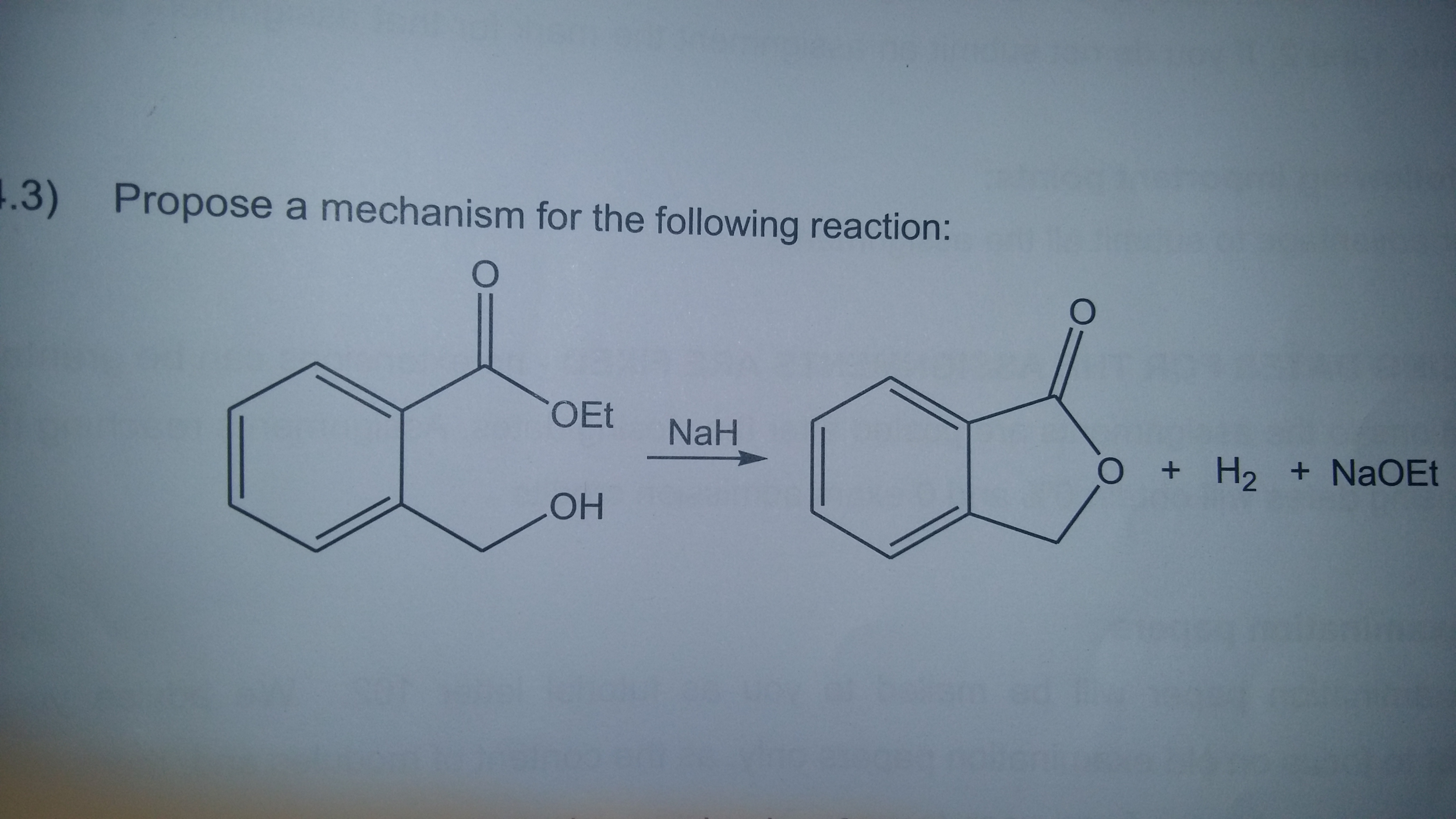
Propose A Mechanism For The Following Reaction

You know, I was staring at this ridiculously complex chemical reaction the other day, feeling utterly lost. It looked like a chemical spaghetti monster had exploded all over the page. And then, I remembered this time in college, trying to assemble IKEA furniture without the instructions. Utter chaos. Screws everywhere, weird wooden dowels, and me, muttering obscenities at a half-built bookshelf. Eventually, after much trial and error (and a minor existential crisis), I figured it out. It was all about understanding how each piece connected to the next, how one step logically led to another. That, my friends, is what we’re doing today with chemistry. We’re ditching the instruction manual and figuring out the IKEA of reactions, one step at a time.
