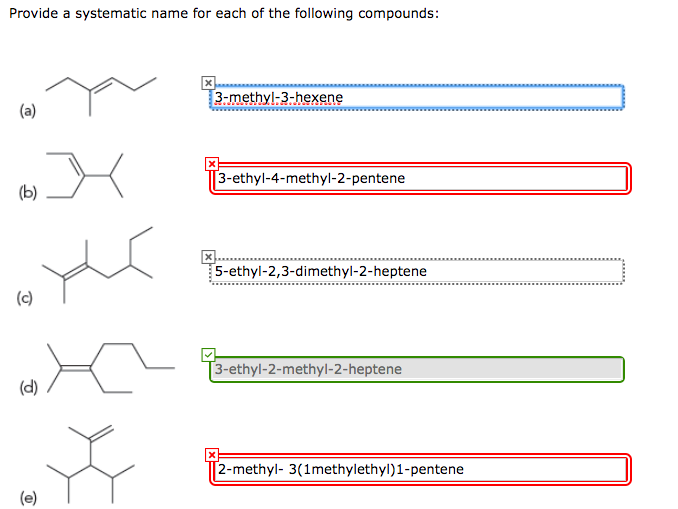
Provide A Systematic Name For The Following Compound:
Hey there, fellow chemistry adventurers! So, you’ve stumbled upon a mysterious molecule and you’re wondering, "What in the name of all that's holy is THIS thing?" Well, fear not, my friends, because today we’re going to crack the code of systematic naming. Think of it like giving your pet rock a proper, fancy scientific name. No more "Rocky," it's now "Lithos Petramorpha," or something equally impressive.
Today, we’re going to tackle a specific compound. Don't worry, we won't be diving into rocket science or quantum mechanics (unless, of course, you've accidentally synthesized a miniature black hole, in which case, please call me!). We’re keeping it fun, breezy, and, dare I say, even a little bit deliciously nerdy.
So, let's imagine you've got this guy right in front of you. It looks a bit like a tiny, intricate LEGO creation, doesn't it? We need to give it its official, universally recognized, "if you want to publish a paper about it, this is what you call it" name. This isn't just for fun; it's how scientists worldwide communicate, so we don't end up with fifty different names for the same exact thing. Imagine trying to order a pizza with fifty different names for pepperoni – chaos!
Unveiling Our Mystery Guest!
Alright, so here's the star of our show. (Now, I’d usually draw a picture here, but alas, my artistic skills are… well, let’s just say they’re best suited for stick figures that look vaguely like terrified spiders. So, you'll have to use your imagination, or, you know, grab a piece of paper and sketch it out as we go. That’s how the real scientists do it, probably while wearing lab coats and looking incredibly serious. Or maybe they’re just really good at doodling.)
Let's break down our compound. It's got a few key features we need to pay attention to. Think of it like meeting a new person. You look at their hair, their eyes, their clothes, and maybe their questionable taste in music before you decide on a nickname. For chemicals, it's similar, but way more structured. And thankfully, no questionable music taste involved!
Step 1: Find the Parent Chain – The Backbone of the Operation!
The first thing we do is identify the longest continuous chain of carbon atoms. This is like finding the main highway in a city. Everything else is going to be attached to this highway, or driving on it, if you want to stretch the analogy. The longer the chain, the "bigger" the parent molecule.
In our case, let's say we’ve carefully counted the carbons and found our longest chain has six carbon atoms. Six carbons? That immediately tells us something important. We don’t call it "one carbon," "two carbons," or even "the slightly-more-than-a-handful-of-carbons molecule." Nope. For six carbons, the parent name is hexane. Easy peasy, right?
Now, if it were a ring of six carbons, it would be cyclohexane. But this one is a straight-up, no-nonsense chain. Think of it as a caterpillar rather than a bicycle wheel. Both have six wheels… I mean, carbons… but they’re structured differently. And the names reflect that. So, our backbone is definitely hexane.
