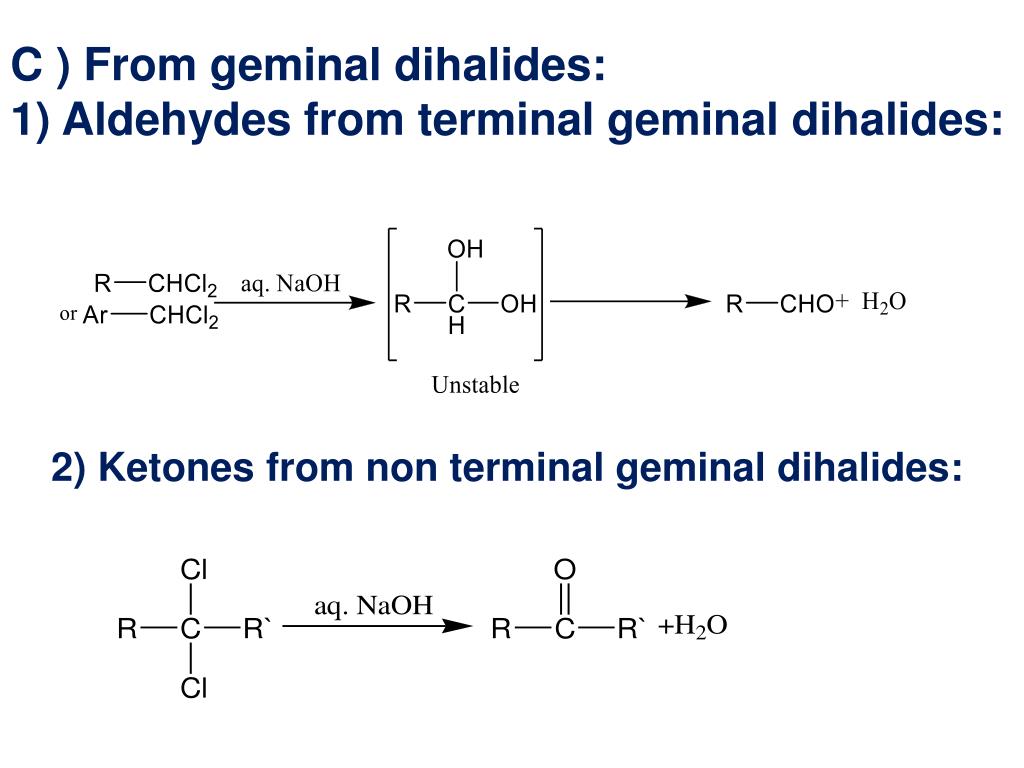
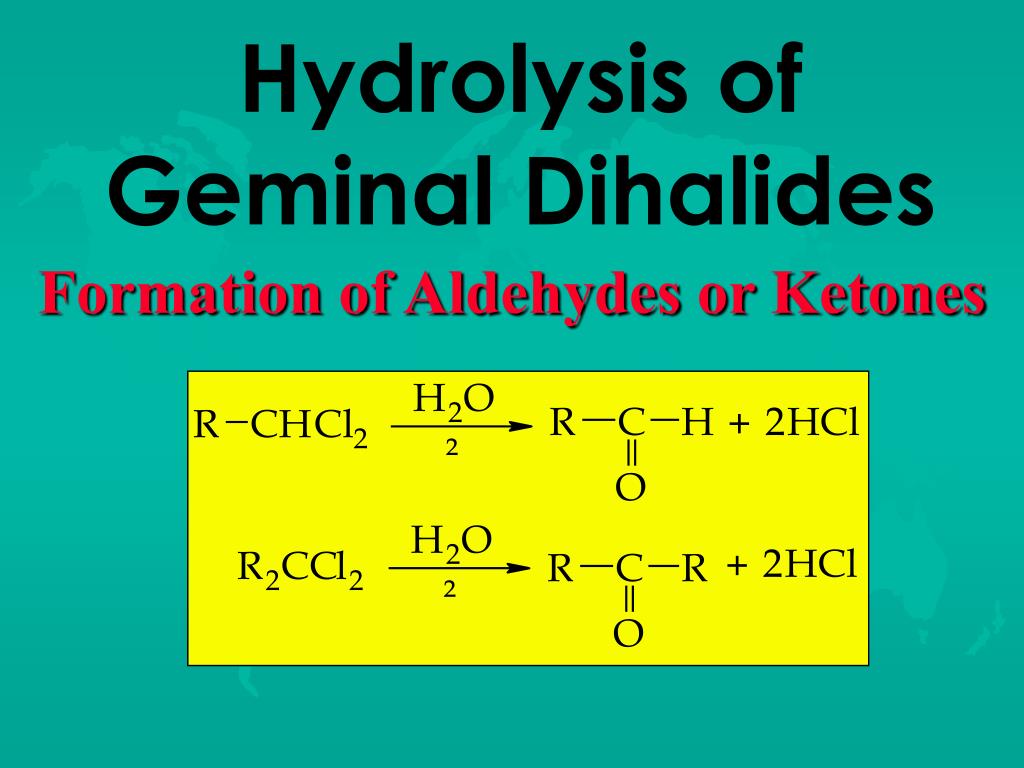
Preparation Of Aldehydes And Ketones From Geminal Dihalides
Alright folks, gather 'round! We're about to dive into a little corner of chemistry that might sound super fancy, but trust me, it's just a bunch of molecules having a party. Today, we're talking about how to whip up some delicious aldehydes and ketones. Now, I know what you're thinking, "What's an aldehyde? What's a ketone?" Don't worry your pretty little heads about it. Think of them as the cool kids of the carbonyl family, the ones that get all the attention. They're in perfumes, flavors, and all sorts of things that make life a bit more interesting. And how do we make them? Well, we're going to start with something a bit… well, let's just say it's got a few extra friends it's trying to get rid of. We're talking about geminal dihalides.
