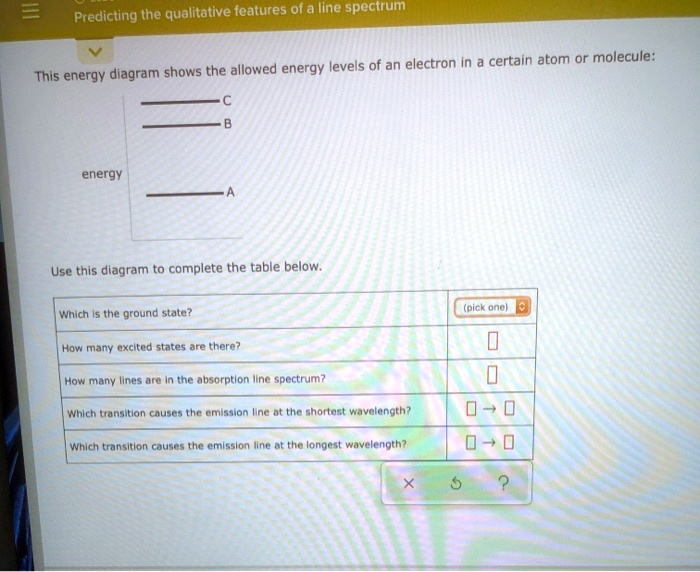
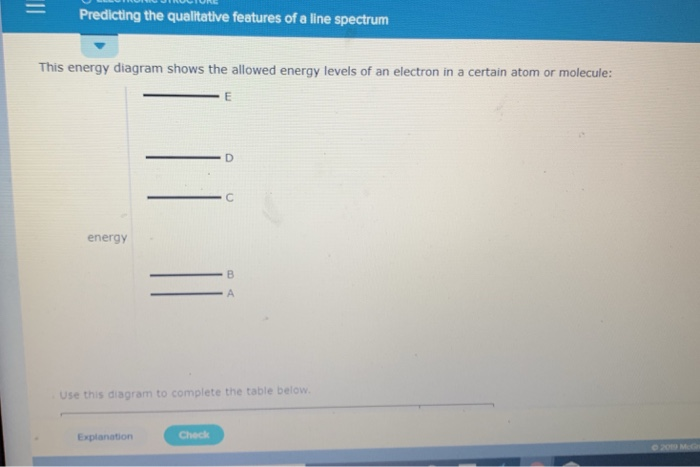
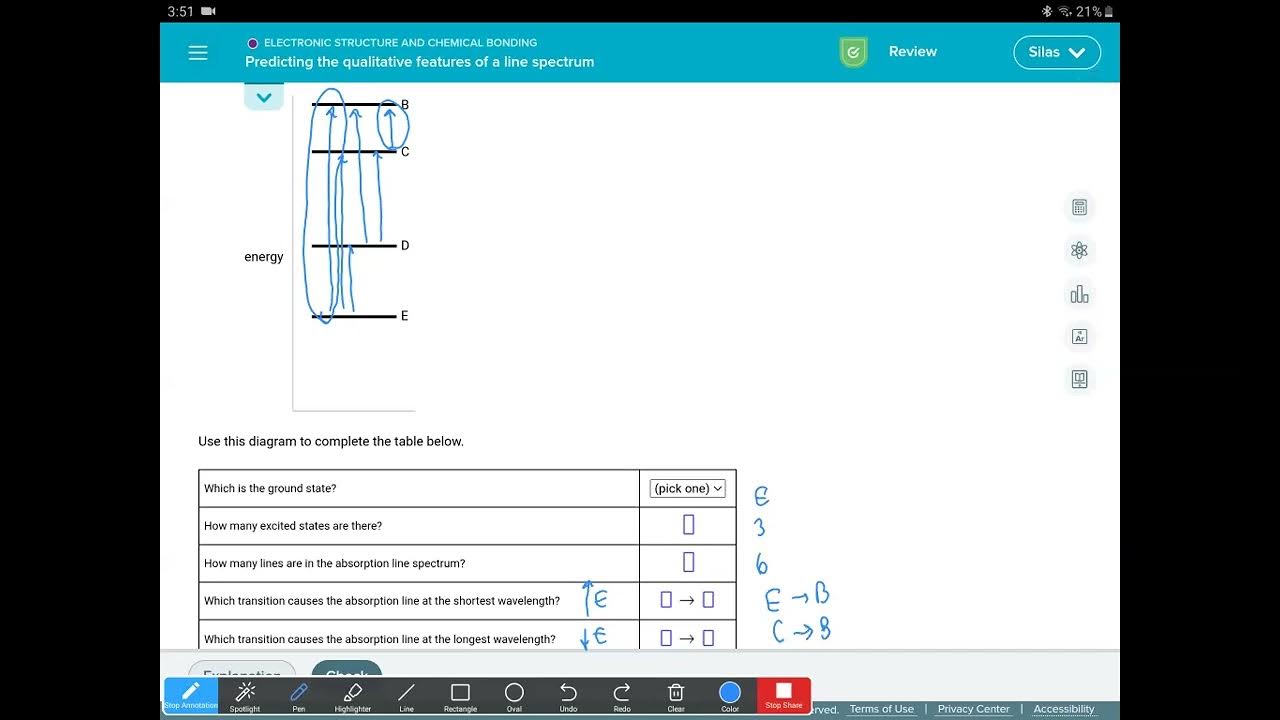
Predicting The Qualitative Features Of A Line Spectrum
So, there I was, wrestling with a particularly stubborn jar of pickles. You know the kind – the ones that have been sitting in the back of the fridge for, let's be honest, a little too long. I tried everything: the rubber grip, banging it on the counter (gently, of course… mostly), even enlisting the help of my perpetually unimpressed cat, Bartholomew, to stare it into submission. Nothing. Just this infuriating, sealed-tight defiance.
