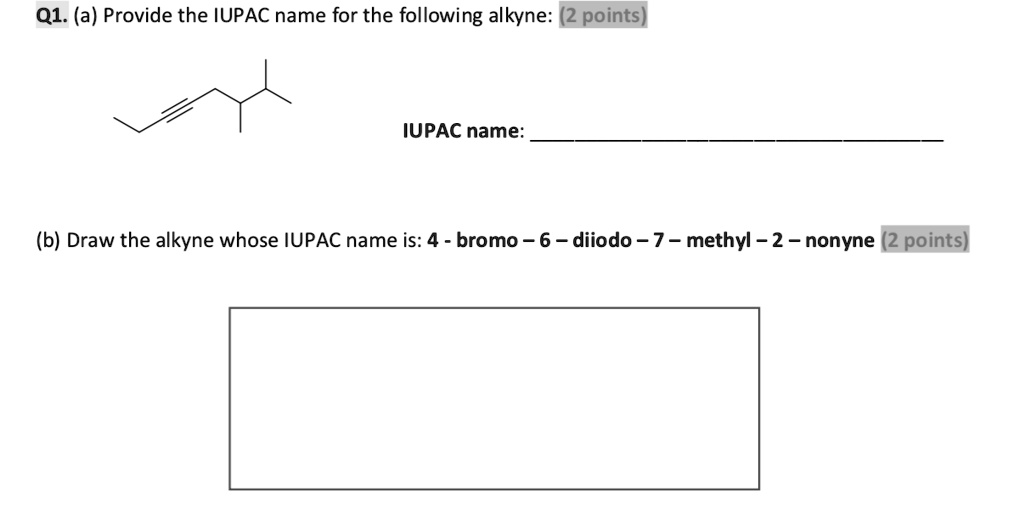
Provide The Iupac Name Of The Following Alkyne.

Ever looked at a jumble of letters and numbers and wondered what secret code it holds? Well, get ready for a little bit of fun because we're diving into the world of naming tricky molecules – specifically, <alkynes>! Think of it like learning a secret handshake for chemists, but way more useful. It’s a fascinating peek into how scientists keep track of all the amazing building blocks that make up our world, from the air we breathe to the medicines that help us.
So, why bother with these fancy names? For the <beginners> out there, it’s your first step into understanding the language of chemistry. It’s like learning your ABCs before you can read a book. For <families> exploring science together, it can be a cool way to discover how everyday things have a hidden structure. Imagine pointing to something and saying, "That's made of molecules with names like this!" For <hobbyists>, whether you’re into crafting, cooking with interesting ingredients, or even just appreciating the natural world, knowing a bit about naming helps you understand the ‘why’ behind a lot of processes.
Let’s get to the fun part: figuring out the IUPAC name of a specific alkyne. The <International Union of Pure and Applied Chemistry (IUPAC)> has a system to make sure everyone around the globe uses the same name for the same thing. This prevents confusion! For alkynes, the key is that they have a special triple bond between two carbon atoms, which we signal with the ending "-yne".
To find the name, we follow a few simple rules:
- Find the longest carbon chain that contains the triple bond. This chain gives us the base name (like methane, ethane, propane, etc., but with an "-yne" ending).
- Number the carbon atoms in that chain. Start numbering from the end that is closest to the triple bond.
- Locate the triple bond. The number of the carbon atom where the triple bond starts tells us its position.
- Add any other groups (like branches) attached to the main chain. These are named and their position is indicated by their carbon number.
Let’s try an example. Imagine an alkyne with 4 carbon atoms in a row, and the triple bond is between the first and second carbon. The longest chain is 4 carbons, so the base name is <butyne>. We number from the end closest to the triple bond, so it starts at carbon 1. Therefore, the IUPAC name is <but-1-yne>! If the triple bond was between the second and third carbon, it would be <but-2-yne>.
For getting started, don’t feel overwhelmed! You can find lots of online resources or even simple chemistry kits that help you build these molecules with sticks and balls. The best tip is to start with the simplest alkynes and work your way up. Think of it as a puzzle, and each rule you learn is a new clue to solve it.
Naming alkynes might seem like a small thing, but it’s a gateway to understanding a whole universe of chemistry. It’s a skill that’s both practical and surprisingly satisfying to master. So, next time you see a chemical formula, remember, it’s just a name waiting to be discovered!
