Process That May Create Alpha Particles Very Briefly

So, you know those tiny, almost invisible things that make up everything? Atoms! They’re usually pretty chill, just doing their atom thing. But sometimes, for reasons only they truly understand, some atoms get a little… energetic.
It's like when you've had too much coffee and start vibrating at a higher frequency. These atoms decide they need to shed some of that extra oomph. And how do they do it? Well, that’s where the fun, or at least the mildly interesting, begins.
Imagine an atom as a tiny solar system. You've got the big, heavy nucleus in the middle. That's like the sun. And then you have electrons whizzing around it like planets. It's a pretty neat setup, most of the time.
But some of these nuclei are just a bit too plump. They’ve got too many protons and neutrons hanging out. It’s like a balloon that’s been blown up a little too much. You can see it’s starting to strain.
This plump nucleus feels a bit… unstable. It’s like trying to balance a bowling ball on a toothpick. It just doesn’t feel right, you know? It craves a bit more equilibrium.
So, what does this overloaded nucleus do? It decides to kick something out. It’s a bit like decluttering. Gotta make space! And what does it decide to toss? Not just any old bit of fluff.
It decides to eject a whole little package. A pre-made, perfectly formed little bundle. This bundle is so special, it even has its own name. It’s called an alpha particle. Fancy, right?
Now, what’s in this little alpha package? It’s not just protons or just neutrons. Nope, it’s a dynamic duo. It’s made of two protons and two neutrons, all snuggled up together. Like a tiny, nuclear bromance.
Think of it as the nucleus having a little snack and then saying, "You know what? I’m going to shed this bit and feel better." It’s a form of atomic diet, I suppose. A very energetic diet.
This ejection process is known as alpha decay. It’s a fancy term for an atom saying, "Peace out!" to a part of itself. And this "peace out" part is our little alpha particle.

It’s not a gentle whisper, though. When an atom undergoes alpha decay, this alpha particle comes shooting out with quite a bit of gusto. It’s got energy, it’s got speed. It’s not messing around.
Imagine a tiny, powerful cannonball, but made of protons and neutrons. And it’s launching itself from the nucleus. This is the birth of an alpha particle, in a nutshell. Or rather, in a nucleus.
So, we have this nucleus, right? It’s feeling a bit crowded in there. Too many particles bumping around. It’s like a tiny apartment with too many residents. Someone’s gotta move out.
And who gets the eviction notice? Not the electrons. They’re generally too light and zippy to be bothered with this particular nuclear drama. It’s the heavier guys in the middle.
The protons and neutrons decide to team up. They form a little clique, a four-person team: two protons and two neutrons. They’re the "Alpha Quartet." Catchy, isn't it?
Then, with a sudden burst of energy, they’re expelled from the nucleus. It’s like a mini-explosion, but contained within the atom. A very specific, targeted explosion.
This expulsion is the key to creating alpha particles. It’s the atom’s way of saying, "I have too much of a good thing, so I’m going to share… by throwing it at you!"
The element that’s doing the shedding changes, by the way. It’s no longer the same element. It’s like it’s lost some of its identity. A bit sad, but also, a new beginning for a lighter atom.
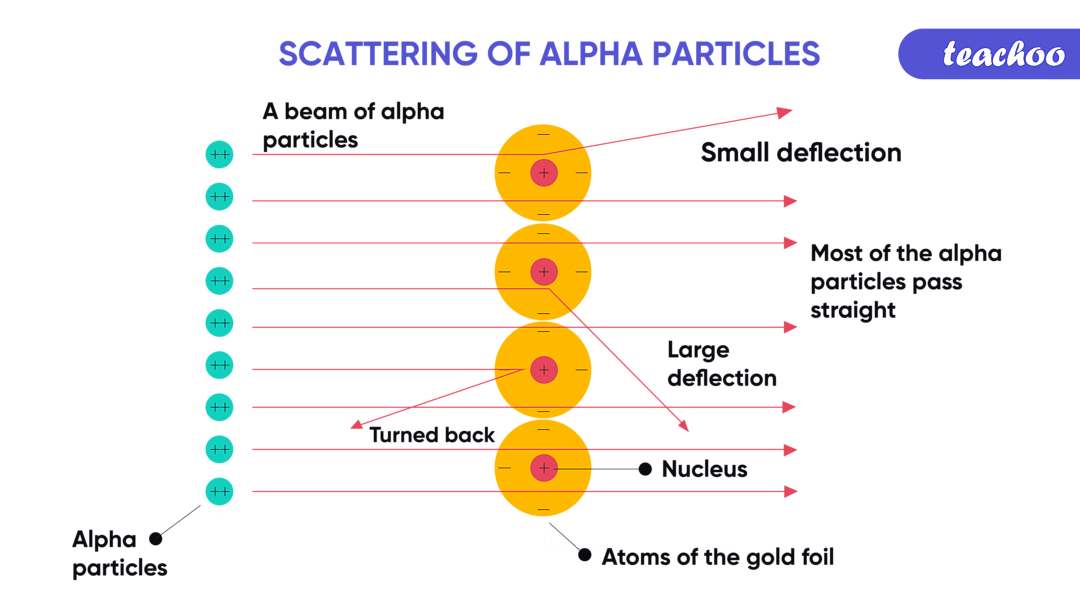
And the ejected alpha particle? It’s a helium nucleus. Yep, the same stuff they put in balloons to make them float! Though this alpha particle is a bit more… radioactive.
So, a plump, unstable nucleus decides it’s time for a change. It’s tired of being so heavy and energetic. It wants a simpler life. Or at least, a less heavy life.
It identifies a specific group of particles within itself – two protons and two neutrons. These guys are the chosen ones. The ones destined for a journey.
With a surge of internal force, the nucleus pushes them outwards. Think of it like a very strong sneeze. A nuclear sneeze, if you will. And out pops the alpha particle.
This is the fundamental process. An atom shedding a specific part of its core. It’s a natural phenomenon, happening all the time in certain radioactive elements.
It’s not a conscious decision by the atom, of course. It’s more like a built-in mechanism for stability. When things get too wobbly, nature has a way of sorting it out.
And sorting it out often involves these little alpha particles making their grand exit. They're the byproduct of an atom's quest for peace. A very energetic, positively charged peace.
The nucleus is left behind, lighter and more stable. The alpha particle goes off on its own adventure, interacting with whatever is in its path. It’s a bit like a child leaving home. A very, very small, very energetic child.