So, the reaction I’ve got simmering in my brain is this beauty:
Reactant A + Reactant B → Product C
Simple enough on paper, right? But how do we get from the starting materials to that glorious final product? It’s not magic. It's a series of perfectly orchestrated dance moves, each with its own tiny reason for existing. We’re going to try and suss out that choreography. Think of it like being a detective. We have the suspects (the atoms and molecules), the crime scene (the reaction vessel), and the evidence (the bonds breaking and forming). Our job is to figure out who did what to whom, and in what order.
Now, this particular reaction, let’s call it the "Enigmatic Enolization," is a bit of a classic. It involves a carbonyl compound, a nucleophile, and some interesting rearrangements. You might have seen something similar in organic chemistry textbooks, looking all neat and tidy with perfectly drawn arrows. But behind those elegant arrows lies a whole lot of molecular drama.
Let's break down our initial players. On one side, we have our carbonyl compound. Imagine a molecule with a double bond between a carbon and an oxygen atom. This C=O group is quite special. The oxygen is more electronegative than carbon, meaning it pulls the shared electrons closer to itself. This creates a partial negative charge on the oxygen ($\delta^-$) and a partial positive charge on the carbon ($\delta^+$). Think of it like a tiny magnet, with one end a bit more attractive to electrons than the other. This makes the carbon atom a prime target for attack by electron-rich species.
And who are these electron-rich species? Well, that’s where our nucleophile comes in. "Nucleophile" literally means "nucleus-loving." Remember, the nucleus of an atom is positively charged. So, anything that’s negatively charged, or has a region of high electron density, is going to be attracted to positive things. It’s like moths to a flame, or me to a good cup of coffee on a Monday morning. They're looking for that electron-deficient carbon.
So, the first logical step in many reactions involving carbonyls and nucleophiles is, unsurprisingly, an attack. Our nucleophile, with its abundance of electrons, sees that slightly positive carbon on the carbonyl group and thinks, "Hey, let's get acquainted!" It swoops in and forms a new bond with that carbon. This is the first big move in our molecular dance.
What happens when this attack occurs? Well, the electrons in the C=O double bond, which are only shared between the carbon and oxygen, have to go somewhere. They can't just stay put if the carbon is busy forming a new bond with our nucleophile. So, one of those electron pairs from the double bond gets pushed onto the oxygen atom. This makes the oxygen atom carry a full negative charge. It's like the electrons in the double bond were a shared resource, and when one side of the partnership got a new suitor, the other side (the oxygen) had to take on more responsibility, becoming negatively charged.
We now have what's called a tetrahedral intermediate. It’s a fancy name for a molecule where the carbon atom, which was originally sp2 hybridized (flat, with a double bond), becomes sp3 hybridized (tetrahedral, with four single bonds). This intermediate is usually quite reactive because it has both a negatively charged oxygen and, depending on the nucleophile, perhaps other charged or polar features. It's a bit of a chemical hot potato.
Now, this tetrahedral intermediate might hang around for a bit, or it might be eager to rearrange. In our "Enigmatic Enolization," the next step often involves a proton transfer. What's a proton in chemistry? It's essentially a hydrogen ion (H$^+$). These little guys are often floating around in the reaction mixture, perhaps from the solvent or from some acidic/basic conditions. If there’s a way for a proton to stabilize the situation, it often takes it.
