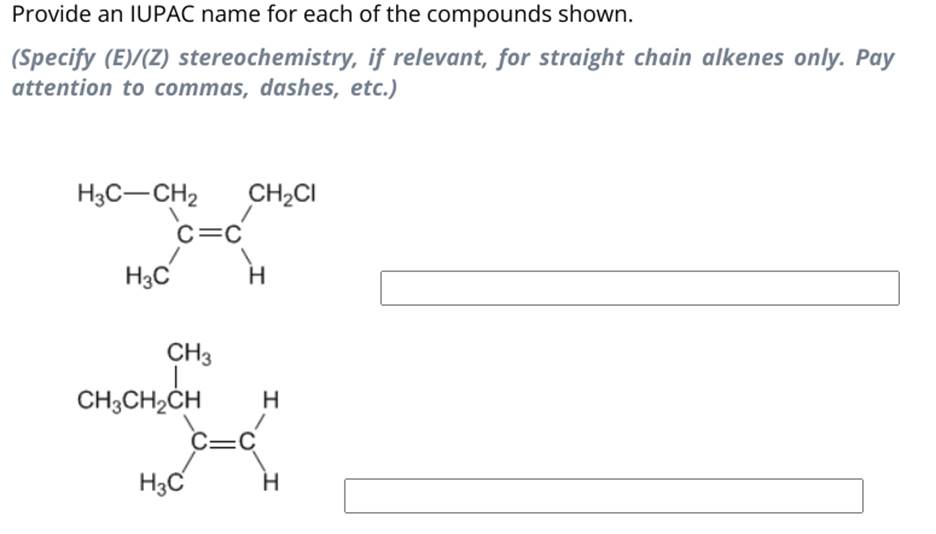
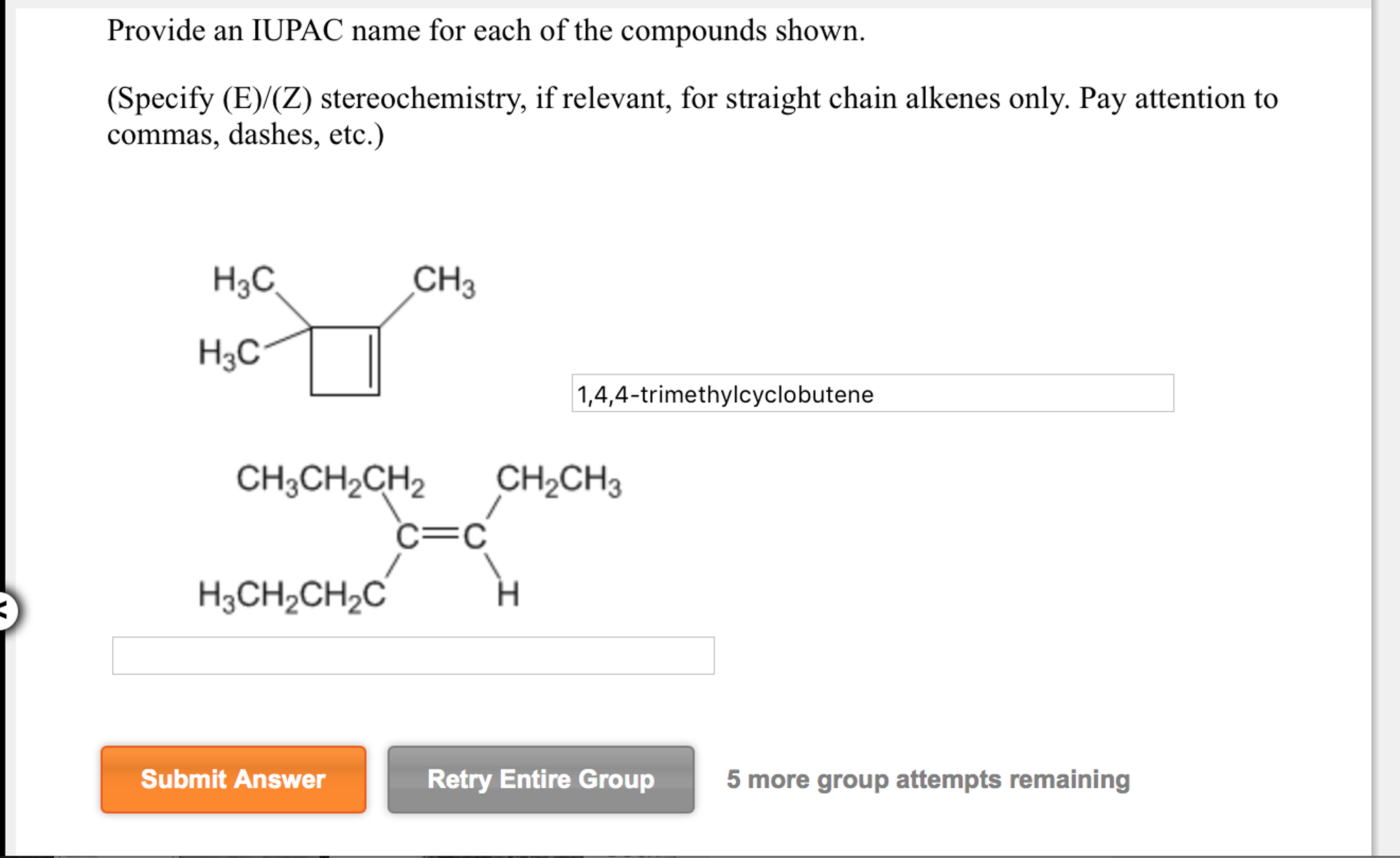
Provide An Iupac Name For Each Of The Compounds Shown

Hey there, chemistry buddies! Ever feel like those IUPAC names for compounds are just a secret code only super-nerds can crack? Well, guess what? You're about to become a secret code cracker yourself! Today, we're diving headfirst into the wonderfully wacky world of naming chemicals, and trust me, it's not as scary as it sounds. Think of it like giving your pet hamster a dignified, official title – it just makes things clearer, right?
We’re going to tackle a few examples, and I promise to keep it light, breezy, and sprinkled with just enough silliness to make sure your brain doesn't fall out. So, grab your favorite beverage (coffee, tea, maybe a mysterious glowing potion you brewed yourself – no judgment here!), and let's get naming!
Let's Decode Some Chemical Cocktails!
Alright, first up, we've got a real classic. Imagine a molecule that's basically a tiny, happy chain of carbon atoms, all linked together like a string of pearls. And sprinkled on this chain are a couple of those famously clingy hydroxyl groups (-OH). You know, the ones that make alcohols… well, alcohols!
This little fella looks something like this:
(Imagine a simple diagram here: a horizontal line of carbons with -OH groups attached to a couple of them. For example, CH3-CH(OH)-CH2-OH or CH3-CH2-CH(OH)-CH2-OH)