So, the next time you hear about alpha particles, just picture a slightly overweight nucleus deciding it’s time for a brisk walk. And that walk involves kicking out a little package of protons and neutrons. Simple, really. Albeit with a touch of atomic drama.
It's like a tiny, nuclear shedding. The nucleus is the old skin, and the alpha particle is the bit that’s sloughed off. Except, it’s not skin, and it’s incredibly fast. And it carries a positive charge, which is, you know, important.
The core of the atom, the nucleus, is where the magic (or the physics) happens. It's densely packed. So packed, sometimes it just needs to let a little something go to feel better. And that "something" is our alpha particle.
It’s a bit like when you’re too full after a big meal and you just need to let out a little… well, you know. Except this is a nuclear version. Much more significant. And less embarrassing, hopefully.
The transformation of the parent atom is crucial here. It’s not just creating an alpha particle; it’s becoming a different element. Losing those two protons changes its identity entirely. Quite the identity crisis, if you ask me.
So, the process is essentially a way for certain heavy elements to achieve a more stable state. They do this by emitting these alpha particles. It’s a natural balance act, on a microscopic scale.
It’s an ongoing cycle for some elements. They’ll keep spitting out alpha particles until they reach a more comfortable atomic configuration. It’s like they’re on a mission for nuclear serenity.
And the alpha particle, this little helium nucleus, is the messenger of this atomic transformation. It’s the tangible evidence that a nucleus has undergone a change. A speedy, positively charged evidence.

We're talking about radioactivity here. And alpha decay is one of its primary forms of expression. It’s the atom’s way of saying, "Look at me! I’m changing!"
It’s not a universal process for all atoms, mind you. Only specific types of atoms, usually the heavier ones, are prone to this. Lighter atoms are generally content with their current nuclear arrangement. They’re not as dramatic.
So, the key ingredients are a heavy nucleus, a desire for stability, and the ability to form and expel a pair of protons and neutrons. It’s a very specific recipe for creating an alpha particle.
Think of it as a natural lottery. Certain nuclei win the "alpha decay lottery" and get to send out these energetic particles. It's a way of shedding excess nuclear baggage.
It’s a process that scientists have studied extensively. They understand it quite well, even if the atoms themselves are a bit mysterious about their motivations. We just observe and describe.
The fundamental idea is that the nucleus is trying to get rid of a bound cluster of two protons and two neutrons. This cluster is very stable on its own. So, if the nucleus can manage it, it’s a good deal for everyone involved.
It’s like a company spinning off a profitable division. The main company becomes smaller, and the new division goes off to do its own thing. Except here, the "division" is an alpha particle, and the "company" is an atom’s nucleus.
And that, my friends, is a glimpse into how these tiny, energetic alpha particles come to be. It’s a story of nuclear shedding and transformation, all happening at speeds we can barely imagine. Pretty neat, if you ask me.
