Propanol And Isopropanol Are Isomers This Means That They Have

Ever stopped to wonder why some cleaning products smell so powerfully sterile, while others have a gentler, almost sweet scent? Or maybe you've noticed that different types of rubbing alcohol seem to tackle different jobs? The secret behind these everyday observations often boils down to a fascinating concept in chemistry: isomerism. And today, we're diving into a classic example that’s both incredibly common and surprisingly simple to grasp: the relationship between propanol and isopropanol. These two chemicals might sound a bit intimidating, but they're actually superstars in our daily lives, and understanding their connection is like unlocking a little cheat code for understanding the world around you.
So, what’s so fun about isomers? Think of them like identical twins who look alike but have distinct personalities and skills. They're made of the exact same "building blocks" – the same number of carbon, hydrogen, and oxygen atoms – but these atoms are arranged in a slightly different way. This subtle difference in structure leads to vastly different properties and uses, making them incredibly useful in a wide array of applications. It's a perfect illustration of how small changes can have big impacts, a principle that resonates far beyond the lab and into so many aspects of our lives.
The Power of Structure: Propanol vs. Isopropanol
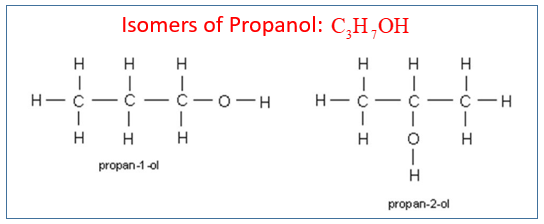
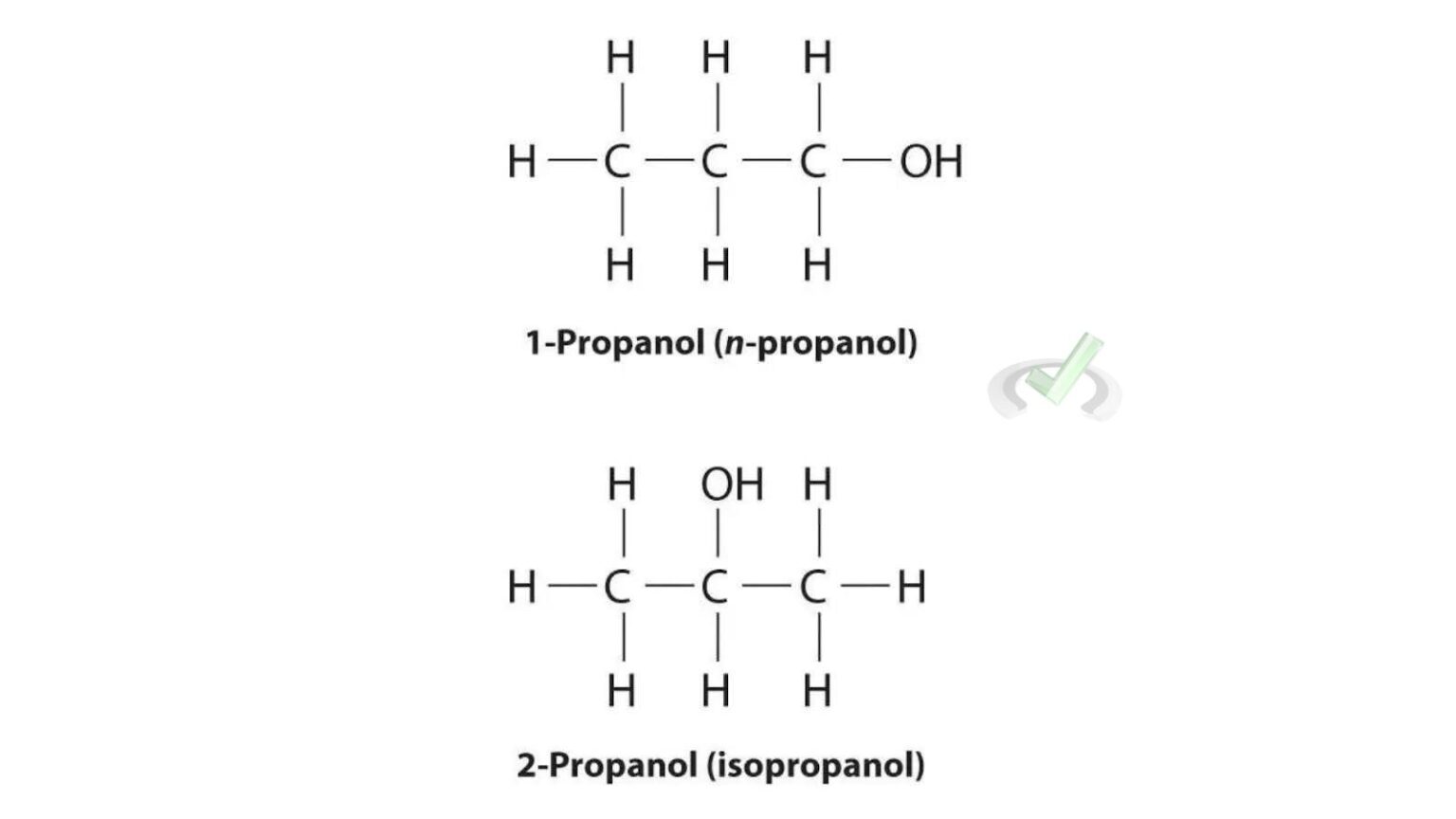
Let’s get down to the nitty-gritty. Both propanol and isopropanol share the same chemical formula: C₃H₈O. This means they both have three carbon atoms, eight hydrogen atoms, and one oxygen atom. They are, in essence, built from the same Lego bricks. However, the way these bricks are connected is where the magic, and the difference, lies.
Imagine you have three carbon atoms lined up in a row. Now, you need to attach the remaining hydrogen and oxygen atoms.
In propanol, also known as n-propanol or 1-propanol, the atoms are arranged in a straight, linear fashion. The oxygen atom is attached to the first carbon atom in the chain. Think of it as a straightforward, uncluttered arrangement. This linear structure gives n-propanol certain properties that make it suitable for specific industrial tasks.
Now, picture that same chain of three carbon atoms. Instead of the oxygen attaching to the end carbon, what if it attaches to the middle carbon? That's where isopropanol comes in!

