Predicting The Relative Boiling Points Of Pure Substances

Ever stared at a bunch of liquids and wondered which one would be the first to throw a hissy fit and turn into a gas? You know, like, which one is the most eager to ditch its liquid life for a more carefree, floaty existence? Well, welcome to the club! We’re not talking about rocket science here, folks. We're talking about good old-fashioned, slightly unscientific, "my gut feeling says so" boiling point prediction.
Think about it. We’ve all got our favorites. Some folks love a good cup of coffee. Others swear by a soothing mug of tea. And then there are the truly adventurous who dabble in things like... well, let's just say acetone for nail polish removal. They all boil, right? Eventually. But which one gets there first? It’s a mystery, a tantalizing puzzle for the casually curious.
Now, before the serious scientists start adjusting their spectacles, let me tell you something. This isn't about precision. This is about instinct. It's about looking at a substance and having a hunch. It’s like knowing your dog wants a walk just by the way it cocks its head. You just know.
My unofficial rule of thumb? Bigger things tend to be lazier. They need more convincing to get going. So, a big, chunky molecule? It's probably going to take its sweet time to boil. It's got more baggage, you see.
Imagine water. It’s pretty common, right? Relatively small. It boils at a respectable 100 degrees Celsius. Now, think about something like, say, cooking oil. It’s a bit more… substantial. It feels like it should take longer to get excited. And guess what? It usually does. It likes to hang around in its liquid form a little longer, enjoying the heat before it decides to bubble up.

And then there are those substances that seem to have a personal vendetta against staying liquid. They are just itching to escape. These are your low-boiling point superstars. They're the party animals of the molecular world. They hear the music of heat and they are gone!
Let’s consider something simple. Alcohol. You know, the kind you might find in hand sanitizer or perhaps something a bit more… celebratory. It’s generally less “heavy” than water. It’s more eager to be free. So, you can bet your last dollar that if you put alcohol and water side-by-side and turned up the heat, the alcohol would be packing its bags and heading for the gas phase way before the water even started to sweat.

It’s all about how much energy these little molecules need to break free from their buddies. If they're clinging to each other like they just saw a spider, it's going to take a lot more oomph to pry them apart. And those who are more independent? They'll be off with a wave and a smile.
Think about the smell. Sometimes, the smell can be a clue, can't it? If something smells really sharp and potent, like it’s really trying to get your attention, it might be a bit of a gas-fiend. It’s got that energetic vibe. On the other hand, if it’s mellow and smooth, it might be content to stay put for a while.
Now, I’m not saying you should start sniffing around industrial chemicals. Please, for the love of all that is safe, do not do that. But in a controlled, kitchen-science kind of way, you can start to get a feel for it. Ever boiled milk? It’s different from water, isn’t it? It seems to have a bit more… reluctance. It’s got those extra bits and bobs in it.
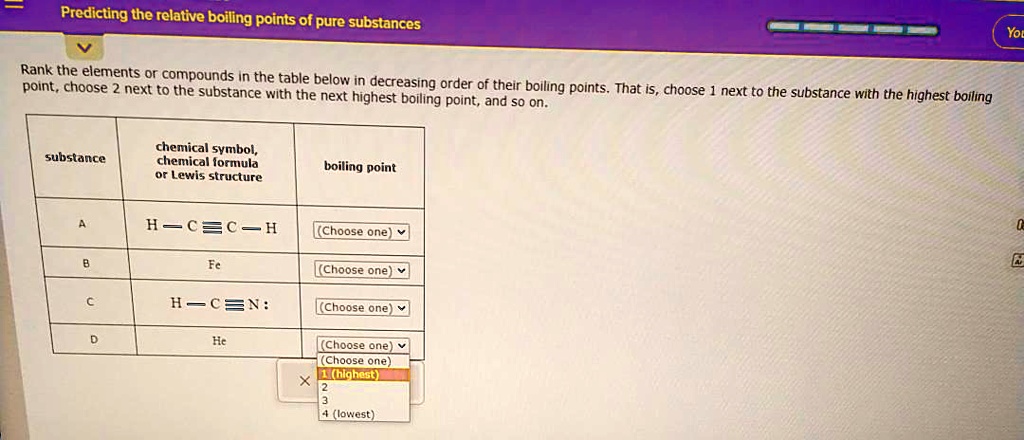
And what about things that are already pretty gassy? Like helium? You’re not going to boil helium on your stovetop. It’s already so eager to be a gas, it basically exists that way unless you go to extreme cold. It’s the ultimate free spirit. It’s so far up the boiling point scale, it’s practically in a different dimension.
So, the next time you’re faced with a variety of liquids, take a good look. Consider their apparent “weight” or “stickiness.” Does it seem like a molecule that’s going to be easily tempted by a little heat, or is it going to need a full-blown rave to get it to vaporize? It’s an art, really. An art of observation and a dash of educated guesswork.

And if you’re wrong? Well, that’s half the fun! Science is all about experimenting, right? Even if your experiment is just a mental one while you’re waiting for your tea to steep. You’re predicting, you’re observing, and you’re learning. All from the comfort of your own kitchen. Who needs a fancy lab coat when you’ve got a curious mind and a pot of boiling water?
It’s about developing that feel. That intuition. That subtle understanding that some things are just naturally more predisposed to change their state. It's a little bit of magic, a little bit of common sense, and a whole lot of fun. So go forth and predict, my friends. Predict with confidence, predict with humor, and predict with the joyful knowledge that you're engaging in a highly unofficial, but undeniably entertaining, scientific pursuit.
