Protein Folding And Protein Structure Worksheet Answers

Hey there, protein-folding enthusiasts! So, you’ve been wrestling with those pesky protein structure worksheets, huh? Don't worry, we’ve all been there. It’s like trying to fold a fitted sheet – looks simple, but suddenly you’re in a tangled mess of amino acids. But fear not! Today, we’re going to dive into the wonderful world of protein folding and, more importantly, shed some light on those worksheet answers. Think of this as your friendly guide, your protein-folding whisperer, here to untangle those knots and bring a smile to your face. Ready to fold some knowledge?
First off, let’s get real for a sec. Protein folding sounds super scientific, and it is, but at its heart, it’s just about a long chain of amino acids getting its act together. Imagine you have a super long string of beads, right? Each bead is an amino acid. Now, this string doesn’t just float around all willy-nilly. It needs to fold up into a specific, 3D shape. Why? Because that shape is, like, its job description! A protein’s function is super tied to its 3D structure. Mess up the shape, and the protein might go on strike, or worse, start causing trouble.
The Nitty-Gritty of Folding: Why Bother?
So, why is this whole folding thing such a big deal? Well, think of it like building with LEGOs. You have all these individual bricks (amino acids), but you need to snap them together in a precise way to build, say, a cool spaceship. Proteins are the LEGOs of your body, and their folded shape is the spaceship. Without the right shape, that enzyme won’t catalyze its reaction, that antibody won’t bind to its target, and that structural protein will just be… floppy. And nobody wants a floppy structural protein, trust me.
The driving force behind all this folding is usually something called the hydrophobic effect. Sounds scary, right? But it’s actually pretty simple. Imagine you have a bunch of oily (hydrophobic) beads and some watery (hydrophilic) beads. In water, those oily beads are going to try their best to avoid the water, like they’re at a pool party and forgot their swim trunks. So, they’ll clump together in the middle, away from the watery surroundings. The hydrophilic beads, on the other hand, are happy to hang out with the water, so they’ll stick to the outside. This is a huge part of how proteins fold into their compact, functional shapes.
Then there are other forces at play, like hydrogen bonds. These are like little sticky notes holding different parts of the chain together. They’re weaker than the covalent bonds that hold the amino acid chain together in the first place, but when you have a ton of them, they create a really stable structure. It’s like a million tiny hugs keeping everything in place.
Levels of Protein Structure: It’s Like Layers of a Cake!
Now, worksheets often talk about different levels of protein structure. Think of these as the different layers you need to understand to get the whole picture. It’s not just one big blob; it’s a carefully organized structure.
Primary Structure: The Amino Acid Sequence
This is the most basic level, the actual sequence of amino acids in the chain. It’s like the order of LEGO bricks on your string before you even start building. For example, you might have a sequence like Gly-Ala-Val-Leu-Ser. This sequence is determined by your DNA, so it’s the blueprint! If there’s a typo in the DNA, there’s a typo in the primary sequence, and that can lead to all sorts of drama. Ever heard of sickle cell anemia? That’s a classic example of a single amino acid change in a protein causing big problems. A real-world tragedy, but a fascinating illustration of how important this primary sequence is.
Worksheets might ask you to identify the primary structure given a list of amino acids or even a short DNA sequence that codes for it. Remember, it’s just the order, no folding yet. Easy peasy, lemon squeezy! Well, maybe not always lemon squeezy, but definitely the foundational step.
Secondary Structure: The Local Folds
Moving on to the next layer! Secondary structure refers to the localized, regular folding patterns that occur within short segments of the polypeptide chain. The two most common ones are the alpha-helix and the beta-pleated sheet. Imagine your string of beads starting to coil up like a spring (alpha-helix) or fold back on itself like a concertina (beta-pleated sheet). These shapes are stabilized by hydrogen bonds between the backbone atoms of the amino acids.
The alpha-helix looks like a corkscrew, with the side chains (the R-groups, remember those? The parts that make each amino acid unique) sticking out. The beta-pleated sheet is formed when segments of the chain lie side-by-side, either parallel or antiparallel, and are held together by hydrogen bonds. It looks a bit like a corrugated piece of cardboard. So, if your worksheet shows a coiled segment, it's likely an alpha-helix. If it shows a zig-zagging, flattened segment, it's probably a beta-pleated sheet. Pay attention to those little diagrams!
Sometimes, worksheets will show you representations of these structures and ask you to identify them. Look for the helical shape for alpha-helices and the parallel or anti-parallel strands for beta-sheets. It’s like a protein structure scavenger hunt!
