Provide The Proper Iupac Name For The Alkene Shown Below

Hey there, chemistry curious folks! Ever feel like some science words are just… a little intimidating? Like they belong in a super-secret lab with beakers bubbling and lab coats swishing? Well, today we’re going to peek behind the curtain of one of those words, and I promise, it’s going to be way more fun than you think. We're talking about naming things, specifically, a molecule that’s a bit like a quirky little character in the grand story of chemistry. And guess what? It's called an alkene!
Now, imagine you’ve got a bunch of LEGO bricks. You can build all sorts of amazing things, right? From a simple house to a towering spaceship. Chemistry is kind of like that, but instead of LEGOs, we have tiny building blocks called atoms. These atoms love to connect and link up in all sorts of ways to make everything we see and touch. Our friendly neighborhood alkenes are special because they have a particular kind of connection: a double bond.
Think of a double bond like a really strong handshake. Most atoms are happy with a single handshake, you know, holding hands one-on-one. But an alkene is like two atoms saying, "Hey, we're extra close! Let's hold hands twice!" This double handshake makes them a bit more reactive, a bit more eager to join in on other chemical adventures. It’s like they’re the life of the chemical party, always ready to mingle and make new friends.
So, why should you, a perfectly normal human being who probably has better things to do than memorize molecular structures, care about this? Well, these alkenes are everywhere! They’re in the air we breathe (some are!), they’re in the food we eat, and they’re even in the plastics that make our lives convenient. Understanding them, even a little bit, is like getting a secret decoder ring for the world around you. Suddenly, that plastic water bottle isn’t just a plastic water bottle; it’s made of molecules that have a story, and that story includes a double bond!
Now, every molecule, just like every person, needs a name. And in the world of chemistry, we have a super-organized system for naming them. It’s called the IUPAC system. Think of it as the ultimate naming convention, ensuring that no matter where you are in the world, a chemist can look at a name and know exactly what molecule you’re talking about. No more confusion, no more accidentally ordering “but-what-even-is-this-ene?” from a chemical catalog!
Let’s get to the fun part: naming the specific alkene you’ve got your eye on. Imagine this alkene is a little family. The IUPAC system helps us figure out the family name and then give each member their own special title. The most important thing for an alkene’s name is finding the longest chain of carbon atoms that includes that special double bond.
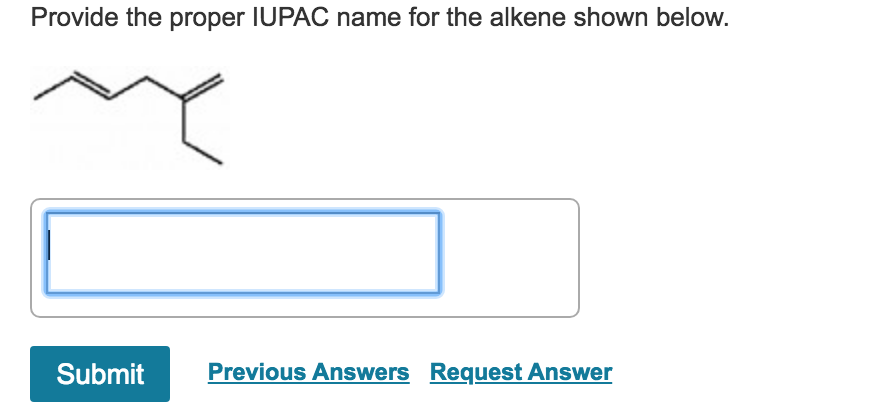
Let's say our alkene looks like this (don't worry if it looks a bit like spaghetti!):
C=C-C-C
The carbon atoms are the ‘C’s, and the ‘=’ sign is our super-strong double bond handshake. We need to find the longest stretch of these carbon atoms that has the double bond in it. In this simple example, it’s a chain of four carbon atoms. Four carbons? In the world of alkenes, that’s a but- prefix. So, we start with “but-“.
Next, we need to tell everyone where that double bond is hanging out. We do this by numbering the carbon chain. The trick is, we always start numbering from the end that's closest to the double bond. So, in our C=C-C-C example, the double bond is right at the beginning. If we number from left to right, the double bond is between carbon 1 and carbon 2. If we number from right to left (C-C-C=C), the double bond would be between carbon 3 and carbon 4.
See? Starting from the left makes the double bond appear earlier in the numbering! So, we start numbering from the left. The double bond is at position 1. So, we add a ‘1-’ to our name.
Finally, because it’s an alkene (remember that double bond handshake?), we add the suffix ‘-ene’. So, putting it all together, our alkene becomes but-1-ene!
It’s like naming your pet. You don’t just call it “Animal.” You give it a specific name, like “Whiskers” or “Buddy.” IUPAC naming is the same, just for molecules. It’s precise and universally understood.
