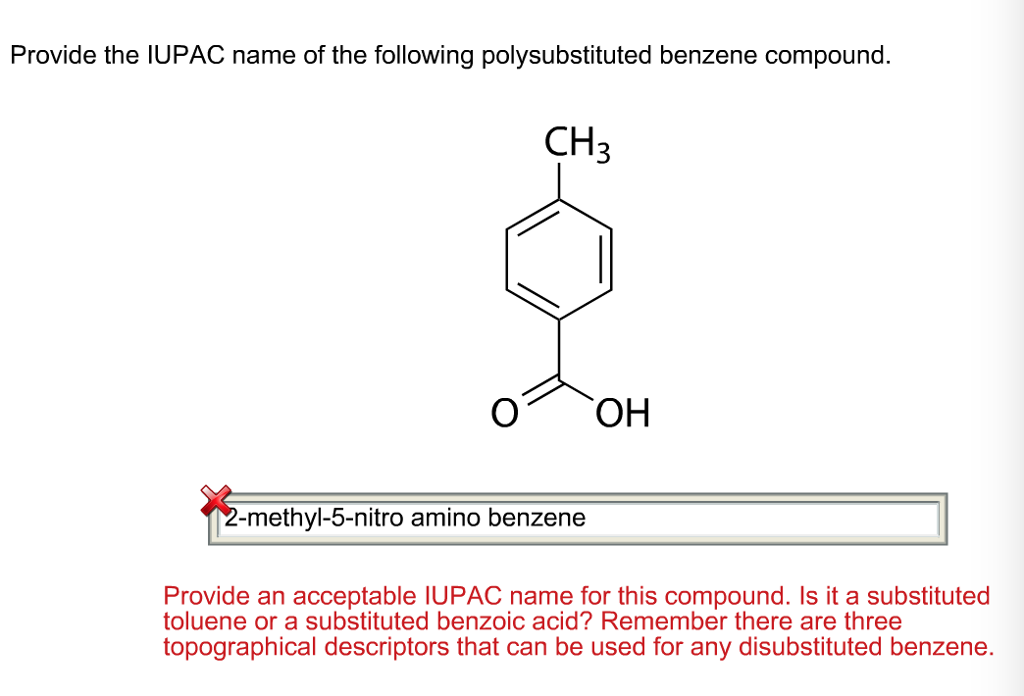
Provide An Acceptable Iupac Name For The Following Polysubstituted Benzene.
Ever wondered how scientists manage to give names to incredibly complex molecules, like those found in everyday medicines or fascinating natural compounds? It's not just a random jumble of letters; there's a clever, standardized system at play called the International Union of Pure and Applied Chemistry (IUPAC) nomenclature. Today, we’re going to peek behind the curtain at how this system works for a common class of molecules: polysubstituted benzenes. Think of it as learning the secret handshake for naming chemical structures, which can be surprisingly fun and incredibly useful!
The purpose of IUPAC naming is to provide a unique and unambiguous name for every chemical substance. This is absolutely crucial for clear communication in science, research, and industry. Imagine trying to order a specific chemical for an experiment if everyone used their own, informal names! Chaos would ensue. For polysubstituted benzenes, meaning benzene rings with multiple different groups attached, IUPAC naming helps us understand the molecule's structure at a glance. This, in turn, can give us clues about its properties, how it might react, and even its potential uses. It’s like having a built-in roadmap to the molecule's identity.
Where do we see this in action? Well, the most direct place is in education. Chemistry students spend a good amount of time learning these naming rules, and it’s a foundational skill for anyone pursuing a career in chemistry, pharmacy, or even biology. Beyond textbooks, these names are critical in scientific literature, patents, and regulatory documents. When a new drug is developed, for example, its IUPAC name is essential for identification and safety. Even in everyday life, the ingredients listed on some advanced cosmetics or cleaning products might subtly hint at the underlying chemistry, and their formal names are derived from these IUPAC principles.
So, how do we approach naming a polysubstituted benzene? The core idea is to identify the parent structure, which in this case is benzene. Then, we identify all the attached groups (substituents) and their positions on the ring. The IUPAC system has specific rules for assigning numbers to the carbon atoms of the benzene ring, ensuring everyone describes the same arrangement. We then list the substituents alphabetically, along with their positional numbers, before the word "benzene." For example, if a benzene ring has a chlorine atom at position 1 and a methyl group (CH3) at position 2, the name would be 1-chloro-2-methylbenzene (though this specific compound also has a common name!).
Ready to explore a bit yourself? You don’t need a lab coat! A great way to start is by looking at simple examples online. Search for "examples of IUPAC naming of polysubstituted benzenes." You’ll find diagrams that clearly show the structure and the corresponding name. Try to identify the parent ring and the substituents yourself before checking the answer. You can also find apps or online quizzes that test your naming skills. It’s a bit like solving a puzzle, and with a little practice, you’ll start to see the elegance and logic behind these chemical names. The more you practice, the more intuitive it becomes!
