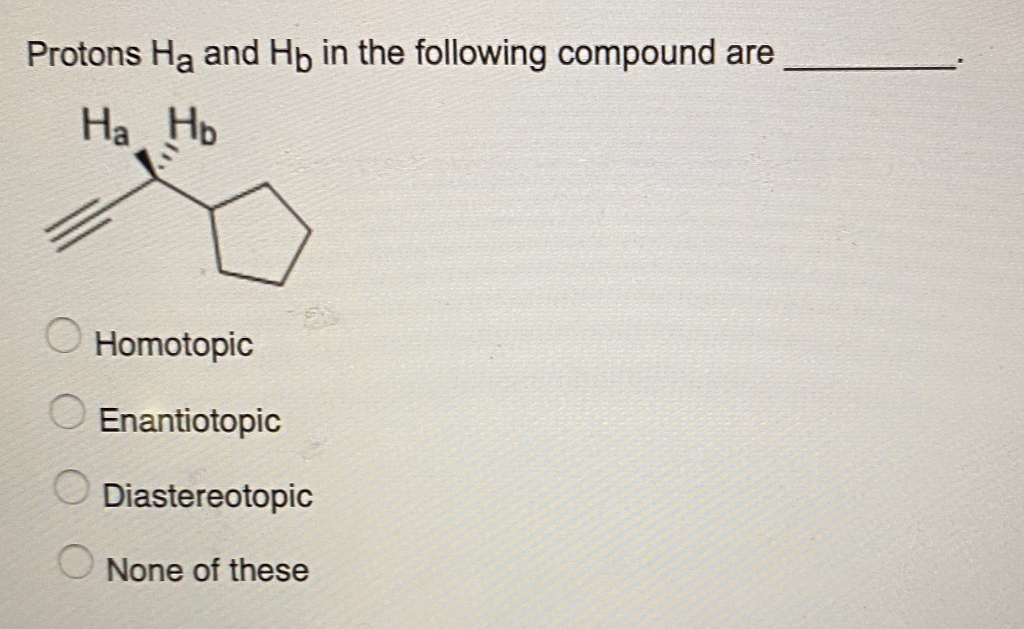
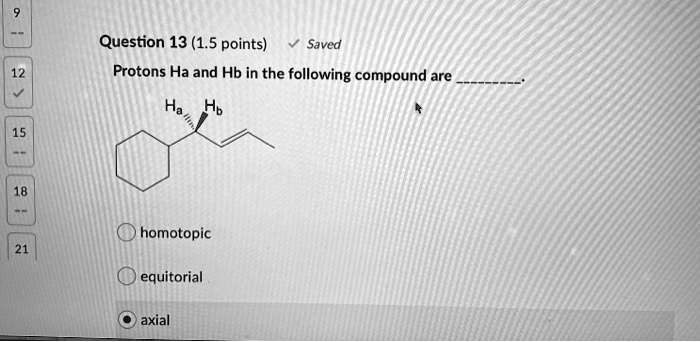
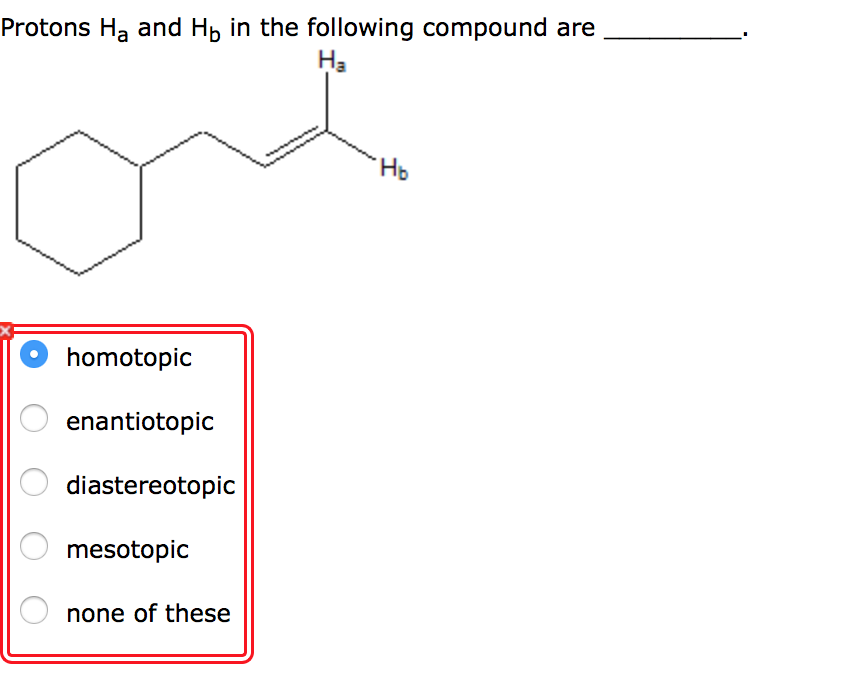
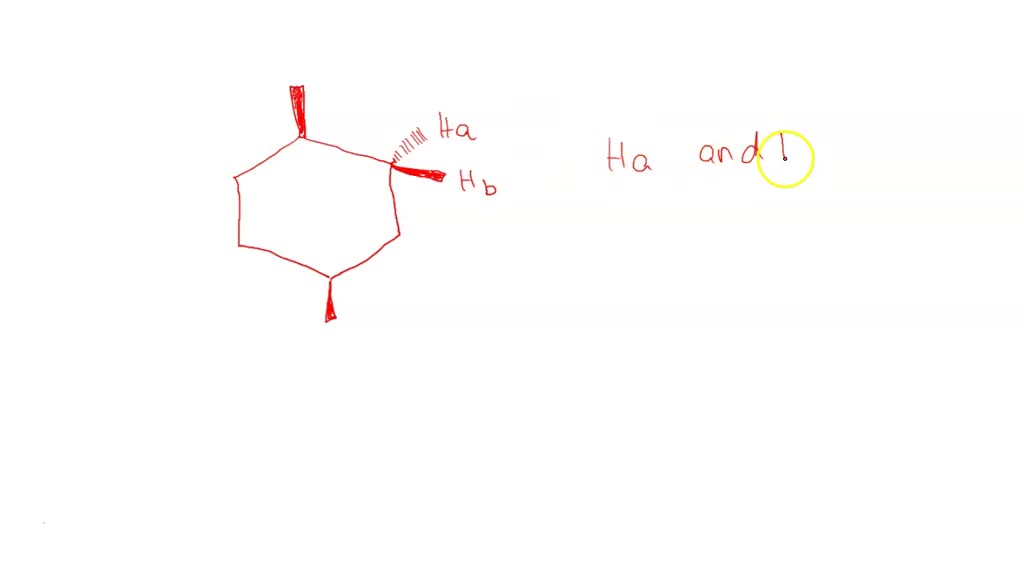
Protons Ha And Hb In The Following Compound Are

Hey there, science curious friends! Ever looked at a fancy chemical name and thought, "Whoa, what's going on there?" It's like trying to read a secret code, right? Well, today we're going to decode a little bit of that magic, and I promise, it's going to be way more fun than deciphering your teenager's text messages. We're going to chat about something called "protons Ha and Hb" in a specific compound. Sounds a bit like a quirky superhero duo, doesn't it? Like, "Here comes Proton Ha, and alongside him, the mighty Proton Hb!"
So, what exactly are these "protons" we're talking about? Think of atoms as tiny Lego bricks that build everything around us – your coffee mug, your favorite comfy chair, even you! And inside these Lego bricks are even smaller bits. Protons are one of these bits. They're like the positive energy centers of the atom. They have a little positive charge, kind of like a friendly nudge of encouragement.
Now, when we talk about protons in a compound, we're not just talking about any old protons. We're talking about protons that are part of a specific molecule – a little group of atoms joined together. Imagine a group of friends holding hands to form a circle. Each friend is an atom, and they're all linked up, forming a molecule. The protons are inside those friends, giving them their unique personalities, if you will.