Tertiary Structure: The Overall 3D Shape
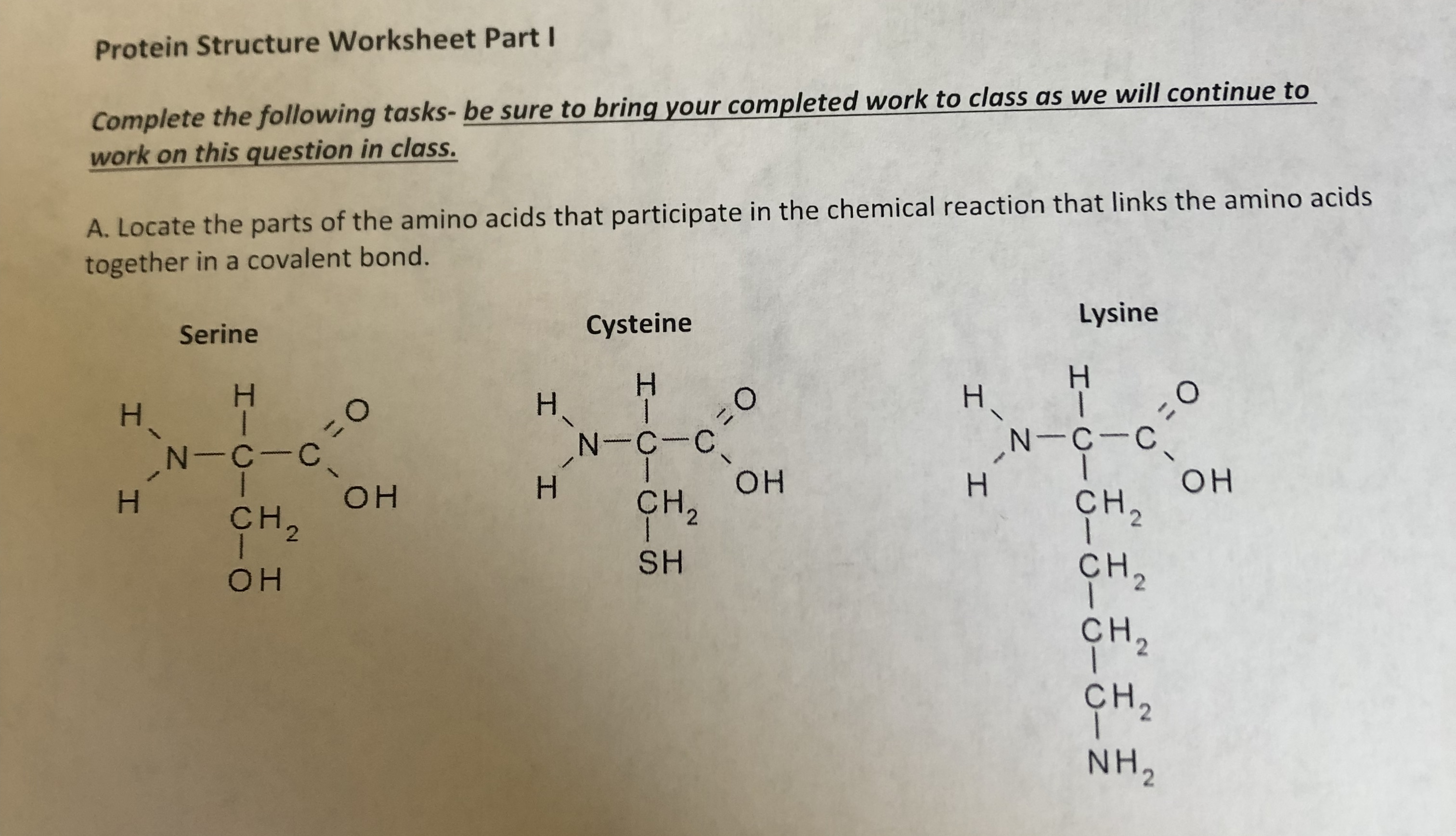
Now we're talking about the real deal! Tertiary structure is the complete, three-dimensional folded shape of a single polypeptide chain. This is where all the local folds (secondary structures) and the regions between them come together to form the protein’s overall conformation. Think of it as taking all those coiled springs and pleated sheets and folding them all up into one compact, functional package. This is where all those different types of bonds and interactions come into play: hydrophobic interactions, hydrogen bonds, ionic bonds (between charged amino acids, like tiny magnets), and even disulfide bridges (covalent bonds between cysteine amino acids – those are super strong!
This is the level that defines a protein’s function. A globular protein, for example, will be tightly folded and soluble in water, perfect for enzymes. A fibrous protein, on the other hand, will be more elongated and stringy, ideal for structural roles like in hair or muscle. Your worksheet might ask you to describe the overall shape or identify the types of bonds stabilizing the tertiary structure. This is where understanding those R-groups and their properties (hydrophobic, hydrophilic, charged) really pays off!