So, how do we give this molecular marvel its proper, dignified IUPAC name? It's all about finding the longest continuous carbon chain. This is like the backbone of our molecule. Once we find that, we count the carbons in the chain. For this guy, let's say our longest chain has three carbons.
Three carbons? That screams "propane" in the world of alkanes, right? But wait, we have those hydroxyl groups! When you've got -OH groups hanging around, your parent name becomes an "-ol." So, instead of propane, we're going to be looking at something like "propanol."
Now, here's where the numbering comes in, and it's like assigning house numbers to our carbon chain. We want to give the lowest possible numbers to the carbons that have the functional groups (our hydroxyls) attached. It's like making sure the most important residents get the most convenient addresses. So, we number our three-carbon chain from left to right or right to left, depending on which way gives our -OH groups the smallest numbers.
If the -OH groups are on carbons 1 and 2, for instance, the name would be propane-1,2-diol. The "1,2" tells us exactly where those hydroxyls are chilling. And the "diol" at the end? That "di" means there are two of them! See? It's like a chemical detective story, piecing together clues.
Let's try another variation of this chain. What if we had something like this:
(Imagine: CH3-CH2-CH(OH)-CH2-OH)
This time, our longest carbon chain is four carbons long. So, our parent name is going to be based on "butane." And since we have those handy hydroxyls, it's going to be a "butanol." But wait, how many -OH groups do we have? Still two! So, it's a "butanediol."