In our specific case, that negatively charged oxygen on the tetrahedral intermediate is a strong base. It's like a thirsty sponge, ready to soak up any available protons. So, if there’s an acidic proton nearby – often on an adjacent carbon atom that’s been deprotonated or is adjacent to the carbonyl group – the oxygen will grab it. This reprotonation event neutralizes the negative charge on the oxygen and puts a hydrogen atom back on it, forming a hydroxyl group (-OH).
But wait, there’s a twist! In the "Enigmatic Enolization," we're not just aiming for a simple alcohol. We're aiming for an enol. An enol is a molecule that has a double bond adjacent to a hydroxyl group. Think of it as a vinyl alcohol. The standard way to get to an enol from our tetrahedral intermediate involves a slightly different proton transfer. Instead of the oxygen grabbing a proton, a different base in the solution (or even another part of the same molecule) might deprotonate a carbon atom next to the original carbonyl carbon. This creates a carbanion – a carbon with a negative charge.
This carbanion is a pretty significant species. It’s electron-rich and wants to get rid of that negative charge. How can it do that? By reforming a double bond! The electrons from the newly formed carbanion can push back towards the oxygen atom. This pushes the electrons from the original C=O double bond (which are now single bonds to the oxygen after the initial nucleophilic attack) back to reform a double bond between the carbon and oxygen. But that's not quite right for an enol. Let's re-evaluate.
Okay, let's backtrack a moment. We have our tetrahedral intermediate with a negatively charged oxygen and a new bond formed by the nucleophile. Now, in the context of forming an enol, the goal is to create a C=C double bond next to an -OH group. This means we need to eliminate something or rearrange. The most common way this happens is through a process that involves the reformation of the carbonyl, followed by deprotonation to form the double bond.
So, let's refine our intermediate step. After the nucleophile attacks the carbonyl carbon, we have that negatively charged oxygen. Now, imagine there was a proton attached to the carbon adjacent to the carbonyl carbon. In many reactions, a base will abstract (remove) this proton. This leaves a negative charge on that adjacent carbon, forming a carbanion. At the same time, the electrons from the C-H bond that was just broken can flow back to form a double bond between that carbon and the carbonyl carbon. Simultaneously, the electrons that were holding the oxygen in its negatively charged state can reform the C=O double bond. This seems a bit crowded, doesn't it?
Ah, but here's the elegant part, and where the "Enigmatic Enolization" gets its name! Often, the tetrahedral intermediate doesn't immediately lose the nucleophile. Instead, a proton is transferred from the alpha-carbon (the carbon next to the carbonyl) to the negatively charged oxygen. This happens internally or with the help of the solvent/base. So, you have a hydroxyl group forming on the original carbonyl carbon, and a negative charge on the alpha-carbon. This is still a tetrahedral intermediate, but now with a protonated oxygen and a deprotonated alpha-carbon.
Now, this negatively charged alpha-carbon is the key! It's got a spare electron pair. The negatively charged oxygen is still holding onto its negative charge. What if the electrons on the alpha-carbon could push back towards the bond connecting it to the original carbonyl carbon? And at the same time, the electrons that were holding the oxygen to that carbon could reform a double bond? This is where the magic happens. The electrons from the alpha-carbon carbanion push inwards, forming a C=C double bond. And as this double bond forms, the electrons from the original C-O single bond (now that the oxygen has a proton) are expelled. But where do they go? They don't get expelled as a separate entity. Instead, they move to reform the double bond with the oxygen, kicking out a leaving group if there was one, or rearranging the bonds to form the final product.
Let's try a more concrete example of the "Enigmatic Enolization" mechanism. Imagine we have an aldehyde or a ketone reacting with a base. The base deprotonates the alpha-carbon, forming a carbanion. This carbanion is resonance-stabilized, meaning the negative charge can be delocalized over the adjacent oxygen atom via a structure called an enolate. This enolate ion is crucial. It's got that negative charge that can act as a nucleophile itself!
