Provide The Kinetic Product Of The Following Reaction.
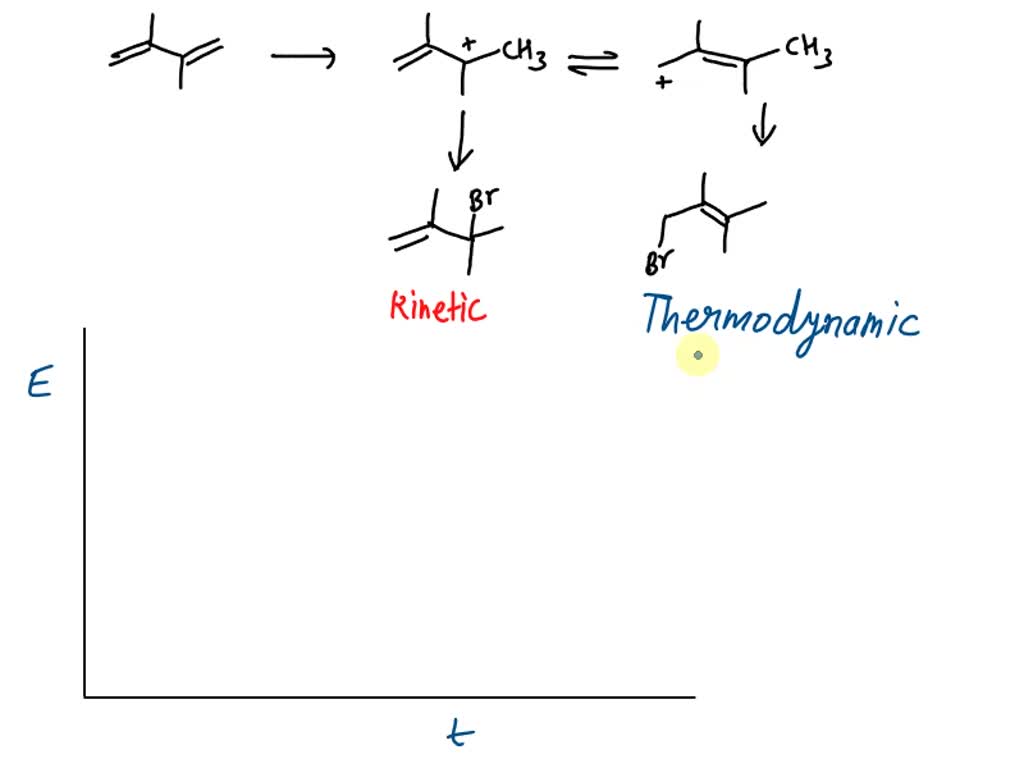
So, you've probably seen those fancy chemistry problems, right? The ones that look like a secret code from outer space. They throw in these crazy names and funny arrows. And then, the big question: "Provide the kinetic product of the following reaction."
Now, I have an unpopular opinion about this. A really unpopular opinion. I think the kinetic product of pretty much any reaction is... well, it's the stuff that gets made first. The speedy stuff. The "get 'er done" molecules.
Think about it. Life is busy. The universe is busy. Things are always happening. Molecules are bumping into each other, doing their little dance. And when they dance, sometimes they make new things. Some of those new things pop out really fast. They're the rock stars of the molecular world, hitting the stage and wowing the crowd before anyone else even gets a chance to warm up.
These are your kinetic products. They're the ones that win the race. They probably didn't have to try that hard. They just sort of... happened. Like when you're making cookies and you accidentally drop a blob of dough on the floor. It's not the prettiest cookie, but it's definitely the first cookie you made, right? That's a kinetic product.
Now, there are other products. The ones that take their sweet time. They might be more elegant. More perfectly formed. They probably involved some deep thinking and careful planning. These are the thermodynamic products. They're the ones that, by the time they show up, the party's already winding down. They're a bit like that friend who always shows up late to the concert, but when they do, they have the best seat.
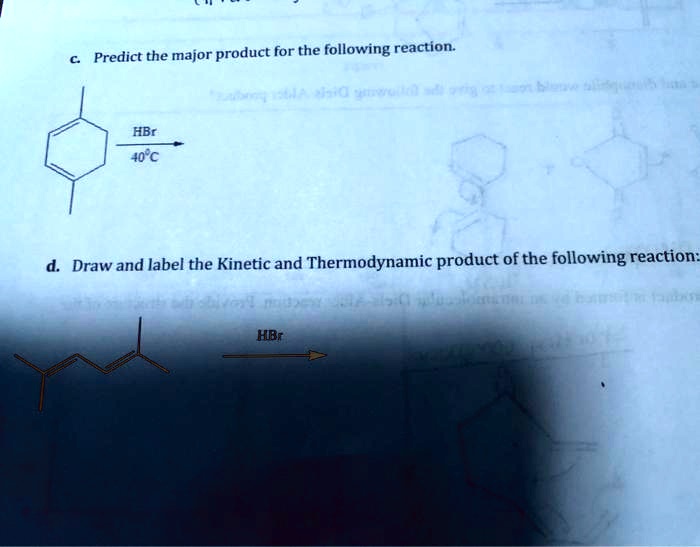
But back to our kinetic pals. They're the rebels. The daredevils. They don't wait for conditions to be just right. They're like, "You know what? Let's just do this now." And boom. Product. It might not be the most stable thing. It might not be the most efficient. But it's there. It's a fait accompli. It's the participation trophy of the molecular world, and honestly, I respect that.