Now for the numbering. We number our four-carbon chain to give the hydroxyl groups the lowest possible numbers. If we number from the end that gives us the 1 and 2 positions for the -OH groups, our name becomes butane-1,2-diol. Easy peasy, right? It's like putting labels on everything so nobody gets confused. No more molecular identity crises!
Aromatic Adventures and Their Nifty Names
Okay, let's switch gears from straight chains to something a bit more… ring-tastic! We're talking about aromatic compounds, the cool kids of the organic chemistry world. Think benzene rings – those lovely, six-carbon rings that are super stable and often smell quite pleasant (though don't go sniffing random lab chemicals, please!).
Here’s a common aromatic structure we might encounter:
(Imagine a benzene ring with a methyl group (-CH3) attached.)
This molecule is a benzene ring with a little methyl group hanging off. Benzene itself is, well, benzene. It's our base, our foundation. When you add a methyl group to a benzene ring, it gets a special, common name that's recognized by IUPAC. This special name is toluene.
So, if you see a benzene ring with a -CH3 group, you can happily call it toluene. It’s like when your friend has a nickname that everyone uses – it just sticks! It's a shortcut, a friendly nod to its common identity.
But what if we have more than one substituent on our benzene ring? This is where things get a little more systematic, but still fun! Let's look at this one:
(Imagine a benzene ring with two methyl groups attached, both on adjacent carbons.)
We've got our benzene ring, and now we have two methyl groups. We already know that a methyl-substituted benzene is toluene. So, our parent name is still going to be related to toluene. But we need to specify where those two methyl groups are sitting on the ring relative to each other.
We number the carbons on the benzene ring starting from the carbon attached to the "parent" group (in this case, we can consider one of the methyl groups as the starting point, though it's a bit of a simplification for illustrative purposes). Then, we number around the ring to give the second substituent the lowest possible number.
In this case, since the two methyl groups are on adjacent carbons, we'll number them 1 and 2. So, we have a dimethyl substituted toluene. The IUPAC name would be 1,2-dimethylbenzene. The "1,2" tells us the methyl groups are next to each other. Simple, right?
Now, what if those methyl groups are on carbons 1 and 3 (meaning there's one carbon in between them)?
(Imagine a benzene ring with two methyl groups attached, with one carbon in between them.)
Using the same logic, our name would be 1,3-dimethylbenzene. The numbers are our GPS coordinates for the substituents on the ring.
And if they're on opposite sides of the ring (carbons 1 and 4)?
(Imagine a benzene ring with two methyl groups attached, on opposite sides of the ring.)
You guessed it! It's 1,4-dimethylbenzene.
However, for these 1,2-, 1,3-, and 1,4- arrangements on benzene rings, IUPAC also allows for the use of prefixes: ortho- (o-), meta- (m-), and para- (p-) respectively. So, our examples could also be called:
- o-xylene (for 1,2-dimethylbenzene)
- m-xylene (for 1,3-dimethylbenzene)
- p-xylene (for 1,4-dimethylbenzene)
Xylene is the common name for dimethylbenzene, and these prefixes are super handy and widely used. It’s like having a secret handshake for these specific ring arrangements!
But wait, what if we have different groups on our benzene ring? Let's say we have a hydroxyl group (-OH) and a methyl group (-CH3) on the same ring.
(Imagine a benzene ring with one -OH group and one -CH3 group attached.)
This is where the alphabetical order of substituents comes into play, and it’s like a polite queue at the grocery store – everyone waits their turn! The parent name here will be based on the group that gets priority. In this case, a hydroxyl group on a benzene ring gives us phenol. So, phenol is our parent name.
Now, where is that methyl group? If it’s on the carbon next to the hydroxyl group (carbon 2, if the hydroxyl is on carbon 1), we’d number it accordingly. Since methyl comes before hydroxyl alphabetically, if we weren’t using the common name "phenol," and were just strictly numbering a substituted benzene, the methyl group would get the lower number. But since "phenol" is the established parent name, we number from the carbon bearing the -OH group.