So, if our "Reactant A" is a ketone or aldehyde and "Reactant B" is a base, the base will attack an alpha-hydrogen. This removes the hydrogen and leaves a negative charge on the alpha-carbon, forming a carbanion. This carbanion can then resonate with the carbonyl oxygen, meaning the negative charge is shared between the carbon and the oxygen. This is the enolate ion. It's like having two potential sites of reactivity, both negatively charged.
Now, what happens next depends on the specific reaction. In many cases, this enolate ion is the key intermediate. It’s electron-rich and can now act as a nucleophile. If there’s an electrophile (an electron-poor species) present, the enolate can attack it. This is a very common way to form new carbon-carbon bonds. Think of the aldol reaction, for example, where one enolate attacks another carbonyl compound.
However, in the specific "Enigmatic Enolization" we are considering, the goal is to form the enol itself. So, after the carbanion is formed and resonates to the enolate, the key step is protonation. But it's not just any protonation. If the reaction is run under slightly acidic or neutral conditions, the enolate can pick up a proton from somewhere in the solution. The most likely place for this proton to land is on the oxygen atom of the enolate. This neutralizes the charge on the oxygen and creates a hydroxyl group, while the double bond reforms between the alpha-carbon and the original carbonyl carbon. Voilà! We have an enol.
So, to summarize the proposed mechanism for our "Enigmatic Enolization":
Step 1: Formation of the Carbanion/Enolate. A base abstracts an alpha-hydrogen from the carbonyl compound. This forms a carbanion which is in equilibrium with its resonance structure, the enolate ion. The enolate ion has a negatively charged oxygen and a double bond adjacent to the carbon that was deprotonated. This is the initialization of our enol formation.
Step 2: Protonation of the Enolate. The enolate ion, with its negatively charged oxygen, acts as a nucleophile and abstracts a proton (H$^+$) from a proton source in the reaction mixture. This proton attaches to the oxygen, neutralizing its negative charge and forming a hydroxyl group (-OH). Simultaneously, the electrons from the alpha-carbon carbanion part of the resonance structure push back to form a carbon-carbon double bond.
Let's visualize this with arrows. The base attacking the alpha-hydrogen creates an arrow from the C-H bond to the alpha-carbon, making it negatively charged. Then, electrons from that carbanion can move to form a double bond between the alpha-carbon and the carbonyl carbon, pushing electrons from the C=O double bond onto the oxygen, creating the negatively charged oxygen. This is the resonance structure. Then, an arrow from the negatively charged oxygen attacks an incoming proton (H$^+$). As the oxygen bonds to the hydrogen, the double bond reforms between the alpha-carbon and the original carbonyl carbon. This is the enol form.
It's important to note that enols are often in equilibrium with their keto forms (the original carbonyl compounds). The keto form is usually more stable. However, under certain conditions, the enol can be the dominant species or a crucial reactive intermediate for subsequent reactions. The "Enigmatic Enolization" describes the pathway to get to this enol form.
So, the entire process is not a single, massive event, but a carefully timed sequence of electron movements. It's like a well-rehearsed play, where each actor (atom or electron) knows its cue and its movement. We go from a stable carbonyl compound, to a reactive intermediate (the enolate), and finally to the enol. Understanding these steps, these electron pushes and pulls, is what allows us to predict what will happen, to design new reactions, and to truly understand the language of chemistry.
And that, my friends, is how we tackle these seemingly overwhelming reactions. We break them down, step by step, looking at where the electrons want to go, and who's feeling a bit electron-hungry or electron-rich. It's a lot like assembling that IKEA bookshelf, really. Once you see how the pieces fit, it all makes sense. Now, if you'll excuse me, I think I have a few more chemical spaghetti monsters to untangle!
