Propose A Mechanism For The Following Transformation.

Hey there, curious minds! Ever looked at a recipe and thought, "How on earth does this happen?" You know, like how simple flour and water somehow magically become a fluffy loaf of bread? Or how that splash of vinegar can turn milk into something chunky and amazing for cheesecake? Well, today we're going to peek behind the curtain of a similar kind of magic, but instead of the kitchen, we're diving into the fascinating world of chemistry. Don't worry, no lab coats or complicated jargon required! Think of this as a friendly chat about how one thing can transform into another, and why that's actually super cool.
So, let's imagine we've got this particular chemical transformation. We start with something (let’s call it “Thing A”) and with a little help, it becomes something else entirely (we’ll call it “Thing B”). Our mission, should we choose to accept it (and trust me, it's a fun mission!), is to figure out the step-by-step story of how Thing A becomes Thing B. This step-by-step story is what we chemists call a "mechanism". It’s like the director’s cut of a chemical reaction, showing you all the little scenes and behind-the-scenes action that lead to the final blockbuster result.
Why should you, an everyday awesome human, care about a chemical mechanism? Good question! Think about it. When you’re trying to fix something around the house, understanding how it broke helps you fix it better, right? If your favorite gadget stops working, you don't just randomly poke at it; you try to understand the faulty part. Similarly, understanding a chemical mechanism helps scientists design new medicines, create better materials for your phone or your clothes, and even find ways to clean up pollution more effectively. It’s all about understanding the "why" and "how" to make things work for us!
Let’s make this super relatable. Imagine you’re trying to get from your cozy couch to the yummy-smelling kitchen for a snack. You don't just teleport there, right? You have a series of actions: you get up, you walk, maybe you open a door. Each of those is a step in your personal journey. A chemical mechanism is just like that – a sequence of smaller, more manageable steps that a molecule takes to get from its starting form to its final form.
Now, the specific transformation we're looking at involves a few key players. We've got our starting molecule, our Thing A. Let's give it a bit more character. Imagine Thing A is like a shy introvert, perfectly happy in its own little space. But then, something happens – something or someone else enters the scene and encourages Thing A to step out of its shell.

In our case, this "something else" is often another molecule, or even just a part of the molecule itself that gets a little bit antsy. Think of it like this: sometimes, a part of a molecule might have a slight negative charge, like a tiny little magnet that attracts positive things. And on the flip side, another part might have a slight positive charge, like a magnet that’s always looking for something negative to cling to. It's a dance of attraction and repulsion, constantly influencing how molecules interact.
So, the first step in our mechanism might be this shy introvert (Thing A) bumping into a slightly more adventurous molecule, let’s call it our "Catalyst Friend." This Catalyst Friend isn't really changed by the interaction, but it has a way of making Thing A a bit more… ready for change. It’s like that friend who nudges you to try something new. The Catalyst Friend might, for instance, gently pull on a certain part of Thing A, making it a little more eager to break a bond or accept a new partner.
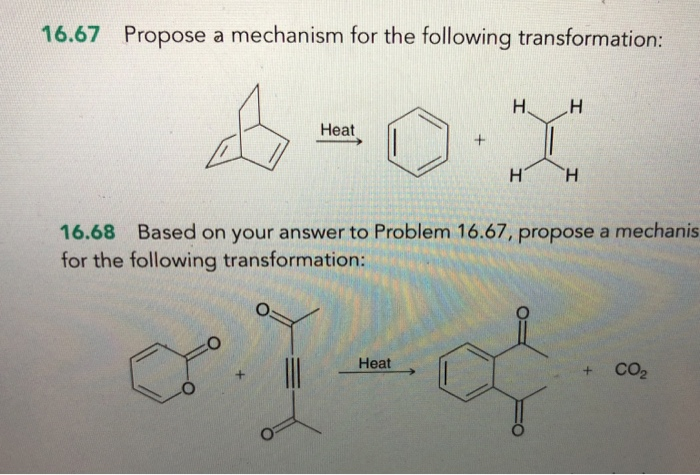
This "nudging" is often the first actual chemical event. Imagine a really strong rubber band. It’s holding two things together. Our Catalyst Friend, or an internal "push" within Thing A, might start to stretch that rubber band. It doesn’t snap immediately, but it’s definitely getting thinner and weaker.
This stretching leads us to the next exciting step. Once that rubber band is sufficiently stretched, it might break completely. This is a big moment! Now, Thing A isn't Thing A anymore. It's become something new, let's call it an "Intermediate." Think of an Intermediate like a teenager – no longer a child, but not quite an adult yet. They’re in a transitional phase, with new properties and behaviors.
