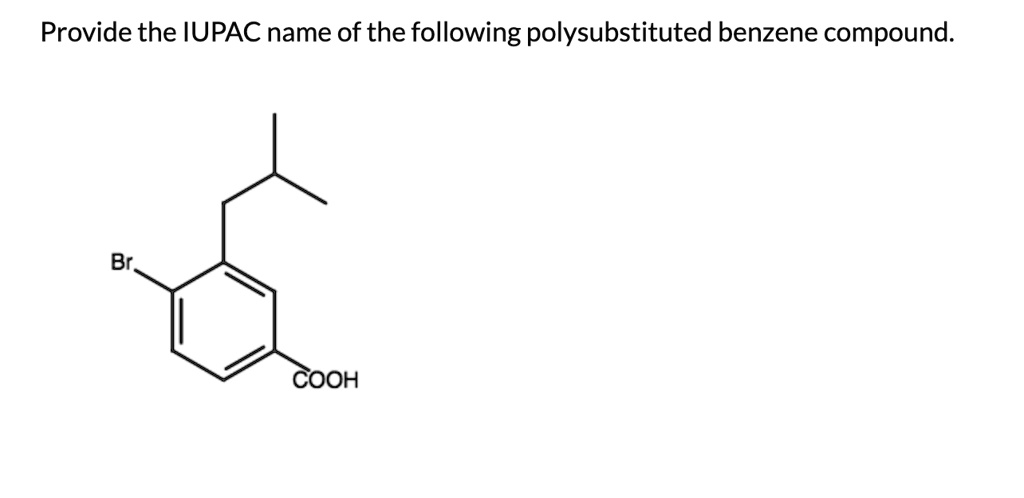
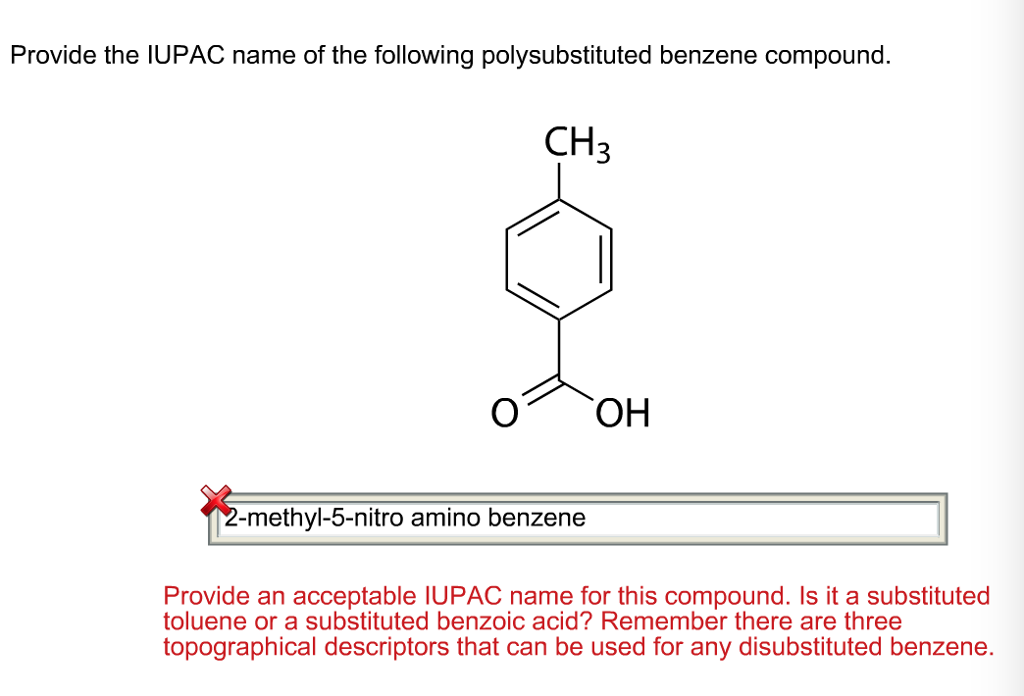
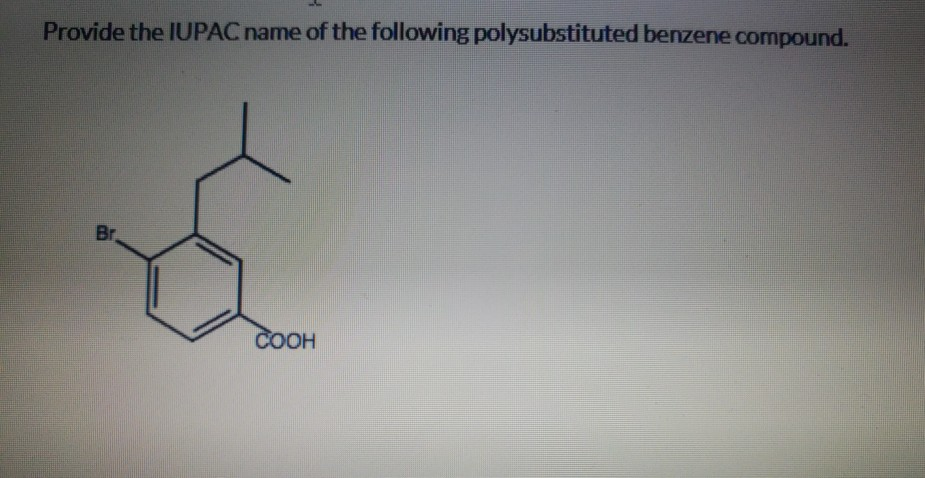
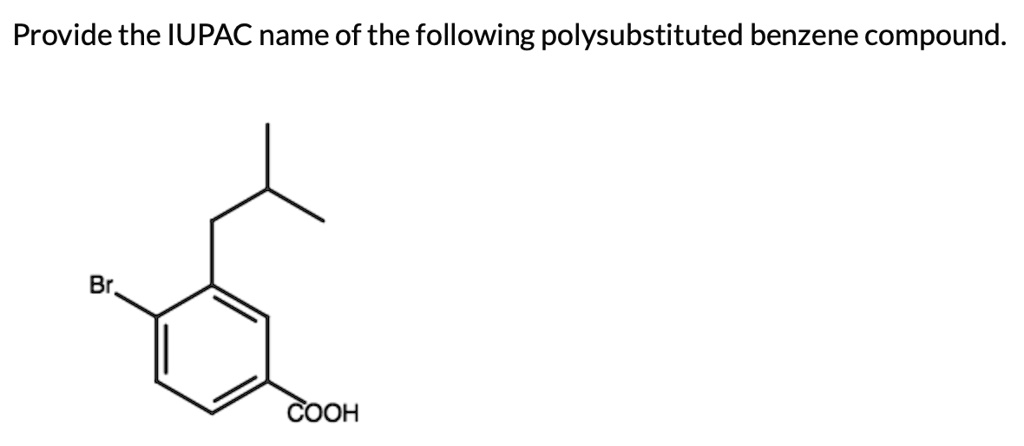
Provide The Iupac Name Of The Following Polysubstituted Benzene Compound.
Hey there, fellow adventurers in the world of chemistry! Ever felt like learning about fancy molecule names is about as exciting as watching paint dry? Well, buckle up, because we're about to turn that frown upside down! Today, we're diving into the wonderfully weird and surprisingly fun world of naming complex chemical structures. And trust me, it’s more like a fun puzzle than a grueling exam.
You see, every molecule, no matter how intricate, has its own unique identity. And in the grand ballroom of chemistry, these identities are given by something called IUPAC names. Think of it as the molecule's official, super-official, "everyone-agrees-on-this" name. It's how scientists across the globe can point to a drawing of a molecule and know exactly which one they're talking about. Pretty neat, right?
Now, sometimes these molecules can get a little… crowded. We're talking about benzene rings, those hexagonal wonders with a delightful pi-electron cloud. And when you start adding multiple bits and bobs, or substituents, to these rings, things can get a bit wild. It's like a party on a ring, and we need a systematic way to introduce everyone!