Sometimes, you’ll see diagrams of proteins that look like abstract sculptures. That’s a representation of tertiary structure. The colors and shapes often represent different secondary structures and the overall fold. It’s like art, but with a biological purpose!
Quaternary Structure: When Proteins Team Up
Finally, we have quaternary structure. This level only applies to proteins that are made up of more than one polypeptide chain (called subunits). Think of it as multiple independent protein sculptures coming together to form a larger, more complex machine. Hemoglobin, the protein that carries oxygen in your blood, is a classic example. It's made of four separate polypeptide chains that fit together perfectly. Another fun one is collagen, which forms long, rope-like structures from multiple subunits. This assembly is held together by similar types of interactions as tertiary structure.
Worksheets might present you with models or diagrams showing multiple interacting protein subunits and ask you to identify the quaternary structure. The key here is recognizing that it’s not just one chain folded up, but multiple chains interacting. It’s like a protein band getting together for a jam session!
Common Protein Folding Worksheet Questions and How to Ace Them!
Alright, let’s get down to the nitty-gritty of those worksheet answers. What kind of questions are you likely to encounter, and how can you conquer them?
1. Identifying Amino Acid Properties
You'll often see questions asking you to predict how a protein might fold based on the amino acids present. For example: "If a protein segment is rich in leucine and alanine, where is it likely to be located within the folded protein?"
The Answer Key Says: Leucine and alanine are hydrophobic amino acids. They want to escape the watery environment. Therefore, they will tend to cluster together in the interior of the folded protein, away from the water. Think of it as the oily part of your salad dressing sinking to the bottom!

Your Smarty-Pants Reply: "Since leucine and alanine are hydrophobic, they'll be found in the protein's core, shielded from the aqueous environment." High fives all around!
2. Recognizing Secondary Structures
You might be shown a diagram of a protein segment and asked to identify it as an alpha-helix or a beta-pleated sheet.
The Answer Key Says: Look for the characteristic shapes. A coiled, spiral structure is an alpha-helix. A more flattened, zig-zagging structure, often shown with arrows indicating the direction of the polypeptide chain, is a beta-pleated sheet.
Your Smarty-Pants Reply: "Ah, that swooshy one is clearly an alpha-helix, and the one that looks like a neatly folded fan is a beta-pleated sheet!" Nailed it!
3. Predicting Interactions in Tertiary Structure
Questions might describe specific amino acids within a protein chain and ask about the type of interaction that might occur between them to stabilize the tertiary structure.
The Answer Key Says: * If you have two positively charged amino acids (like lysine or arginine), they will repel each other (ionic repulsion). * If you have a positively charged amino acid and a negatively charged amino acid (like aspartic acid or glutamic acid), they will be attracted to each other (ionic bond, salt bridge). * If you have two nonpolar amino acids (like valine or isoleucine), they will likely interact via hydrophobic interactions. * If you have polar uncharged amino acids (like serine or threonine), they can form hydrogen bonds with each other or with the surrounding water.

Your Smarty-Pants Reply: "Okay, so a lysine is hanging out with an aspartic acid? That's a classic salt bridge forming! And those two valines are definitely going to huddle up to avoid the water." You're basically a protein whisperer now.
4. Understanding Denaturation
Worksheets often touch on what happens when a protein loses its shape – denaturation. This can be caused by heat, extreme pH, or certain chemicals. Think of it as a protein having a really bad day and forgetting how to fold correctly.
The Answer Key Says: Denaturation disrupts the non-covalent bonds and interactions that maintain the protein’s secondary, tertiary, and quaternary structures, leading to a loss of function. Crucially, denaturation does not break the primary structure (the peptide bonds). You can sometimes refold a denatured protein if the conditions are right, but if the primary structure is broken (like through digestion), it’s game over for that specific protein molecule.
Your Smarty-Pants Reply: "So, if you boil an egg, you're denaturing the proteins, but you're not breaking them into individual amino acids. That's why you can't un-boil an egg! Fascinatingly irreversible, usually." You're basically a culinary scientist now, too!
The Joy of Understanding
Look, I know these worksheets can sometimes feel like deciphering an ancient scroll. But every time you correctly identify an alpha-helix, predict a hydrophobic interaction, or explain denaturation, you’re unlocking a little piece of the incredible complexity of life. Proteins are the workhorses of our cells, and understanding how they fold is like understanding how the world around you works on a microscopic level.
So, the next time you’re staring at a worksheet, take a deep breath. Remember the LEGO analogy, the pool party, the tiny hugs. You’ve got this! Each question you solve is a step closer to truly appreciating these amazing molecular machines. And who knows, maybe this knowledge will even help you fold your next fitted sheet a little more successfully. 😉 Keep exploring, keep learning, and keep that curious mind engaged. You’re doing great!
