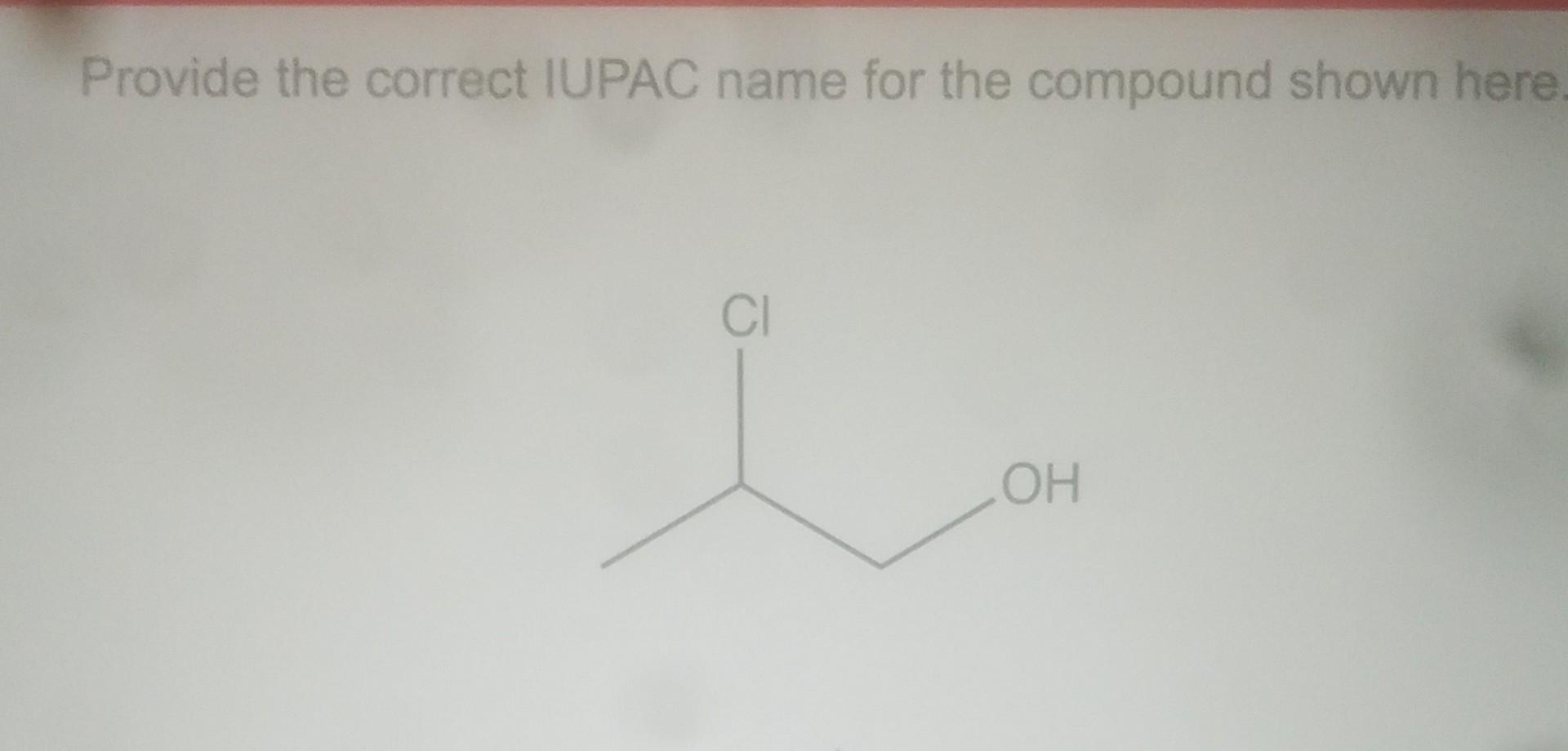
Provide The Correct Iupac Name For The Compound Shown Here.

Hey there, science nerds and curious cats! Ever stared at a jumble of letters and numbers and wondered what on earth it’s supposed to be? That’s kind of how chemistry can feel sometimes. It’s like a secret language for molecules. But guess what? We’re about to crack the code on one super interesting compound. Get ready to flex those brain muscles!