So, let's get to the fun part. We're going to tackle a polysubstituted benzene compound. What does that even mean? "Poly" just means "many," and "substituted" means we've attached other chemical groups to our trusty benzene ring. So, it's a benzene ring with lots of things hanging off it. Imagine a plain donut getting a whole bakery's worth of toppings! Yum!
Now, the challenge: how do we give this delicious-looking (but probably not edible!) molecule its proper IUPAC name? It's not as scary as it sounds. It's all about following a few simple, yet super smart, rules. Think of it like a treasure hunt, and the IUPAC rules are your map!
First things first, we need to identify the parent structure. In our case, the undisputed star of the show is the benzene ring. So, "benzene" will be the core of our name. Easy peasy, lemon squeezy!

Next, we look at the substituents. These are the little extras that have joined the benzene party. Each one has its own name, like a methyl group (CH3), an ethyl group (C2H5), a chlorine atom (Cl), or a nitro group (NO2). They're like the guests at our molecular bash!
Here's where the real puzzle-solving comes in: numbering the ring. This is crucial! We need to assign numbers to the carbon atoms of the benzene ring so we can tell everyone where each substituent is located. And the golden rule here is to give the substituents the lowest possible numbers. It's like finding the shortest route to get to all the cool attractions in a new city – you want the most efficient way!
So, you'd look at your molecule and figure out a starting point and a direction that minimizes the numbers assigned to your substituents. Sometimes, there's a bit of a tug-of-war between different numbering schemes, and you have to choose the one that's most minimal. It's like a strategic game of chess, but with atoms!
Once we've got our numbers, we list the substituents in alphabetical order, before the parent name. So, if you have a methyl group and a chloro group, "chloro" comes before "methyl" alphabetically. And don't forget to include those numbers to tell us exactly where each one is!
Let's imagine we have a benzene ring with a chlorine atom and a methyl group. If we number them so chlorine is at position 1 and methyl is at position 2, the name would be 1-chloro-2-methylbenzene. See? Not so bad!
What if we have three or more substituents? The same principle applies! You find the numbering that gives the lowest set of locants. For example, if you have a nitro group, a bromine atom, and an ethyl group, you’d find the numbering that results in the smallest possible numbers for their positions. Then, you’d list them alphabetically: bromo, then ethyl, then nitro. So, it might be something like 1-bromo-3-ethyl-4-nitrobenzene (just an example, the actual numbering would depend on the specific structure!).
It's really about building the name layer by layer. You're not just memorizing names; you're understanding a logical system. And when you grasp the system, you can decode any polysubstituted benzene. It's like learning a secret language that unlocks endless molecular possibilities!
Why is this fun, you ask? Because it gives you the power to understand and describe the building blocks of so many things around you! From medicines that heal us to materials that build our world, complex organic molecules are everywhere. Knowing their names, even the fancy IUPAC ones, connects you to that hidden world. It's like having a backstage pass to the molecular universe!
Think about it: every time you see a complicated chemical formula in a textbook or an article, you'll have the tools to at least start to decipher it. You'll see the benzene ring, you'll spot the substituents, and you'll know the rules of the naming game. That's not just knowledge; that's empowerment!
And the beauty of it is, the more you practice, the easier it gets. It's like learning to ride a bike – a little wobbly at first, but soon you're cruising! So, don't shy away from those complex structures. Embrace them!
The IUPAC naming system for polysubstituted benzenes is a testament to human ingenuity – a way to bring order to complexity. It’s a system designed to be clear, unambiguous, and, dare I say, even a little elegant. So, the next time you encounter one of these molecular party animals, don't be intimidated. See it as an opportunity to flex your intellectual muscles, to solve a chemical riddle, and to appreciate the incredible diversity of the molecular world.
And remember, every complex name is just a series of simple rules waiting to be applied. So go forth, my friends, and name those molecules with confidence and a smile! The world of chemistry is vast and fascinating, and with tools like IUPAC naming, you're perfectly equipped to explore it. Happy naming!