Finally, in a moment of sheer, unadulterated frustration (and a healthy dose of pickle craving), I remembered a trick. I ran the lid under hot water for a minute. And poof! It popped open. Just like that. All that metal and glass, once a solid, unyielding barrier, suddenly yielded to a simple change in… well, its qualitative feature. It went from "cold and contracted" to "warm and expanded."
And it got me thinking. We often focus on the how much of things, the quantities. How many pickles? How much does it cost? But sometimes, the real magic, the actual unlocking of a problem, lies in understanding the what kind – the qualitative features. And that's precisely what we're going to chat about today, but instead of pickles, we're diving into the fascinating world of line spectra.
The Whispers of the Universe: What is a Line Spectrum Anyway?
Okay, so maybe "whispers" is a bit poetic for what is essentially light. But honestly, when you think about it, light carries so much information! Imagine each atom, each molecule, like a tiny, unique instrument. When you "play" it – by heating it up, or zapping it with electricity, or whatever other scientific shenanigans you get up to – it doesn't just emit a continuous, mushy hum of light. Oh no, it’s much more sophisticated than that.
Instead, it emits light at very specific, discrete wavelengths. Think of it like a musical instrument playing only certain notes, not a smooth slide up and down. When you spread this light out using something like a prism (remember those from school? Science class was way cooler than I gave it credit for), you don't get a rainbow. You get a series of bright, distinct lines, each at its own precise color (or wavelength). This, my friends, is a line spectrum. It’s like the atom's unique fingerprint. Pretty cool, right?
And it’s not just for excited atoms. When light from a distant star passes through a gas cloud, some of those wavelengths get absorbed by the atoms in the cloud. This absorption also happens at specific wavelengths, creating dark lines in the otherwise continuous spectrum of the star. This is called an absorption spectrum. It’s like the atom's "imprint" in reverse, showing us what it didn't let through.
Why Should We Care About These Lines? (Besides the Obvious Cool Factor)
This is where things get really interesting. These line spectra aren't just pretty patterns. They are packed with information. Think about it: if each element has a unique spectral fingerprint, what does that mean?
It means we can actually tell what elements are present in a distant star, a nebulae, or even a sample right here in the lab, just by looking at the lines in its spectrum. We don't need to fly there and grab a sample (which, let's be real, is a bit of a logistical nightmare for most stars). We can analyze the light they send us. It’s like being a cosmic detective, piecing together clues from afar.
But it’s not just about what is there. These lines can also tell us about the conditions under which the light was produced or absorbed. This is where we start talking about predicting qualitative features.
Predicting The Qualitative Features: It’s Not Just About “Is It There?”
So, we know we can identify elements. That's a great starting point. But what else can these spectral lines tell us? This is where the "predicting qualitative features" part comes in. It's about understanding the nature of the light source, not just its composition.
The Subtle Nuances of Intensity
Let's start with something seemingly simple: the intensity of the spectral lines. You might think, "Okay, some lines are brighter than others. So what?" Well, the brightness of a spectral line is related to how many atoms are emitting or absorbing light at that particular wavelength. More atoms, brighter line. Less atoms, fainter line.
This seems straightforward, but it gets more nuanced. The intensity of a line can also be affected by the temperature of the source. At different temperatures, atoms will be in different energy states. Some energy transitions (which produce specific spectral lines) are more probable at certain temperatures than others. So, by looking at the relative intensities of different lines from the same element, we can actually make educated guesses about the temperature of that element's environment.