So, if the methyl group is on the carbon next to the -OH, we call it 2-methylphenol. If it's on the carbon with one carbon in between, it's 3-methylphenol. And if it's on the opposite side, it's 4-methylphenol.
Again, for these, we also have those handy common prefixes:

- o-cresol (for 2-methylphenol)
- m-cresol (for 3-methylphenol)
- p-cresol (for 4-methylphenol)
Cresol is the common name for methylphenol. See how those common names sneak in and make things a little friendlier?
Carbonyl Crew: Aldehydes and Ketones
Let's dive into the world of carbonyl compounds! These are molecules that have a carbon-oxygen double bond (C=O). They're super important and come in a couple of main flavors: aldehydes and ketones. The difference is where that C=O group is located in the molecule.
First, the aldehydes. In an aldehyde, the C=O group is at the end of a carbon chain. It’s like the leader of the pack, always at the forefront.
Consider this little guy:
(Imagine: CH3-CH2-CHO)
Here, we have a three-carbon chain. The longest continuous carbon chain is three carbons, which would normally be propane. But because we have that aldehyde group (-CHO) at the end, our parent name ends in "-al." So, propane becomes propanal. The "al" suffix signals that it's an aldehyde, and its position at the end of the chain is implied by the naming convention.
What if our chain was longer? Say, four carbons:
(Imagine: CH3-CH2-CH2-CHO)
This is a four-carbon chain, so our base is butane. With the aldehyde group, it becomes butanal. Easy, right? It’s like giving a specific title to the boss of the chain.
Now, let's look at ketones. In ketones, the C=O group is in the middle of a carbon chain. It’s like the distinguished middle child, not at the edge but definitely important!
Let's take this molecule:
(Imagine: CH3-CO-CH3)

Our longest carbon chain here is three carbons. So, we start with propane. Since we have a carbonyl group in the middle, our parent name will end in "-one." So, propane becomes propanone. And this specific propanone is so common, it has a super-famous common name: acetone!
But what if the chain is longer, and the carbonyl group isn't smack in the middle? Like this:
(Imagine: CH3-CH2-CO-CH2-CH3)
Our longest carbon chain is five carbons. So, we start with pentane. With the carbonyl group, it becomes a "pentanone." Now, we need to specify where that carbonyl group is. We number the chain from the end that gives the carbonyl group the lowest number. In this symmetrical molecule, it doesn't matter which way we number it – the carbonyl will be on carbon 2 or carbon 4, and we choose the lower number!
So, the name is pentan-2-one. The "2" tells us exactly where that little C=O buddy is hanging out. It's like giving directions to that crucial intersection in the middle of town.
What about this one?
(Imagine: CH3-CH2-CH2-CO-CH3)
This is a six-carbon chain, so we start with hexane. It's a ketone, so it'll be a hexanone. Where's the carbonyl? We number from the end that gives it the lowest number. Numbering from the right gives us the carbonyl on carbon 2. So, it's hexan-2-one.
These naming rules, while they might seem a bit like a puzzle at first, are designed to be super logical. They're like the universal language for chemists, ensuring that when someone says "hexan-2-one," everyone on the planet knows exactly which molecule they're talking about. No more playing molecular charades!
The Joy of Naming!
Phew! We've decoded some tricky names and uncovered some molecular identities. See? IUPAC naming isn't some dark art meant to intimidate you. It's a system, a set of rules that brings clarity and order to the amazing diversity of chemical compounds. Think of it as giving each molecule its own unique ID card, complete with its family lineage and its specific address.
Every time you correctly identify a functional group, find the longest carbon chain, and apply the numbering rules, you're doing more than just memorizing. You're building a deeper understanding of how these tiny building blocks of the universe are put together. You're becoming a molecular detective, solving mysteries with every compound you encounter!
So, next time you see a chemical formula that looks like a jumbled mess, take a deep breath, put on your naming hat, and dive in! You've got this. Embrace the process, have fun with it, and remember that each name you decipher is a little victory, a step closer to mastering the beautiful language of chemistry. Keep exploring, keep learning, and keep smiling – the world of molecules is waiting for you!