Imagine a carbon atom that's holding onto two identical halogen buddies. Like, double the trouble, double the chlorine, or bromine, or iodine. These are our geminal dihalides. They're a bit… clunky. Two big halogens stuck to the same carbon can't be that comfortable, right? It’s like wearing two coats in July. Something's gotta give!
So, what do we do with these slightly awkward molecules? We give them a little nudge. A little push. We introduce them to a trusty sidekick. And that sidekick, my friends, is usually a base. Think of a base as that friend who's always ready to help you clean up a mess or, in this case, help a molecule shed some baggage. And when I say "shed some baggage," I mean those two halogens are about to make a dramatic exit.
It's a chemical glow-up, really. From a slightly overloaded carbon to something sleek and, dare I say, more functional.
Now, the exact way this happens is where things get a bit… theatrical. We're not just going to ask those halogens nicely to leave. Oh no. We're going to encourage them. We're going to introduce some heat, some really good vibes (which in chemistry means the right temperature and solvent), and let our base do its magic. It’s like throwing a surprise party for the carbon atom where the guests of honor are none other than a very fancy aldehyde or a sophisticated ketone.

When you have a geminal dihalide with the two halogens on the same carbon, and you bring in a strong base, something truly wonderful happens. That carbon starts to feel the pressure. It’s like, "Okay, two halogens are too much. I need to simplify!" So, it starts to break bonds. And as it breaks those bonds, it gets ready to form a new, much more exciting relationship. This is where the double bond magic happens. A double bond between the carbon and an oxygen atom. That, my friends, is the signature of our aldehyde and ketone pals.
Let’s break it down a smidge, but not too much, because we want to keep it light. The base comes in and snatches away a proton (which is just a fancy word for a hydrogen atom that's lost its electron). This creates a little opening, a vulnerability. Then, one of the halogens decides it's had enough and bails. It takes its electron buddies and exits stage left. This leaves our carbon with a positive charge, looking a bit lonely. But fear not! Because the remaining halogen, seeing its friend go, also decides, "You know what? I'm out too!"

Here's the really neat part. As the second halogen leaves, the carbon atom, which is now craving some stable relationships, forms a beautiful double bond with an oxygen atom that was just hanging around, minding its own business. It’s like the carbon finally found its soulmate in oxygen. And poof! You’ve gone from a cluttered, overloaded carbon to a perfectly formed aldehyde or ketone.
The difference between an aldehyde and a ketone is subtle, like the difference between a fancy appetizer and a gourmet main course. It's all about where that double bond to oxygen is located and what else is attached. But for our purposes today, the important thing is that we’ve successfully transformed our slightly awkward geminal dihalide into something much more desirable. Something that smells good, tastes good (in some cases, maybe don’t go tasting random chemicals!), and generally makes the world a more interesting place.
It's honestly one of those transformations that makes you feel like a bit of a chemist wizard. You take something that seems a little… meh, and with a few clever steps, you turn it into something that’s actually quite brilliant. And all it takes is a little bit of patience, the right reagents (that's chemistry lingo for ingredients), and a willingness to let those halogens go. Because sometimes, shedding the extra baggage is exactly what you need to become your best, most functional self. Or in this case, your most delicious, fragrant self!
So, next time you’re enjoying a nice pastry or smelling a lovely perfume, just spare a thought for the humble geminal dihalide. It might have played a starring role in that delightful experience, and it all started with a little chemical shake-up. It’s a bit like how you might get rid of a messy roommate to have a more organized and enjoyable living space. Except, you know, with molecules and double bonds. Much cleaner, in the long run!
