Pressure Has An Appreciable Effect On The Solubility Of
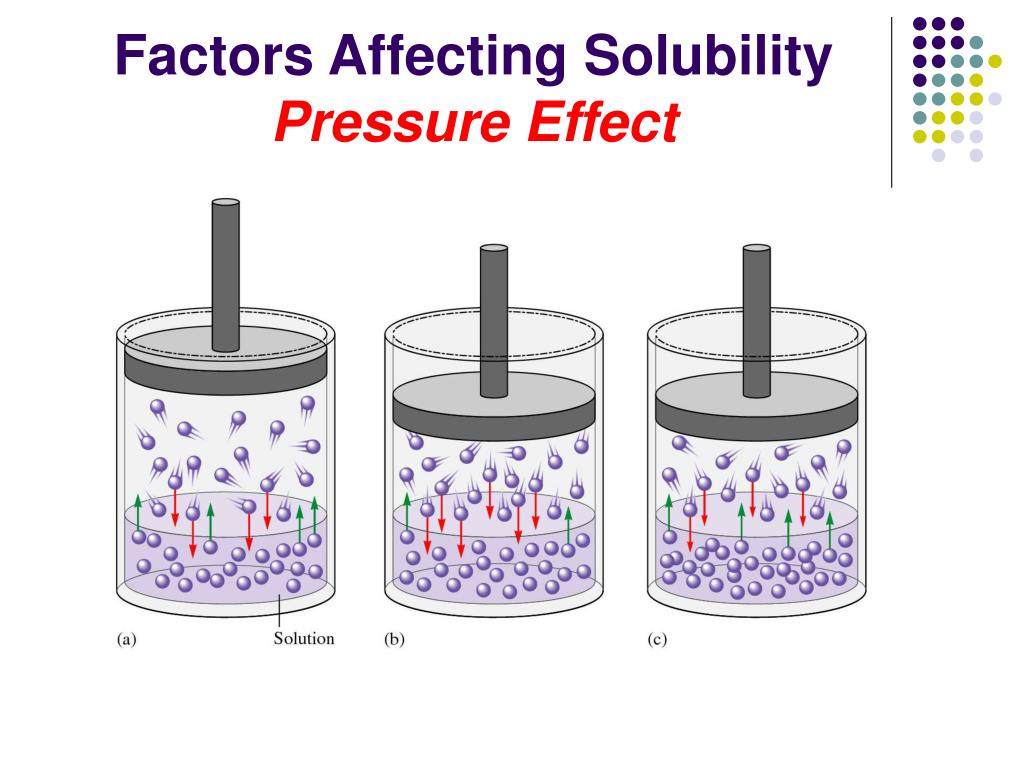
Hey there, science curious friends! Ever wondered why some things dissolve like a dream and others put up a real fight? Well, get ready to have your mind gently blown, because today we’re diving into the surprisingly exciting world of how a little thing called pressure can make a huge difference in how easily stuff dissolves.
Think about it. We’re all about the easy life, right? When you’re making lemonade, you want that sugar to vanish into the water pronto. Nobody wants crunchy sugar at the bottom of their drink!
Now, imagine you’re trying to dissolve something tricky, something that’s being a bit stubborn. What if I told you that you could give it a little nudge, a gentle push, and suddenly it’s like, “Okay, fine, I’ll dissolve!” That, my friends, is where our star player, pressure, comes in.
It’s like a secret handshake for dissolving. A little bit of squish, and poof, things change!
The Soda Pop Sensation!
Let’s talk about your favorite fizzy drink. You know, the one that makes that delightful “psssst!” sound when you open it? That’s pressure showing off its dissolving skills!
Inside that sealed can or bottle, there’s a whole lot of carbon dioxide gas chilling out. It’s been squeezed in there at a pretty high pressure. This squishing action forces the gas to dissolve really well into the liquid.
But then, you pop the top! Suddenly, that high pressure is gone. It’s like a dam bursting! The dissolved gas, feeling all free and adventurous, can’t wait to escape. So it bubbles up and out you go!
This is a super fun example of how increasing pressure can make gases dissolve more in liquids. It’s the reason your soda is so delightfully bubbly!