Imagine a gas cloud. If it's relatively cool, certain spectral lines might be more prominent. If it's superheated, other lines will dominate. It's like looking at Bartholomew: when he's curled up asleep, he has a certain "intensity" of coziness. When he's chasing a laser pointer, his "intensity" of chaos is significantly higher. Different states, different observable qualities.
The Doppler Effect: Is It Coming Towards Us or Running Away?
This one is a classic and incredibly powerful. You've probably experienced the Doppler effect with sound. Think about an ambulance siren. As it approaches you, the pitch sounds higher. As it moves away, the pitch sounds lower. This happens because the sound waves are compressed or stretched by the motion of the source.
The same principle applies to light! If a star or galaxy is moving towards us, the light waves it emits will be compressed, shifting their wavelengths towards the bluer end of the spectrum. This is called a blueshift. Conversely, if it's moving away from us, the light waves will be stretched, shifting their wavelengths towards the redder end. This is a redshift.
So, by comparing the observed wavelengths of spectral lines in an object to their known laboratory wavelengths, we can tell if that object is approaching or receding from us, and even how fast! This is how we discovered that the universe is expanding, a truly mind-blowing realization. It’s not just about what's in the star, but also about its motion. Qualitative feature number two, unlocked!
The Broadening of Lines: Things Are Complicated Out There
Sometimes, spectral lines aren't perfectly sharp. They can be broadened. And just like intensity and redshift, this broadening tells us something important about the conditions in the light source.

One common cause of broadening is the motion of atoms within the source. Even if the bulk of the gas cloud is moving away from us (causing a redshift), individual atoms within that cloud are constantly jiggling and moving around. Some will be moving towards us, some away, and some will have velocities perpendicular to our line of sight. These random motions cause their emitted or absorbed light to be slightly shifted in different directions. When we observe the whole ensemble, the spectral line appears broadened. The faster the atoms are moving, the broader the line. So, line broadening can be a proxy for temperature or the degree of turbulence within the gas.
Another factor that can cause broadening is the density of the gas. In very dense environments, atoms can interact with each other more frequently. These interactions can disrupt the energy levels of the atoms, leading to a broadening of the spectral lines. So, if you see a particularly broad line, it might be a clue that you're looking at a dense region.
And then there’s pressure broadening, which is related to density. The more the atoms are bumping into each other, the more the spectral lines get "squashed" and spread out. It's like trying to have a quiet conversation in a crowded, noisy pub – the clarity of your words gets a bit compromised, doesn't it?
The Polarization of Light: A Twist in the Tale
This is a more advanced, but equally fascinating, qualitative feature. Sometimes, the light emitted by an object is polarized. What does that mean? Light waves have an oscillating electric field that vibrates in a certain direction. If these vibrations are predominantly in one direction, the light is polarized.
Polarization can occur in several ways. For instance, if light is scattered off particles, it can become polarized. Also, in the presence of strong magnetic fields, the energy levels of atoms can be split (this is the Zeeman effect). When atoms in these split energy levels emit light, the emitted light can be polarized in specific directions, depending on the orientation of the magnetic field.
By measuring the polarization of spectral lines, we can gain insights into the strength and orientation of magnetic fields in astronomical objects. This is incredibly important for understanding phenomena like solar flares, the behavior of stars, and even the structure of distant galaxies.
Putting It All Together: A Symphony of Information
So, you see, a line spectrum is not just a collection of bright or dark lines. It's a rich tapestry of information, woven from the fundamental properties of atoms and the environment they inhabit.
By analyzing the presence of specific lines, we identify elements. By looking at their intensities, we get clues about temperature and abundance. By observing their shifts (redshift/blueshift), we understand their motion. By studying their broadening, we infer density, turbulence, and temperature. And by measuring their polarization, we can even probe the presence and strength of magnetic fields.
It’s this combination of qualitative features that allows us to paint a detailed picture of incredibly distant and inaccessible objects. It’s how we know that stars are made of hydrogen and helium, how we chart the expansion of the universe, and how we study the violent processes happening on the surfaces of other worlds.
Next time you look up at the night sky, remember that those distant pinpricks of light are not just passive points of illumination. They are actively communicating with us, sending us their stories encoded in the subtle nuances of their spectral lines. And with a bit of scientific detective work, we can decipher those stories, predicting the qualitative features of their existence. Pretty amazing, wouldn't you say? Now, if only I could apply this same scientific rigor to that stubborn pickle jar...
