nothing is truly static. Everything is in motion, in flux. Even water, in its purest form, is a swirling, dynamic entity.
And that, my friends, is kind of amazing. It’s the subtle magic of chemistry, happening all around us, all the time.
Pure water is never truly pure. It's always got a little bit of sparkle, a little bit of sass, thanks to its tendency to autoionize. It’s the drama queen of the liquid world, and we wouldn’t have it any other way.
So, let’s appreciate the autoionizing water. The water that’s not afraid to break a few bonds. The water that’s always ready for a chemical adventure.
It's a little bit of chaos in an orderly world. A splash of unpredictability. And it’s happening right now, in every drop.
It just goes to show, you never quite know what’s going on beneath the surface. Even with something as simple as water. It’s a constant source of wonder, and a gentle nudge to remember that complexity often hides in plain sight.
So, cheers to the H₂O molecules that decide to shake things up. The ones that become hydronium and hydroxide. They’re the unsung heroes of chemical reactions, the silent movers and shakers.
And that, in a nutshell, is why water is so fascinating. It’s not just a drink; it’s a tiny, dynamic universe in a glass.
It’s a secret that’s out in the open, if only you choose to look. And once you see it, you can’t unsee it. The autoionizing water is everywhere.
