Primary Factor That Differentiates One Amino Acid From Another

Ever wondered what makes life, well, alive? It’s a pretty big question, but one of the coolest answers lies in the tiny, unsung heroes of our bodies: amino acids. Think of them as the Lego bricks of everything from your muscles to your mind. We’ve got about 20 different kinds, and they all team up in an astonishing variety of ways to build the complex machinery that makes you, you! So, what’s the secret sauce that makes one amino acid unique from another? It’s not rocket science, but it’s absolutely fascinating, and understanding it is key to appreciating the incredible chemistry happening inside you right now.
This isn’t just some dry biological fact; it’s the foundation of understanding how our bodies function, how we get energy, how we repair ourselves, and even how our brains send signals. It’s also incredibly useful for anyone interested in health, fitness, nutrition, or even just the science of cooking! Knowing what these little guys do can empower you to make smarter choices about what you eat and how you take care of yourself. It’s like getting a peek under the hood of the most amazing machine ever created – your own body!
The Secret Ingredient: The "R" Group!
So, what’s the big difference? Imagine a basic amino acid structure. It’s like a little central hub with a few standard attachments. You’ve got a central carbon atom, a hydrogen atom, an amino group (which is just a nitrogen atom with a couple of hydrogens), and a carboxyl group (carbon, oxygen, and hydrogen). These are the common parts, the universal blueprint shared by all 20 amino acids. They’re like the four basic connector pegs on every Lego brick.
But here’s where the magic happens, the primary factor that sets them apart: the side chain. Scientists affectionately call this the "R" group. This R group is attached to that central carbon atom, and it’s the only part that varies from one amino acid to another. Think of it as the unique shape or color of each Lego brick. This seemingly small difference has a monumental impact on the amino acid's properties and how it behaves.
These R groups can be incredibly diverse! Some are simple, like a single hydrogen atom in glycine (the smallest amino acid – talk about a basic brick!). Others are slightly more complex, like in alanine, with a short carbon chain. Then you have R groups that are quite elaborate:
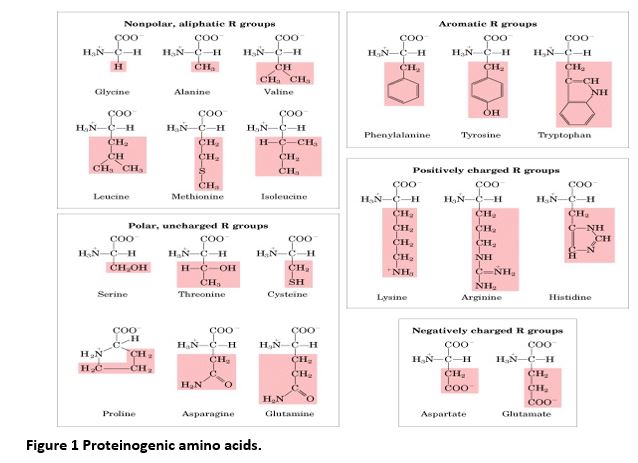
Some are positively or negatively charged, allowing them to interact with other charged molecules. Others are bulky and hydrophobic, meaning they don't like water and tend to hide away from it. Then there are those that are polar, meaning they have a slight electrical attraction and are happy to mingle with water.
It’s these R groups that dictate everything about an amino acid’s personality. Do they prefer to be on the outside of a protein, interacting with the watery environment of our cells, or do they tuck themselves away on the inside? Will they form strong bonds with other amino acids, or will they keep their distance? Will they carry a positive charge, a negative charge, or be neutral?

Why This "R" Makes All the Difference
The incredible diversity of these R groups allows for the formation of an almost infinite number of different proteins. Proteins aren’t just random strings of amino acids; they fold up into specific, intricate 3D shapes. This shape is absolutely critical for the protein's function. Think of a key needing to fit perfectly into a lock – the precise shape matters!
The unique R groups are like the little nubs and grooves on the key that allow it to interact with the lock in a specific way. A protein that acts as an enzyme, speeding up chemical reactions, will have an R group positioned just so in its active site to grab onto its target molecule. A protein that provides structural support, like in your muscles, will have R groups that allow it to form long, strong fibers. And proteins that act as signals in your nervous system will have R groups that can interact with receptors on other cells.
So, the next time you hear about amino acids, remember the humble R group. It’s the unsung hero, the tiny detail that gives each amino acid its unique character and, collectively, allows for the vast complexity and wonder of life itself. It’s a beautiful example of how small differences can lead to enormous outcomes, a principle that applies far beyond the world of biology!