These Intermediates are often quite reactive. They’re like that teenager who’s full of energy and maybe a little unpredictable. They have unfinished business, and they’re looking to connect with something or someone to achieve a more stable state. In our transformation, this Intermediate will then interact with yet another molecule, or perhaps an electron, or even a proton (which is just a tiny positively charged particle – think of it as a tiny, eager helper).
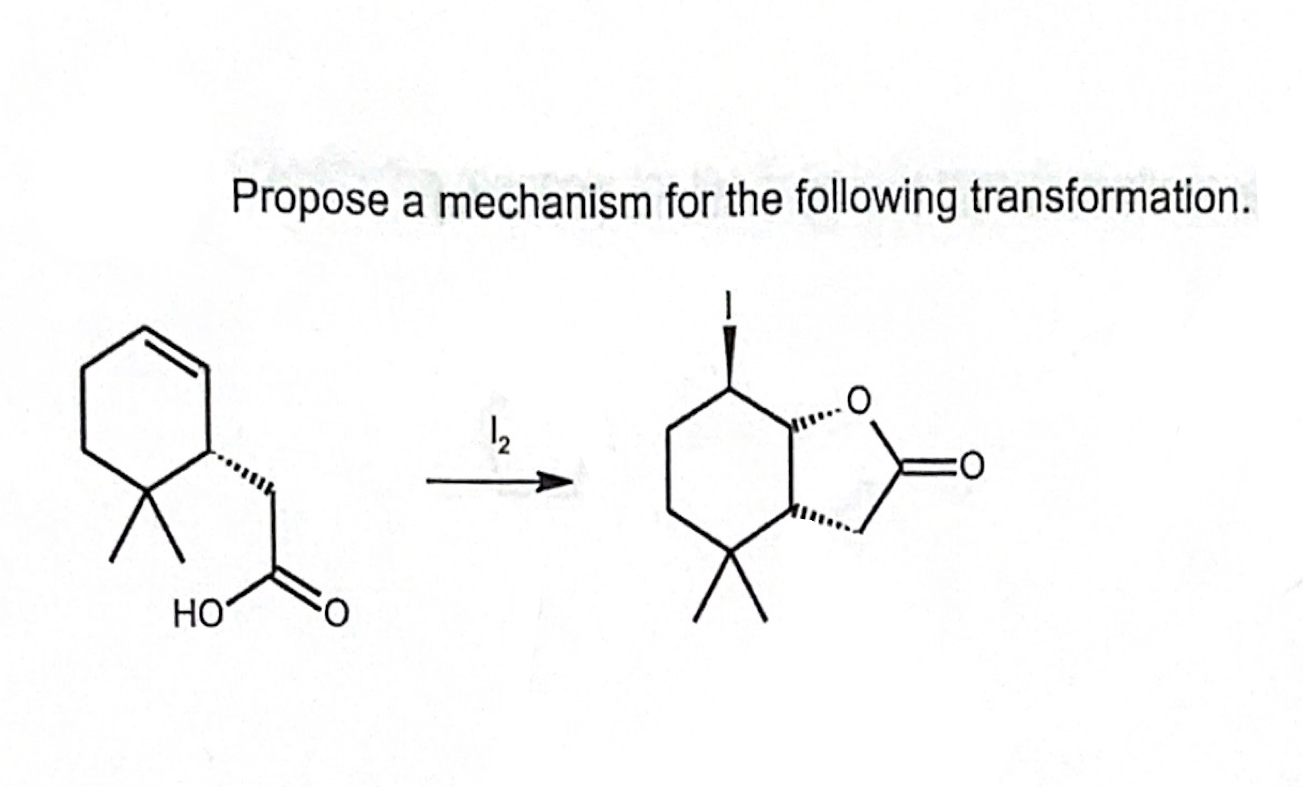
Let’s say our Intermediate needs to grab something. Imagine it's like someone who’s dropped their keys and is looking around frantically. It sees another molecule, our "Helper Molecule," and thinks, "Aha! You have the spare key I need!" It then takes something from the Helper Molecule. This could be an electron, or an atom, or a group of atoms. This step is crucial because it’s what leads us closer to our final product, Thing B.
