Product Of 3 4-dichloro-1-nitrobenzene And Sodium Methoxide
Ever wondered what goes on behind the scenes to create the vibrant colors in your clothes, the effective medicines that keep you healthy, or even the sturdy plastics in everyday objects? It’s often a fascinating dance of chemical reactions, and today, we’re peeking into one such captivating transformation. Forget dusty textbooks; think of this as a glimpse into the ingredient list of innovation! The journey we're about to explore involves a rather specific pairing of chemicals that, when brought together, unlock a world of possibilities for creating entirely new and useful substances. It’s like having a secret recipe that allows chemists to build complex molecules step-by-step, much like an architect designs a building. The beauty lies in its precision and the diverse applications it enables. So, buckle up, and let’s dive into the exciting realm of how a particular chemical reaction fuels progress in so many areas of our lives.
The Magic Behind the Molecule: Unlocking New Possibilities
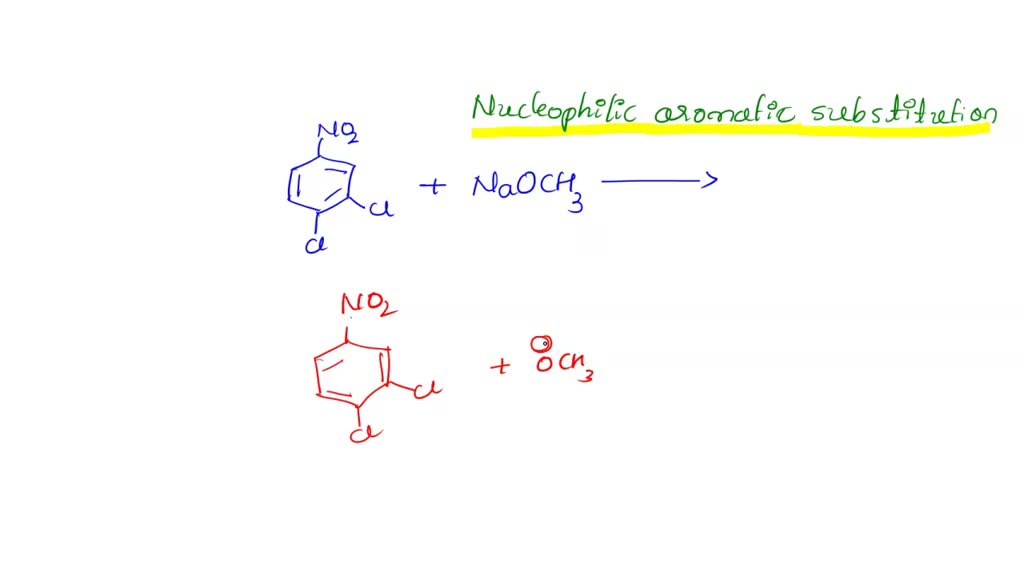
At its heart, the process we’re discussing is about skillfully transforming one chemical compound into another, often with significant upgrades in functionality. The star players in this particular act are 3,4-dichloro-1-nitrobenzene and sodium methoxide. Now, these names might sound a bit technical, but imagine them as specialized building blocks. 3,4-dichloro-1-nitrobenzene is a molecule with a specific arrangement of atoms, featuring chlorine and nitro groups attached to a benzene ring. These attachments make it ripe for further chemical modification. Think of it as a raw material with pre-existing features that make it ideal for certain types of construction.
Enter sodium methoxide. This is a powerful chemical reagent, essentially a source of methoxide ions. In simple terms, it’s like a very specific hammer or screwdriver in our molecular toolbox. When sodium methoxide interacts with 3,4-dichloro-1-nitrobenzene, a fascinating substitution reaction occurs. One of the chlorine atoms on the benzene ring is replaced by a methoxy group (derived from the sodium methoxide). This might sound like a small change, but it's a crucial step that opens up a cascade of new chemical properties and potential uses for the resulting molecule. This new compound, which we can affectionately call the “product of 3,4-dichloro-1-nitrobenzene and sodium methoxide,” is now significantly different from its starting material and ready for its own set of adventures.
This reaction isn't just a random mixing of ingredients; it's a carefully orchestrated transformation that yields valuable intermediates for a wide array of industries.
The purpose of this reaction is primarily to create a more versatile intermediate. The initial 3,4-dichloro-1-nitrobenzene has its uses, but the introduction of the methoxy group can dramatically alter its reactivity. This makes the new compound a much more amenable starting point for synthesizing a broad spectrum of other, more complex organic molecules. These can include pharmaceuticals, agrochemicals (like pesticides and herbicides), dyes, and advanced materials. The ability to precisely control these chemical transformations is fundamental to modern chemical synthesis.
Benefits that Ripple Through Our World
So, what are the real-world benefits of this seemingly specialized reaction? The advantages are far-reaching and touch many aspects of our daily lives, often in ways we don’t even realize.
One of the most significant benefits is its role in the pharmaceutical industry. The product of this reaction can serve as a key building block for synthesizing active pharmaceutical ingredients (APIs). Many life-saving drugs, from pain relievers to treatments for chronic diseases, rely on complex organic molecules that are built piece by piece. This reaction provides a reliable way to introduce specific functional groups that are essential for a drug’s efficacy and how it interacts with the body. The precision offered by such reactions means that chemists can design and produce molecules with targeted therapeutic effects, minimizing unwanted side effects.

Secondly, the agrochemical sector heavily relies on such precise chemical synthesis. Imagine the need for effective and targeted ways to protect crops from pests and diseases, or to manage weeds that compete for vital resources. The compounds derived from the reaction of 3,4-dichloro-1-nitrobenzene and sodium methoxide can be transformed into potent herbicides, insecticides, and fungicides. These agrochemicals play a crucial role in ensuring food security by increasing crop yields and quality. The development of more environmentally friendly and specific agrochemicals is a constant goal, and reactions like this are vital to achieving it.
Beyond health and agriculture, the dye and pigment industry also benefits immensely. The unique electronic properties of aromatic compounds, often modified through reactions like this, are what give them their color. The resulting molecules can be further processed to create vibrant and durable dyes used in textiles, paints, and inks. The ability to fine-tune the molecular structure allows for a vast spectrum of colors and performance characteristics, from fade resistance to specific shades.

Furthermore, this reaction contributes to the development of specialty chemicals and advanced materials. These can include polymers with specific properties for high-performance applications, or intermediates for electronic materials. The modular nature of organic synthesis, where each reaction builds upon the last, is what allows us to engineer materials with incredible strength, flexibility, or conductivity. The product of 3,4-dichloro-1-nitrobenzene and sodium methoxide is a versatile platform for creating such innovations.
In essence, this chemical transformation is a quiet enabler of modern life. It’s a testament to the power of chemistry to provide the foundational components for countless products that improve our health, sustain our food supply, and add color and functionality to our world. The ability to control and manipulate molecules at this level is what drives innovation and allows us to address complex challenges across a multitude of industries.