So, you see this picture, right? (Imagine a cool, slightly complex-looking molecule diagram here!) It looks like a tiny, intricate construction project, doesn't it? A bunch of atoms holding hands, some in straight lines, some making little bends. And those letters and numbers? They’re not random scribbles. They’re the official guest list for this particular molecular party.
Decoding the Molecular Mayhem
Let’s talk about the name. It’s called the IUPAC name. What’s IUPAC? It’s basically the International Union of Pure and Applied Chemistry. Think of them as the world’s fanciest club for naming things. They have rules, very strict rules, about how to name every single chemical out there. It’s like getting a really precise nickname for your pet, but way more important.
Why do we need such fancy names? Well, imagine you’re at a party and you ask for “that fizzy drink.” The bartender might bring you cola, or sprite, or even a fancy sparkling cider. It’s confusing! Chemistry is like that. If we just called everything “that thing with the rings,” we’d be in a whole heap of trouble. IUPAC names are unambiguous. One name, one molecule. Period.
The Stars of the Show: Carbon and Hydrogen
Take a peek at our molecule. You’ll notice a lot of Cs and Hs. That’s because this bad boy is an organic compound. Organic chemistry is basically the chemistry of life, and carbon and hydrogen are its superstar duo. They’re like the peanut butter and jelly of the molecular world, sticking together in all sorts of cool ways.
See those long chains of Cs? That’s like the molecule’s backbone. And the Hs? They’re often hanging off, like little accessories. The way they’re arranged, the number of carbons, the number of hydrogens, where everything is attached – that’s what gives the molecule its unique identity. And its name!
Let’s Get Down to Business: The Actual Name!
Okay, drumroll please… The correct IUPAC name for the compound shown here is… (wait for it…) 2-methylpentan-3-one!
Whoa, that sounds like a mouthful, right? But let’s break it down. It’s actually pretty logical once you get the hang of it. Think of it like a secret code you're unlocking.
The “Pentan” Part
First, let’s look at "pentan." In IUPAC naming, the end of the name tells you the main structure. "Pent-" means five. So, our molecule has a backbone of five carbon atoms lined up in a row. It’s like the main street of our molecular town.
If it was "ethan," it would be two carbons. "Propan," three. "Butan," four. And if it was "hexan," it would be six. So, "pentan" tells us we're dealing with a five-carbon chain. Simple as that!
The “-one” Ending
Next, we have the "-one." This tells us we have a ketone. What’s a ketone? Imagine a carbon atom double-bonded to an oxygen atom. That’s the functional group that makes something a ketone. It’s like the molecule wearing a fancy, double-bonded oxygen hat. Ketones are super common and have some really interesting properties. They’re often used as solvents, and some even have fruity smells!

The “3” in the Middle
Now, where is this ketone party happening? That’s where the number "3" comes in. We number the carbon atoms in our five-carbon chain from one end to the other. The "3" tells us that the double-bonded oxygen (the ketone part) is attached to the third carbon atom in the chain. So, it’s right smack in the middle of our five-carbon street.
The “2-methyl” Twist
Finally, we have the "2-methyl." This is where things get a little more interesting. "Methyl" is a group made of one carbon atom and three hydrogen atoms (CH3). It’s like a small, attached branch or a little sidekick to our main chain.
The "2-" tells us where this methyl group is hanging out. It’s attached to the second carbon atom of our five-carbon chain. So, on carbon number two, we have a regular hydrogen (or whatever else is supposed to be there if the picture isn't a pure hydrocarbon) and also this little methyl group sticking out.
It’s like our five-carbon street has a little detour or an extra house built on the second block. This might seem small, but it changes everything about the molecule’s shape and how it interacts with other molecules. It’s the difference between a plain vanilla ice cream and one with chocolate chips!