Step 2: Identify the Substituents – The Fun Little Extras!
Next up, we look for anything that’s not part of that main carbon chain. These are called substituents, or sometimes functional groups if they’re particularly fancy. Think of them as the cool accessories or the extra toppings on our pizza. They’re what make our hexane molecule more than just a boring old six-carbon chain.
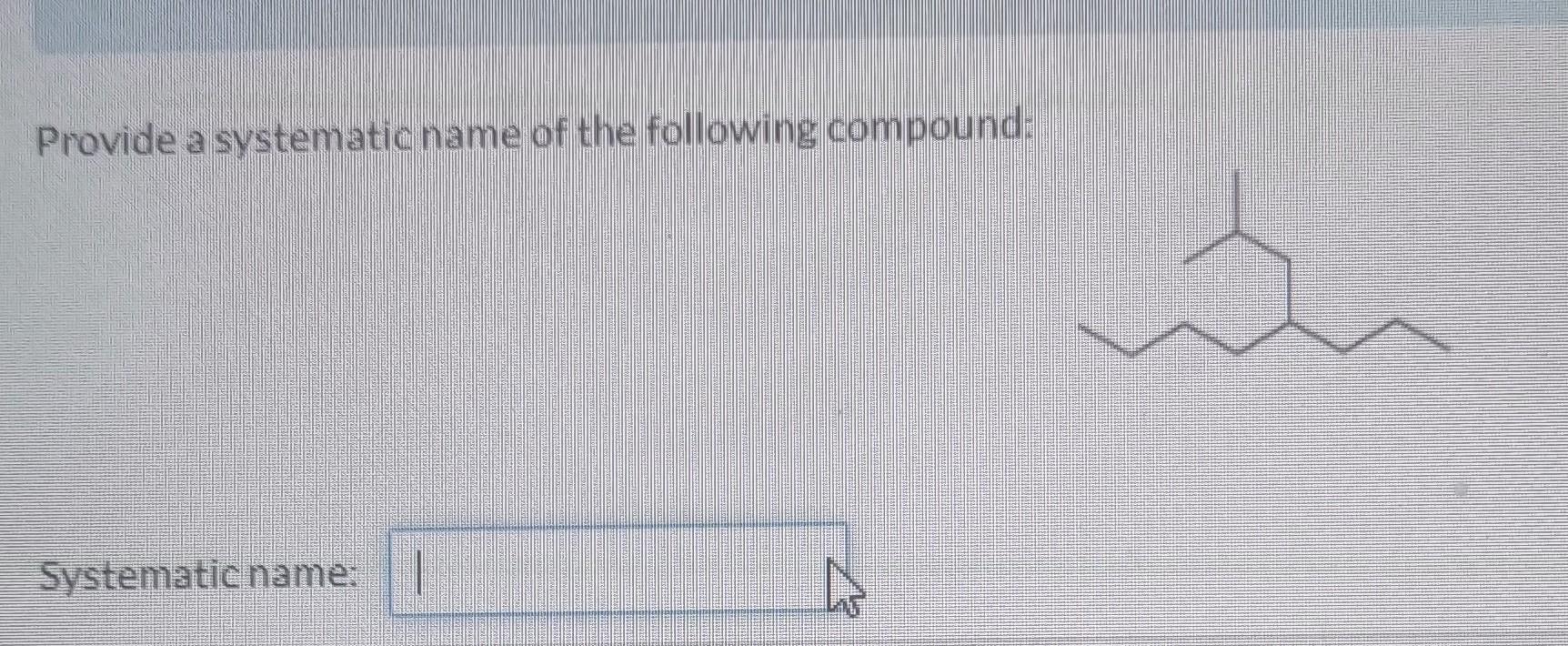
Let’s say our molecule has a couple of these little extras hanging off the hexane chain. We need to figure out what they are. Common substituents include things like:
- A single carbon atom with three hydrogens attached (CH₃). This guy is called a methyl group. It’s like the tiny but mighty little sibling of the carbon chain.