Beyond the Bubbly
While soda is a fantastic and delicious example, the principle applies to other situations too. It’s not just about making things disappear into water, though water is pretty darn good at dissolving things.
Imagine you have a really stubborn stain on your favorite shirt. You’ve tried all sorts of fancy stain removers. What if, in some magical (and slightly exaggerated) lab, we could crank up the pressure on that stain?
Under immense pressure, even the most determined stain molecules might throw in the towel and dissolve into the cleaning solution. It’s like giving the solvent a super-powered hug to coax the stain into leaving!
Of course, we don’t have giant pressure chambers for our laundry (yet!), but it’s fun to think about the possibilities.
The Deep Sea Secret
Let’s take a trip to the deepest parts of the ocean. Down there, it’s dark, cold, and unbelievably… squishy. The pressure is absolutely immense!
Fish and other sea creatures have to be pretty special to survive. They’re built to handle all that squeezing.
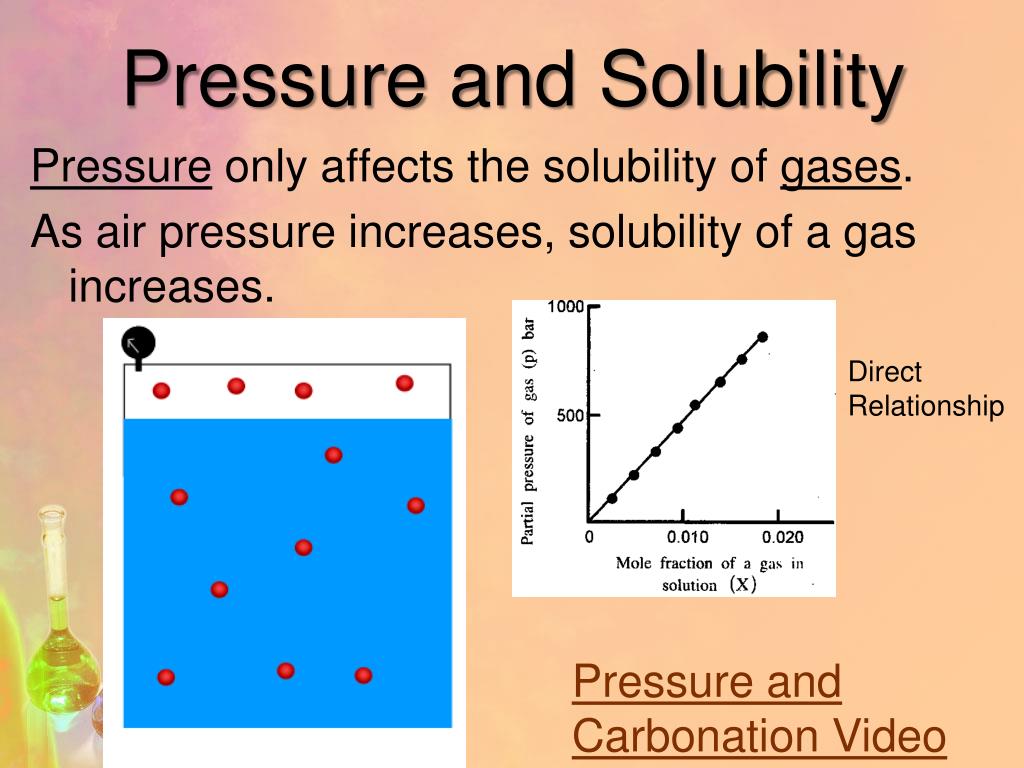
But what about the gases in the water? The same gases we breathe, like oxygen, are also dissolved in the ocean. And guess what? At those crushing depths, more oxygen and other gases can be dissolved into the water than you’d find near the surface.
This is a big deal for marine life! They’re essentially living in a highly pressurized, super-oxygenated soup.
It's a reminder that pressure is a constant, powerful force shaping our planet and the life within it.
Scuba Divers and the Bends
Now, this is where things get a little more serious, but still fascinating. Scuba divers know all about the effects of pressure on dissolving gases.
When divers go down deep, they breathe air from tanks. Because they are under increased water pressure, more nitrogen from the air dissolves into their blood than would normally happen at the surface.

This isn't a problem while they're down there. Their bodies are used to it.
The trouble starts when they ascend too quickly. If they rise too fast, the surrounding pressure decreases rapidly. The dissolved nitrogen, which was happily chilling in their blood, suddenly wants to escape, just like the carbon dioxide in your soda!
This can form tiny bubbles in their bloodstream and tissues, causing a condition called decompression sickness, or the "bends." It's a painful and potentially dangerous reminder of how crucial controlled pressure changes are for the human body.
So, when you see a scuba diver, remember they’re navigating a world where pressure is king and dissolving gases are playing a vital, and sometimes tricky, role!
A Sweet Example: Candy Making!
Let’s get back to something a little lighter, shall we? Think about making hard candy or caramel.
When you’re heating sugar and water to make these treats, you're essentially creating a very concentrated sugar solution. If you were to then increase the pressure on this super-sweet liquid, you could actually dissolve even more sugar into it!

This is a bit of a playful exaggeration for home cooks, as we don't usually mess with pressure when making candy. But in industrial settings, understanding how pressure affects solubility is key to getting that perfect, smooth texture in your favorite sweets.
It’s like giving the sugar molecules a little nudge and saying, “Come on, there’s plenty of room in here for you!”
The Takeaway: Pressure is a Powerful Pal!
So, there you have it! Pressure isn't just some abstract scientific concept; it’s a real, tangible force that has an appreciable effect on the solubility of things all around us.
From the satisfying fizz in your soda to the mysterious depths of the ocean, pressure is constantly at work, influencing how well things dissolve.
It’s a reminder that even the seemingly simple act of dissolving is a complex dance, and pressure is one of the most important dance partners!
Isn't science just the coolest? Keep exploring, keep questioning, and remember that even a little bit of squish can make a big, bubbly difference!