Imagine a bunch of single molecules milling about. They're bored. They're looking for excitement. They bump into each other. Wham! Maybe they form a new bond. Maybe they rearrange themselves. If it happens quickly, without much fuss, that's your kinetic product. It’s the equivalent of a spontaneous combustion, but in a good way. Or at least, a way that gets you an answer on a test.
It's like this: You're trying to build a Lego castle. You've got a gazillion bricks. You start snapping them together as fast as you can. You're not thinking about structural integrity. You're not thinking about the perfect turret. You're just trying to get something built. That first, wobbly, slightly lopsided structure? That's your kinetic product. It's a testament to speed, not necessarily to long-term planning.
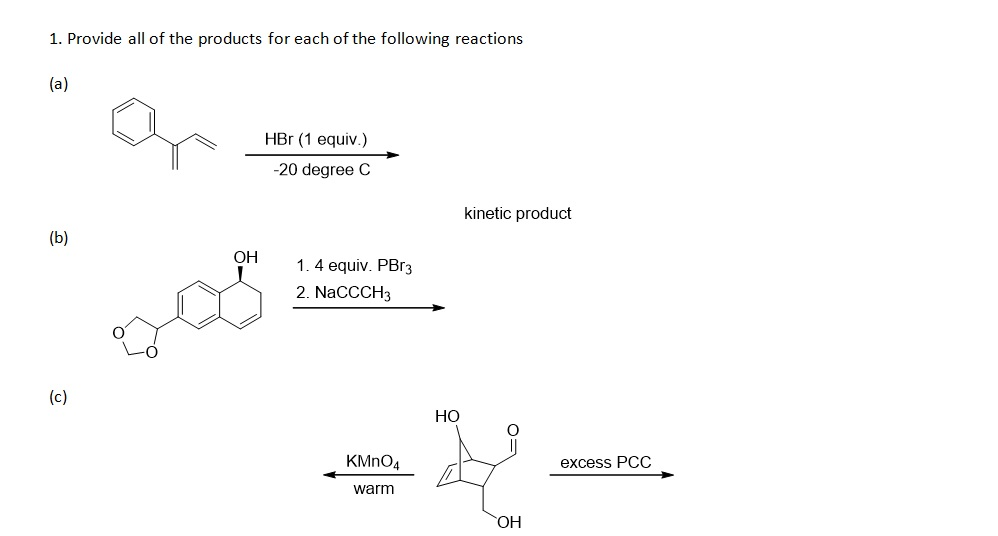
My entirely unofficial, highly unscientific, but admittedly fun take is that the kinetic product is just whatever shows up the quickest. No judgment, just pure, unadulterated speed.
And you know what? Sometimes, that's exactly what we need. We need things to happen. We need progress. We don't always have the luxury of waiting for the "thermodynamically favored" outcome. Sometimes, "good enough, and fast" is the best we can do. And for that, I salute the kinetic product.
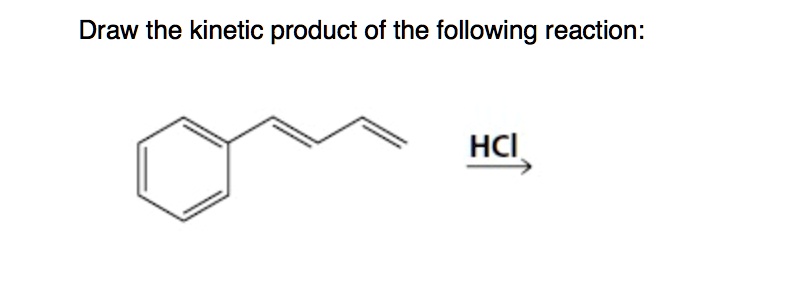
So, when you see that question – "Provide the kinetic product of the following reaction" – take a deep breath. Don't overthink it. Think about what molecule would be the first to say, "Let's get this party started!" That's probably your kinetic product. It's the molecule that's got places to be, people to see, and chemical reactions to, well, react to.
It’s the molecular equivalent of saying, "I'm here, I'm doing something, and I'm doing it now!" No apologies. No lengthy explanations. Just pure, unadulterated, in-your-face product. That’s the kinetic spirit. And frankly, it’s a spirit I can get behind. It’s the underdog. It’s the quick thinker. It’s the one that doesn’t get bogged down in the details.
So, next time you're faced with a chemical reaction and asked for its kinetic product, just picture the molecules zipping around, bumping into each other with gusto, and forming something, anything, as quickly as humanly (or molecularly) possible. That’s your answer. It’s simple. It’s fast. And it’s, in my humble, slightly quirky opinion, the most entertaining way to think about it. It’s the thrill of the chase, the excitement of the moment, the pure, unadulterated joy of a reaction that just can't wait to be born.
