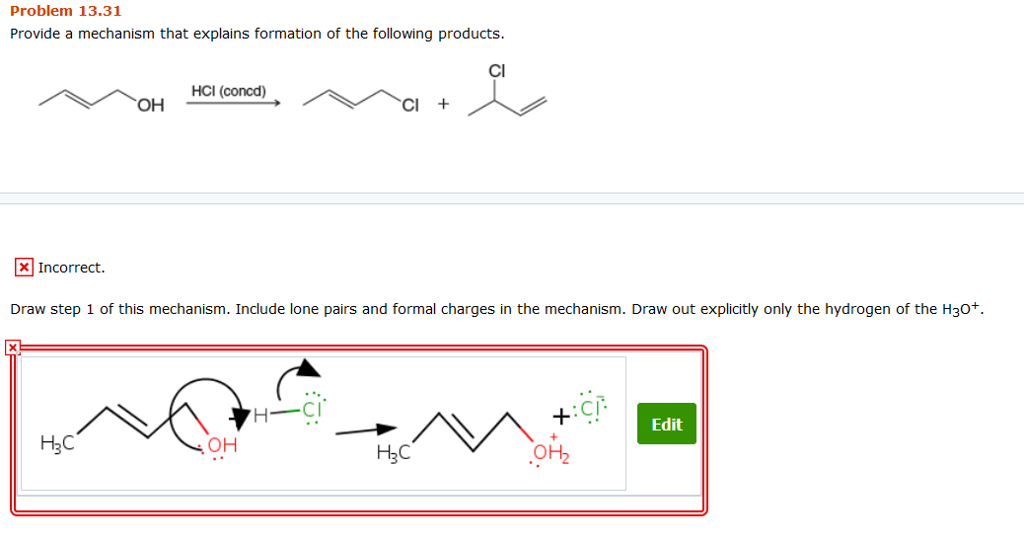
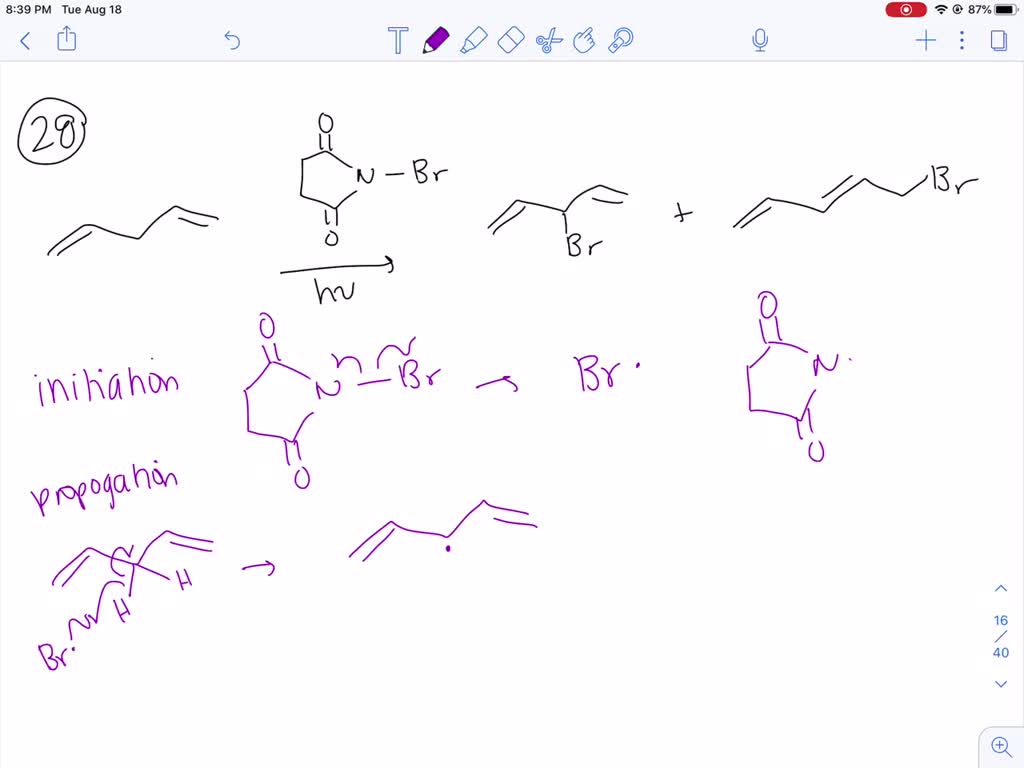
Provide A Mechanism That Explains Formation Of The Following Products
Ever looked at a perfectly formed snowflake and wondered how it got those intricate arms? Or maybe you've been fascinated by how a tiny seed can grow into a towering tree? There's a hidden world of remarkable transformations happening all around us, and understanding the mechanisms behind them can be surprisingly fun and deeply rewarding. Today, we're going to peek behind the curtain and explore a fascinating concept that explains how simple ingredients can come together to create the diverse and often beautiful products we see in nature and beyond.
At its core, this is all about cause and effect. We're talking about the specific, step-by-step processes that lead to a particular outcome. Think of it like a recipe. Just as following the right steps with the right ingredients leads to a delicious cake, nature and science follow precise pathways to build everything from the smallest molecule to the grandest ecosystem. Understanding these mechanisms allows us to not only appreciate the complexity of the world but also to harness these processes for our own benefit.
The benefits of understanding these formation mechanisms are vast. In education, it's the backbone of subjects like chemistry, biology, and physics. Imagine a biology class explaining how DNA replicates – that's a mechanism at play! Or a chemistry lesson detailing how water forms from hydrogen and oxygen – another mechanism! In our daily lives, these principles are everywhere. When you see rust form on metal, that's a chemical mechanism. When your phone battery charges, it's an electrochemical mechanism. Even understanding why bread rises involves the fascinating mechanism of yeast fermentation.
Let's consider a common example: the formation of salt crystals. You might think salt is just… salt. But when you see those perfect little cubes, there’s a mechanism at work. Dissolve some table salt (sodium chloride) in water. As the water evaporates, the dissolved salt ions (sodium and chloride) start to get closer together. They arrange themselves in a very specific, repeating pattern – a crystal lattice – driven by their electrical charges and the available space. This ordered arrangement is the mechanism that results in those beautiful, symmetrical salt crystals you might find left behind on a plate.
Another everyday marvel is the formation of a rainbow. It’s not magic; it’s optics! When sunlight, which is made up of all the colors of the spectrum, hits raindrops, it bends, or refracts. Because each color bends at a slightly different angle, the white light is split into its constituent colors, creating the arc we see. The mechanism here is the interaction of light waves with water droplets, causing dispersion.
So, how can you explore these ideas yourself? It's simpler than you think! Try the salt crystal experiment at home – it’s a fantastic visual. Observe how water evaporates from a shallow dish of salt water over a few days and see the crystals form. For rainbows, next time it rains and the sun comes out, look for that familiar arc. Notice the order of the colors – red on the outside, violet on the inside. You can even experiment with a garden hose on a sunny day to create miniature rainbows. By paying attention to the "how" behind everyday phenomena, you'll start to see the world with a whole new sense of wonder and understanding. It’s a journey of discovery that’s always in progress!