The "Ha" and "Hb" are just ways chemists give specific protons nicknames. It's like in a family with a few Johns – you might call one "John the Tall" and another "John the Younger." In our chemical compound, "Ha" refers to a proton that's in a particular spot, and "Hb" refers to another proton in a slightly different spot. It all depends on where these protons are hanging out within the molecule.
Why should you even care about these tiny guys and their little nicknames? Well, these protons, and where they are, can tell us a ton about how a molecule behaves. It’s like knowing if your friend is a morning person or a night owl – it tells you a lot about their daily habits and how they might interact with you. Similarly, the position of these protons can influence how a molecule reacts with other molecules, how it dissolves in water, or even how it smells!
Think about it this way: imagine you have two identical twins. They look the same, but one spent their childhood learning to play the piano, and the other learned to play the guitar. Even though they started from the same place, their experiences (like the protons' positions!) have made them unique and shaped their abilities. The same goes for our chemical friends. Protons in slightly different environments within a molecule can lead to surprisingly different behaviors.
So, this "compound" we're talking about? It's just a specific arrangement of atoms. And within that arrangement, there are these protons, labeled Ha and Hb, because they're in distinct neighborhoods within the molecule. This distinction is important because it can influence how that molecule interacts with the world.
Let's get a little more specific without getting too technical. Imagine a molecule is like a little dance troupe. Each dancer (atom) has a role. Protons are like the energy and personality of each dancer. Now, if one dancer is always on the left side of the stage and another is always on the right, they'll have different experiences during the performance. Maybe the dancer on the left gets more spotlight, or maybe the dancer on the right has to interact with the backdrop more. These different positions and interactions are what "Ha" and "Hb" are trying to capture.
These protons, depending on their chemical "neighborhood," can behave a bit differently. It’s like how a word can sound slightly different depending on the accent of the person saying it. The core word is the same, but the pronunciation (or in our case, the chemical behavior) can vary. Protons in different chemical environments might be more or less likely to be "shared" with another molecule, or they might influence the electrical charge distribution in different ways.
This is super important in a lot of areas, even if you don't realize it! For example, in medicine, understanding how a drug molecule interacts with your body often comes down to the fine details of its atoms and their arrangement, including the behavior of specific protons. It's like knowing exactly which key fits which lock. If the protons (Ha and Hb) in a drug molecule are in a certain position, they might be able to "unlock" a specific target in your body, like a receptor on a cell.
Or consider the fascinating world of flavors and scents. Why does one molecule smell like roses and another like a skunk? It’s often down to subtle differences in their structure, and those differences can be influenced by the electronic environment around specific protons. So, the "Ha" and "Hb" protons are, in a way, contributing to the unique scent profile of our compound. It’s like a chef carefully selecting ingredients and understanding how they'll combine to create a delicious dish!
In simpler terms, when chemists talk about "Protons Ha and Hb," they're just being very precise about which protons they're interested in within a given molecule. They’ve given them little labels to distinguish them because these protons, even though they're both protons, are in different "places" or have different "friends" (other atoms) nearby. And these differences matter! They influence how the molecule acts, how it connects with other molecules, and ultimately, what its properties are.
It’s a bit like looking at two identical water bottles. One has a sticker on it, and the other doesn't. They’re both water bottles, but the sticker (like the different proton environments) gives them a slightly different identity or purpose in certain contexts. One might be a promotional giveaway, the other just a plain bottle.
So, next time you see something like "Protons Ha and Hb," don't let it intimidate you! Just remember it's a way of pointing to specific, important parts of a molecule that contribute to its overall story. These little protons are the unsung heroes, dictating how molecules interact and, in turn, shaping the world around us in ways we might never even think about. They're the tiny details that make all the difference, from the medicine that heals you to the food that tastes delicious!
It’s a reminder that even the smallest things can have a big impact. These protons, with their humble positive charge, are fundamental building blocks of reality, and understanding their subtle differences is key to unlocking so many scientific mysteries. So, let's give a little cheer for protons Ha and Hb – the tiny but mighty influencers of the molecular world!