Now, the Helper Molecule, having given up its "spare key," might be a bit unhappy. It might be left with a positive charge, or an incomplete bond. This is where the cycle of chemical reactions often comes into play. Remember our Catalyst Friend from the beginning? It might reappear here, not to be changed itself, but to help the Helper Molecule get back to its original, stable form. It's like a supportive friend who helps someone after they’ve helped someone else. The Catalyst Friend might offer a little nudge to the Helper Molecule, allowing it to regain its original structure, ready to help out again in another round of transformations.
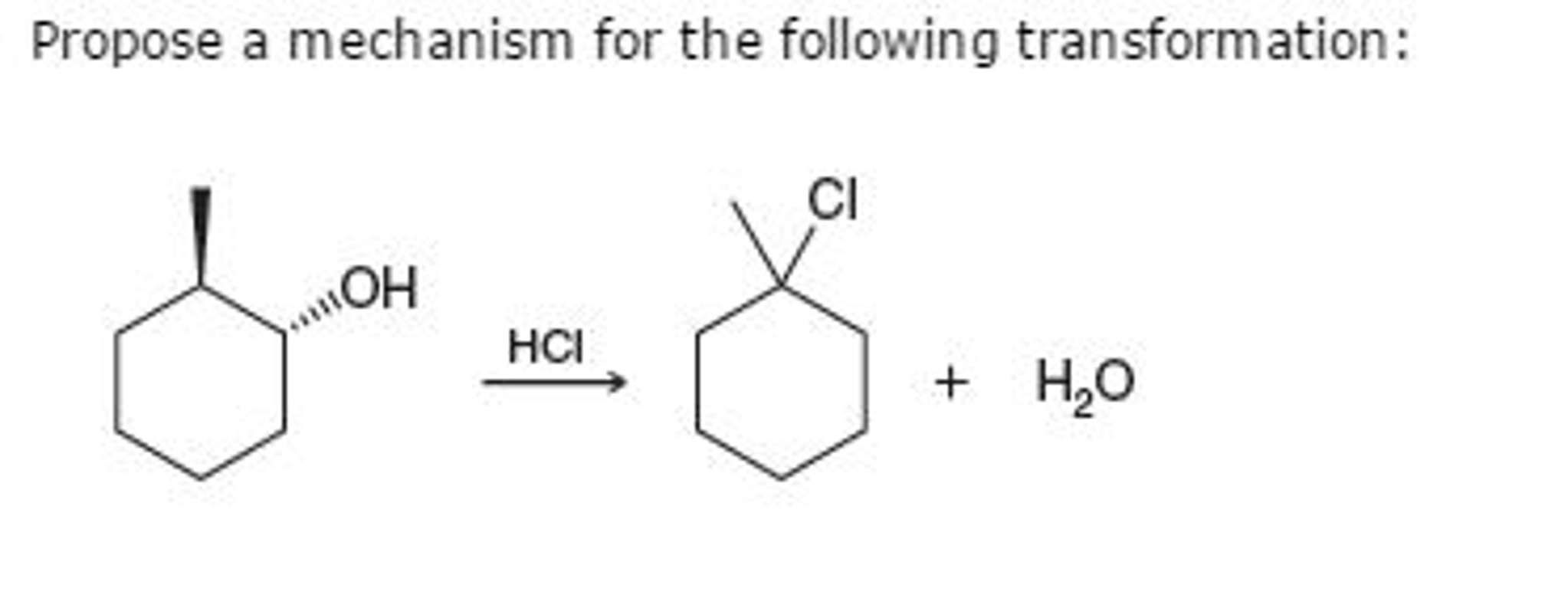
And what about our Intermediate? After taking what it needed from the Helper Molecule, it’s now transformed into our glorious Thing B – the final product! It’s like the teenager who, after all the growing and experimenting, has finally found their footing and become a stable, happy adult. Thing B is the desired outcome, the fluffy bread, the creamy cheesecake.
So, to recap, our mechanism looks something like this:
- Initiation: Our shy introvert (Thing A) encounters the friendly nudger (Catalyst Friend or an internal "push").
- Bond Breaking: A connection within Thing A gets stretched and eventually breaks, forming a reactive Intermediate.
- Intermediate Reactivity: The Intermediate, like a teenager, seeks out and takes something from a Helper Molecule.
- Product Formation & Regeneration: This act of taking leads to our final product (Thing B) and often regenerates the Catalyst Friend, ready for the next cycle.
It’s a beautiful, intricate dance of molecules, each step carefully choreographed. And by understanding this dance, we can not only appreciate the complexity of the world around us but also learn to guide these transformations to our advantage. It’s about unlocking the secrets of matter, one tiny, elegant step at a time. Pretty neat, huh?
