Process That May Create Alpha Particles Very Briefly Crossword

Hey there, word nerds and science fans! Ever think about tiny, invisible things that make up everything? Yep, we're diving into the wonderfully weird world of alpha particles. Don't worry, this won't be a boring lecture. Think more like a fun chat over coffee about something seriously cool.
So, what's the deal with these alpha particles? Imagine them as little cosmic bullets. Super small. Like, impossibly small. They're actually a part of an atom's nucleus. Yep, the very heart of an atom! And sometimes, atoms get a little… unstable. They're like a toddler who's had too much sugar. They need to release some energy.
And how do they release that energy? BAM! They might shoot out an alpha particle. It's basically a helium nucleus. Crazy, right? Two protons and two neutrons chilling together. Think of it as a tiny, speedy package deal from an atom that's had enough.
Now, why is this fun to talk about? Because it's all about transformation and decay. Things changing! It’s like a magic trick happening all around us, all the time. Except, you know, science.
We're talking about radioactive decay here. That's the fancy term for this atomic shedding. Some elements are just built that way. They’re a bit… dramatic. They can’t hold it all in, so they let loose these alpha particles. It’s their way of saying, "Peace out, I'm changing!"
Think about elements like uranium or radium. They’re the rockstars of radioactive decay. They’ve got that certain je ne sais quoi that makes them emit these alpha particles. It's their signature move. Their claim to fame.
And the coolest part? This process, this release of alpha particles, it's been happening for billions of years. We're talking ancient history, cosmic origins, the whole shebang. These little bullets have been zipping around since the universe was just a baby.

So, how do we get these alpha particles? It's not like you can just make them appear with a flick of your wrist. They are born from the nucleus of certain heavy atoms. When these atoms decide they've had enough of their current configuration, they go through a process called alpha decay.
It’s kind of like an atom having a family reunion and deciding to split up the furniture. The nucleus says, "Okay, this arrangement isn't working anymore. Let's send out these two protons and two neutrons as a package. See ya later!"
And this "package" is the alpha particle. It's surprisingly hefty compared to other particles, like beta particles or gamma rays. Imagine it as the slightly heavier cousin who shows up to the party with a whole picnic basket. It's got some substance!
This is where things get really interesting, and I promise we’re keeping it light. Because these alpha particles, while small, have a bit of a punch. They’re positively charged, thanks to those protons. That means they interact with stuff around them. They don't just zoom through like ghosts.
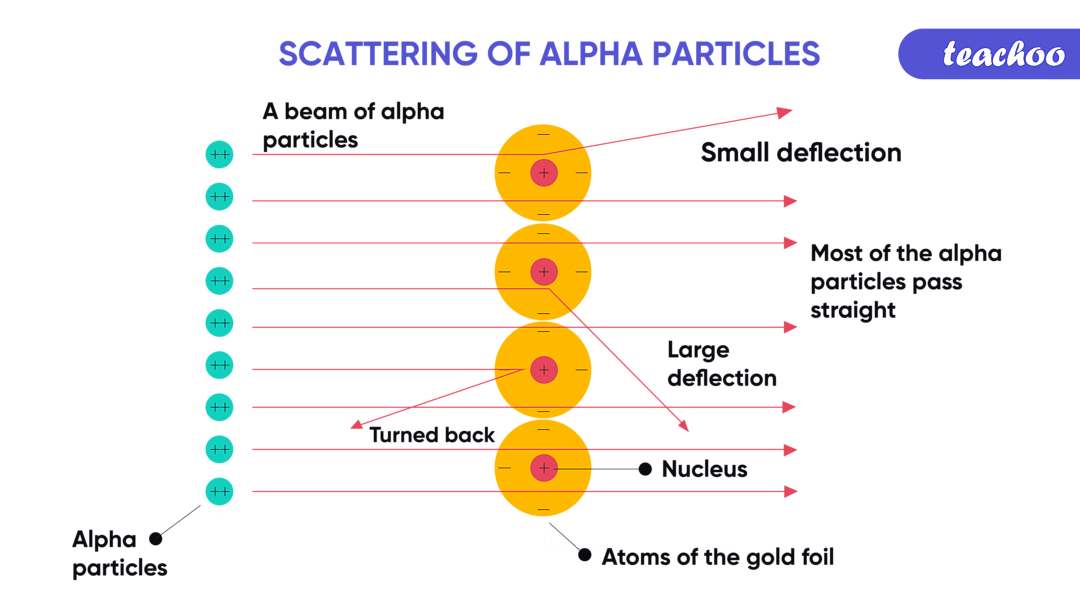
They're actually quite good at bumping into things. Like a tiny, energetic bowling ball. They can knock electrons off other atoms. This is called ionization. It's a fancy word for making atoms a little bit grumpy by messing with their electrons. Think of it as the alpha particle giving other atoms a little jolt.
But here’s a quirky fact: while they're energetic, alpha particles don't travel very far. They get stopped pretty easily. A piece of paper? Game over for an alpha particle. Your skin? Nope, it’s a tough barrier. This is actually a good thing! It means they’re not super dangerous if they're outside your body. It's all about where they are and what they're doing.
The real "uh-oh" moment is if you inhale or ingest something that's emitting alpha particles. Then those little bullets are right there, inside you, doing their ionization thing. That’s where things can get a bit more serious. But again, this is just for fun, so let's focus on the cool science!
The process of creating alpha particles is fundamental to understanding nuclear physics. It's one of the primary ways heavy, unstable nuclei become more stable. They're trying to find their happy place, their most comfortable atomic arrangement. And alpha decay is how some of them get there.

Think about it like a puzzle. The nucleus is trying to find the best fit. If it's got too many pieces (protons and neutrons), it might decide to spit out a perfect little group of four (two protons, two neutrons) to make the rest fit better. Voilà! Alpha particle created.
And this whole thing, this spontaneous spitting out of alpha particles, it’s called spontaneous fission for some really heavy elements, but alpha decay is the more common and widely recognized process for many radioactive isotopes.
The "very briefly" part of our imaginary crossword clue is key. Alpha decay happens very quickly once the conditions are right within the nucleus. It's not a slow, drawn-out process for the atom itself. It's a sudden emission. Like a sneeze. A very, very tiny, atomic sneeze.
So, to recap this little bit of atomic fun: Alpha particles are basically tiny helium nuclei. They're shot out by unstable, heavy atoms. This happens through alpha decay. It's a form of radioactive decay. These particles ionize things. They’re stopped easily by physical barriers.

Why is this fun? Because it's a glimpse into the hidden workings of the universe. It’s about the constant dance of change and stability happening at the most fundamental level. It’s the universe playing its own, incredibly subtle, game of Jenga.
And if you ever see a crossword clue like "Process that may create alpha particles very briefly," you'll know the answer is likely something along the lines of ALPHA DECAY. See? Science can be a puzzle! A very, very small puzzle.
It’s just fascinating to think that these energetic little packets are constantly being released from elements all around us, from the earth beneath our feet to the cosmic dust in space. They're the universe's tiny, energetic messengers, quietly transforming and shaping the world in ways we can't even see.
So, next time you're looking at a crossword puzzle, or just contemplating the vastness of the universe, remember the humble alpha particle. It's a tiny powerhouse, a byproduct of atomic drama, and a key player in the ongoing story of matter. Pretty neat, huh?