- Two carbon atoms with five hydrogens attached (CH₂CH₃). This one is an ethyl group. It’s the slightly older, more established sibling of the methyl group.
- Halogens like chlorine (Cl), bromine (Br), or fluorine (F). These are usually named as chloro, bromo, or fluoro, respectively. They’re like the sparkly, metallic decorations.
For our example, let’s imagine we have two methyl groups attached to our hexane chain. So, we’ve got our hexane backbone, and then two little CH₃ buddies clinging on. They’re not part of the main six, so they get their own names.
Step 3: Number the Chain – Where Are These Guys Hanging Out?
Now for the really crucial part: figuring out where these substituents are attached. We need to give them an address. To do this, we number the carbon atoms in our parent chain. The catch? We have to number it in a way that gives our substituents the lowest possible numbers. It’s like finding the shortest route to a coffee shop – you want the most efficient path, right?
So, we number the hexane chain from either end. Let's say we number it 1, 2, 3, 4, 5, 6 from left to right. Or we could number it 6, 5, 4, 3, 2, 1 from right to left. We need to pick the direction that puts our substituents on the smallest numbers.
If, for instance, our two methyl groups are on the second and fourth carbon atoms when numbering from the left, we’d have substituents at positions 2 and 4. If we numbered from the right, they might be at positions 3 and 5. Since 2 and 4 are smaller than 3 and 5, we’re definitely going to number from the left. This is the golden rule: lowest possible numbers for the substituents. It’s like getting the best seats in the house!
So, we've decided: numbering starts from the end that gives the lowest numbers. Let's say our methyl groups are on carbons 2 and 4.

Step 4: Putting it All Together – The Grand Finale!
Now we assemble our fancy scientific name. We have our parent name (hexane), our substituents (methyl), and their locations (2 and 4).
When we have multiple identical substituents, we use prefixes to indicate how many there are. So:
- Two methyl groups? That's dimethyl.
- Three methyl groups? That's trimethyl.
- Four? tetramethyl. And so on.
It’s like a little chemical counting game. Very satisfying, if you ask me.
So, we have two methyl groups. Therefore, they are dimethyl. And they are located at positions 2 and 4.
The final name is constructed by putting the substituent names first, with their locants (the numbers), followed by the parent name. We separate the numbers with commas and the numbers from the substituent names with hyphens. And we put the numbers in ascending order.
So, our compound would be named: 2,4-dimethylhexane.
Ta-da! We’ve successfully given our mystery molecule its official title. It's no longer a nameless entity; it's a precisely identified chemical specimen. Pretty neat, huh?
A Quick Recap of Our Naming Adventure
Let’s do a quick mental run-through to make sure it all sinks in. We:
- Found the longest carbon chain – that's our parent name (hexane).
- Identified the groups hanging off that chain – our substituents (methyl).
- Numbered the chain from the end that gave the substituents the lowest possible numbers (2 and 4).
- Combined the information, using prefixes for multiple identical groups and locants to show their positions (2,4-dimethylhexane).
It’s a systematic process, like following a recipe. If you stick to the steps, you’ll end up with a delicious chemical name every time. And unlike some recipes, there's no risk of burning anything (unless you’re actually heating it up, which we aren’t doing in this naming exercise. Please be careful in the lab!).
What if it Was More Complicated?
Okay, I know what you might be thinking. "That was… almost too easy. What if it has different groups attached? What if it’s a ring? What if it smells like old socks?"
Well, my friends, the world of organic chemistry nomenclature is vast and, at times, gloriously complex. There are rules for double and triple bonds (alkenes and alkynes), rings (cycloalkanes), and a whole menagerie of functional groups like alcohols (ending in -ol), ketones (ending in -one), and carboxylic acids (ending in -oic acid). Each has its own set of rules and its own special suffix.