Isopropanol, also called isopropyl alcohol or 2-propanol, has its oxygen atom attached to the second carbon atom in the chain. This seemingly minor shift in attachment point creates a branched structure. It's like taking that linear Lego construction and giving it a slight bend or a side branch. This "branched" nature is crucial and dictates how isopropanol behaves.
This difference in arrangement might seem tiny, but it’s a prime example of isomerism. They are structural isomers because the atoms are connected in a different structural order, even though the total number of atoms is the same.
Purpose and Benefits: Why Do We Care?
Why is it useful to know that these two molecules are isomers? Because their different structures lead to different physical and chemical properties, making them suited for entirely different, yet equally important, purposes.
N-propanol (the straight-chain one) is often found in applications where a less volatile solvent is needed. You might find it used as a solvent in the production of printing inks, as a cleaning agent for electronic components, or as an ingredient in varnishes and lacquers. Its slightly different boiling point and evaporation rate compared to isopropanol make it ideal for these specific industrial processes.
Isopropanol (the branched one), on the other hand, is the superstar of our medicine cabinets and cleaning supplies. This is the rubbing alcohol you’re most likely familiar with.
Its primary benefit as isopropyl alcohol is its potent antiseptic and disinfectant properties. When you use it to clean a wound, it effectively kills bacteria and other germs. It evaporates quickly, leaving surfaces clean and germ-free.
Beyond its medical uses, isopropanol is also a fantastic degreaser and solvent for many common substances. It's used in:
- Cleaning electronics: Its ability to dissolve oils and grime without leaving residue makes it perfect for cleaning circuit boards and other sensitive electronic equipment.
- Window cleaners: It helps cut through grease and grime, leaving windows streak-free.
- Hand sanitizers: A key ingredient in many hand sanitizers, providing a quick and effective way to kill germs on the go.
- Antifreeze: In some formulations, it can be used as an antifreeze due to its ability to lower the freezing point of water.
So, the next time you reach for a bottle of rubbing alcohol to clean a cut, or you see a cleaning product with a distinct, sharp scent, remember the fascinating world of isomerism. It's a testament to how the precise arrangement of atoms, even in the simplest molecules like propanol and isopropanol, can create entirely different substances with unique and valuable roles in our everyday lives. It’s a small piece of chemistry that makes a big difference!