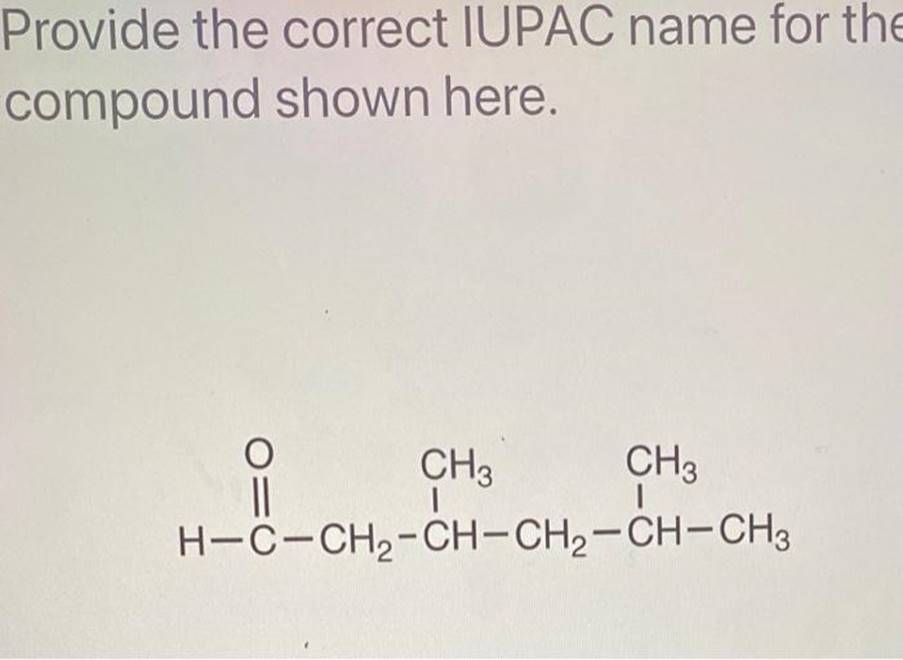
Why is This So Fun to Talk About?
Because it's like solving a puzzle! Each part of the name gives you a clue about the molecule’s structure. And once you know the rules, you can name any organic molecule you see. It’s like learning a secret handshake with the universe.
Think about it: chemists all over the world can look at "2-methylpentan-3-one" and immediately picture the exact same arrangement of atoms. No confusion, no ambiguity. That’s pretty darn cool.
Plus, these molecules are everywhere! They make up medicines, plastics, fuels, and even the flavors and fragrances we enjoy. Understanding their names is the first step to understanding how they work. It's like getting the secret ingredient list for everything around us.
Quirky Facts and Funny Details
Did you know that some ketones are actually used in nail polish remover? Yep, those powerful little molecules can dissolve tough stuff. And some have really pleasant smells, like the ketones found in fruits. So, the next time you’re smelling a juicy peach, you can thank a ketone!
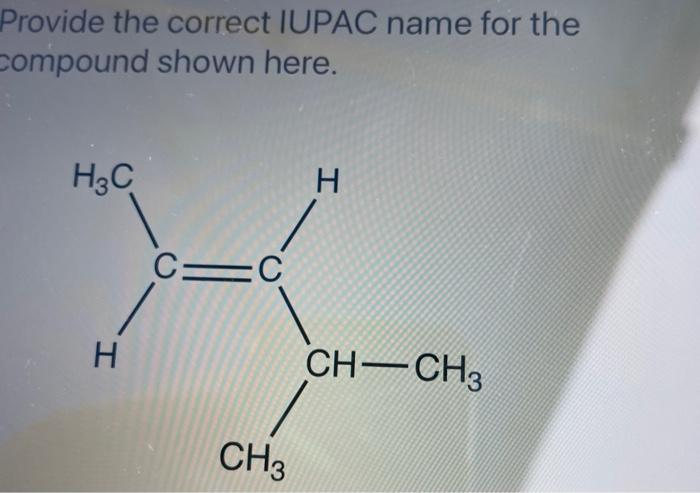
And the naming system itself is kind of a funny story. Imagine chemists arguing for hours about what to call things. Eventually, they got together and said, “Okay, let’s make some rules, people!” And thus, IUPAC was born. It's a testament to the need for order, even in the wild world of chemistry.
Inspiring Curiosity
So, next time you see a chemical name that looks like a tongue twister, don't be intimidated. Break it down. Look for the root words. See where the numbers lead you. It’s a journey of discovery, a little adventure into the tiny, invisible world that makes up our big, visible one.
This molecule, 2-methylpentan-3-one, is just one example. There are millions of organic compounds out there, each with its own unique story and IUPAC name. The more you learn, the more you realize how intricate and fascinating the world of chemistry truly is.
Keep exploring, keep asking questions, and who knows? You might just discover your own favorite molecular mystery to solve!