But the fundamental principles remain the same: find the longest chain, identify the substituents, number strategically, and put it all together in a logical order. It’s like learning to read. First, you learn the alphabet, then words, then sentences. We’re just learning the chemical alphabet and sentence structure today!
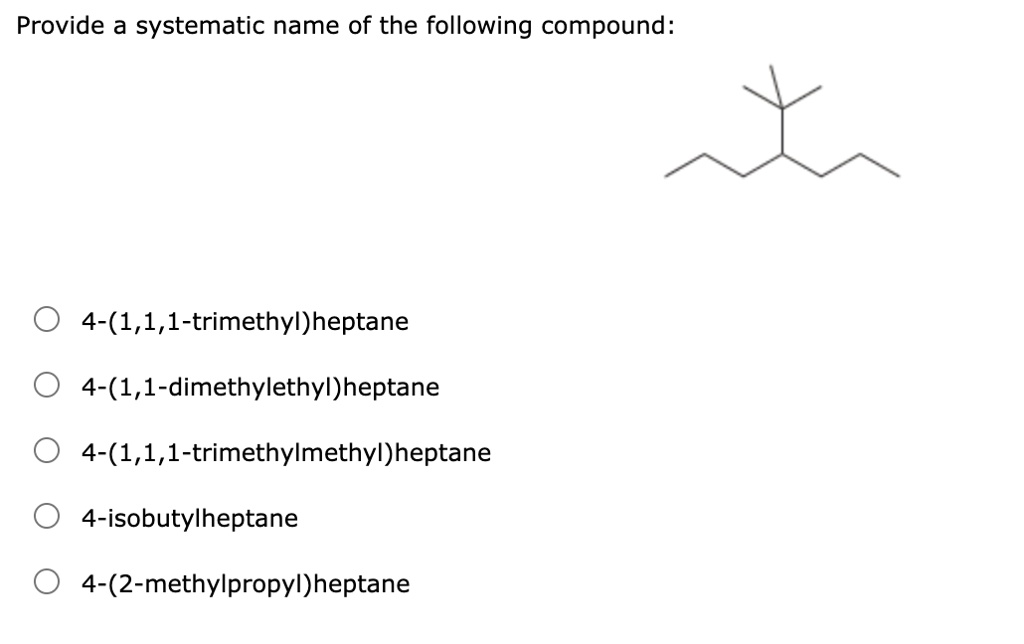
And don’t even get me started on stereochemistry. That’s where molecules can be mirror images of each other, like your left and right hands. But we’ll save that for another day, perhaps when we’re all feeling particularly adventurous and have consumed copious amounts of caffeine. For now, let’s bask in the glory of our 2,4-dimethylhexane.
Why Bother with All This Naming Stuff?
You might be wondering, "Why all this fuss? Can't we just draw it?" And yes, drawing is fantastic, and chemists do it constantly. But names are powerful. They're concise. They’re a shortcut to understanding the structure of a molecule.
When you hear "2,4-dimethylhexane," a chemist immediately pictures that six-carbon chain with two methyl groups on carbons 2 and 4. It tells them about its size, its shape, and hints at its potential chemical behavior. It’s like having a secret handshake for molecules.
This systematic naming convention is a testament to human ingenuity – our ability to create order out of complexity. It allows scientists across the globe to communicate clearly, to build upon each other’s discoveries, and to collectively unravel the mysteries of the universe, one molecule at a time.
So, the next time you encounter a chemical formula, don't feel intimidated. See it as a puzzle, a challenge, and an opportunity to learn. Embrace the systematic approach, follow the rules, and you'll find yourself speaking the beautiful, logical language of chemistry in no time. And remember, even the most complex molecules started with a simple carbon chain and a few brave substituents. You've got this!
Keep exploring, keep questioning, and never stop being amazed by the tiny, fascinating world of molecules. You’re well on your way to becoming a nomenclature ninja, and that’s something to truly smile about!