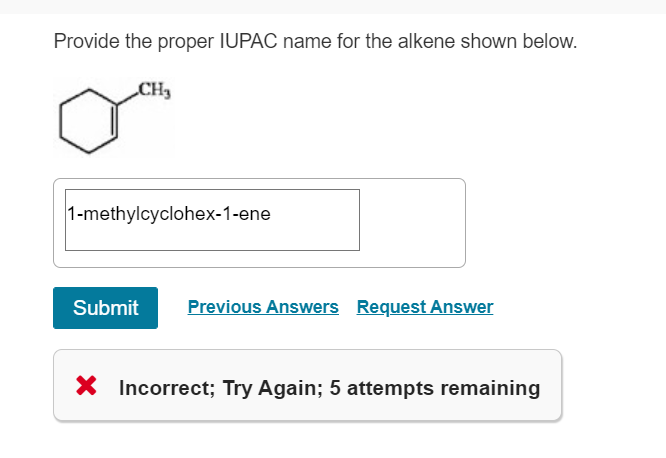
Let’s try another one. Imagine this: C-C=C-C-C. What’s the longest chain that includes the double bond? It’s five carbon atoms! So, our prefix is pent-.
Now, where’s the double bond? If we number from the left, it’s between carbon 2 and carbon 3. If we number from the right, it’s between carbon 3 and carbon 4. Which end is closer to the double bond? The left end! So, we number from the left.
The double bond starts at carbon number 2. So, we add ‘2-’ to our name. And finally, we add the ‘-ene’ suffix. Voila! We have pent-2-ene.
It’s like giving directions. You don’t just say “go north.” You say “turn left at the third street, then go two blocks.” The numbers in the IUPAC name are like those street numbers, telling us exactly where the action (the double bond) is happening.

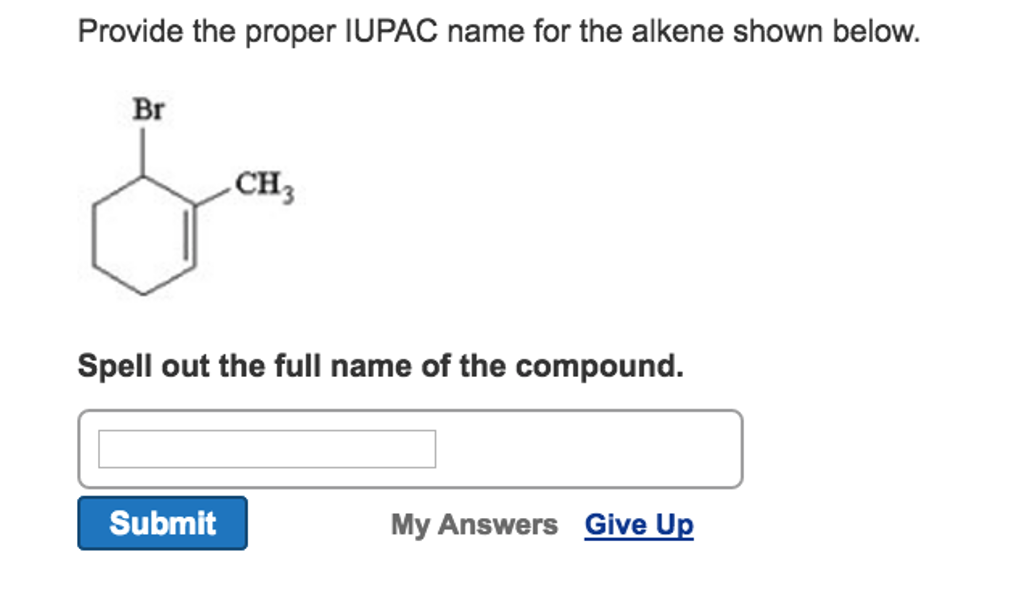
Now, sometimes molecules can get a bit trickier. They might have branches, like little arms coming off the main chain. But the core principle remains the same: find the longest chain with the double bond, number it correctly, and identify the position of that double bond. You might even see prefixes like “methyl” or “ethyl” for those little branches, but that’s a story for another day!
Why bother with all this precision? Well, think about medicine. A slight change in a molecule's structure can completely change its effect on our bodies. One isomer might be a life-saving drug, while another might be completely inactive or even harmful. The IUPAC name is like the molecular fingerprint, ensuring that the right molecule is synthesized, tested, and used. It’s a matter of safety, efficacy, and scientific progress.
Even in everyday materials, understanding these names helps us appreciate the chemistry behind them. When you hear about bioplastics or sustainable materials, knowing that certain alkenes are key components can make those concepts more tangible. It’s about demystifying the world and realizing that even the most complex substances are built from simple rules and predictable patterns.
So, the next time you see a chemical formula or hear a fancy chemical name, don't run away! Take a deep breath, remember our LEGO analogy, our strong handshakes, and the importance of a good name. The IUPAC name for the alkene shown below is a little puzzle, and once you understand the pieces, it’s incredibly satisfying to solve. It’s not just about memorizing; it’s about understanding the language of the universe, one molecule at a time. And isn't that pretty cool?
