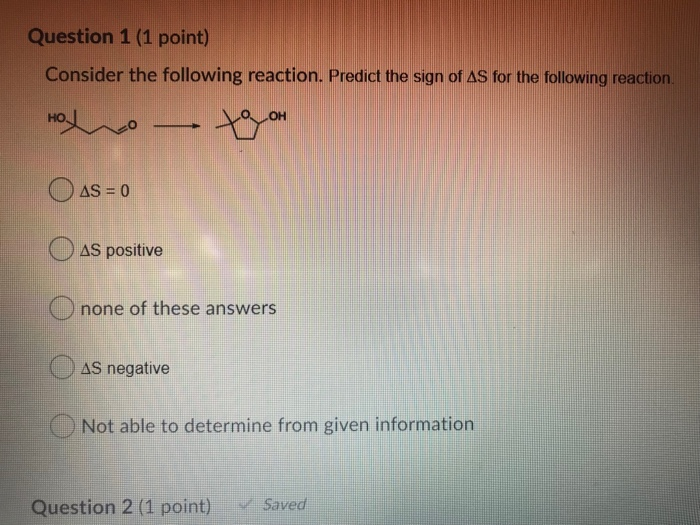
Predict The Sign Of Delta S For The Following Reaction
Hey there, science enthusiasts and folks who just kinda know things without knowing how they know them! Ever looked at a messy room and thought, "Yeah, that's definitely going in one direction: chaos"? Or maybe you've seen a perfectly organized pantry and wondered, "How did that even happen? It must have taken a lot of effort." Well, guess what? You've already been dabbling in thermodynamics, my friends. Specifically, you've been intuiting the second law of thermodynamics, which, in a nutshell, talks about the universe's unwavering love for messiness. And today, we're going to peek behind the curtain at one of its favorite little helpers: Delta S. Think of Delta S as the universe's official mood ring for how things are spreading out, getting mixed up, or generally becoming less… ordered. It's like the universe's way of saying, "Let's all just chill and mingle!"
Now, before you start picturing me in a lab coat, furiously scribbling equations on a whiteboard, let me assure you, we're keeping this super chill. We're not going to get bogged down in complex math. Instead, we're going to use our everyday experiences, the ones where you've seen something go from neat to noteworthy, or from a single entity to a whole bunch of dispersed bits. We're going to predict the sign of Delta S for different reactions, which basically means figuring out if things are getting more spread out (positive Delta S, hooray for mingling!) or less spread out (negative Delta S, aww, a bit too much order for my liking!).
Let's start with a scenario most of us can relate to. Imagine you've just baked a batch of cookies. They're fresh out of the oven, smelling like pure happiness and a little bit of temptation. All the ingredients – the flour, sugar, chocolate chips – were separate before they went into the mixing bowl. Then you went to work, mixing, stirring, maybe even a bit of enthusiastic mashing. When those cookies come out, all those separate bits are now beautifully integrated, all snuggled up together in a delicious, edible package. So, if we were to think about the mixing process itself, before they even bake, what happened to the order of things? We took distinct ingredients and made them a jumbled, delightful mess in the bowl, right? That's a classic example of an increase in entropy, or a positive Delta S. The universe is much happier when things get mixed up, like a giant cosmic potluck where everyone brings something delicious and it all gets blended into one amazing feast.
Now, let's flip that. Imagine you have a perfectly arranged deck of cards. Every suit is in order, from ace to king. It's a picture of supreme order. Now, hand that deck to a toddler. What happens? Poof! Cards are everywhere. Scattered. They’re under the couch, they’re stuck to the dog, they’re probably in the jam jar. Is that more or less ordered than the original deck? Definitely less. The cards have gone from a tightly controlled, predictable arrangement to a state of delightful chaos. This change, from order to disorder, means we've seen an increase in entropy. So, for that card-shuffling toddler scenario, Delta S is positive. The universe is basically applauding the spread! It's like the universe saying, "Yes! More freedom! Less rigid structure!"
The Great Phase Transition: From Solid as a Rock to Gaseous Goodness
Let's talk about states of matter, which are basically different levels of "chill" for molecules. Think about ice. Ice is super organized. The water molecules are all locked in a rigid, crystalline structure. They're like tiny soldiers standing at attention, not moving much except for a little shimmy. It's the epitome of order. Now, what happens when we melt that ice? We get liquid water. The molecules are still pretty close together, but they can now slide past each other. They’re not standing at attention anymore; they’re more like a crowd at a concert, milling around but still in close proximity. This is a step towards less order, a bit more freedom to move.
But let's take it a step further. What happens when we boil that liquid water? We get steam, which is a gas. Now, those water molecules are like teenagers let loose at a party. They're zipping around everywhere, bouncing off walls, and generally occupying way more space than they did when they were liquid. They are far, far apart and moving with wild abandon. Is this more or less ordered than liquid water? Oh, it's way less ordered! The molecules are scattered, spread out, and have tons of freedom. So, going from solid ice to liquid water, and then from liquid water to gaseous steam, represents a significant increase in entropy. For these phase transitions, Delta S is positive. The universe loves it when things break free and spread out like that. It’s like watching your carefully arranged bookshelf suddenly get redecorated by a mischievous windstorm – more spread, more entropy!
Think of it like this: If you have a neatly arranged box of LEGOs, that's your solid. If you spill them all over the floor, that's your liquid (a bit more spread out, but still somewhat contained). If you then somehow manage to get those LEGOs to float around the entire room independently, that's your gas. The messier and more dispersed, the higher the entropy.
Mixing Things Up: The Joy of a Good Blend
Here’s another everyday scenario. You're making a salad. You’ve got crisp lettuce leaves, bright red tomatoes, maybe some crunchy cucumbers, and little cubes of cheese. Everything is separate, in its own little pile. Now, you toss it all together. What happens? The lettuce mixes with the tomatoes, the cheese mingles with the cucumbers, and suddenly you have a unified, delicious salad. Before, you could easily pick out the tomatoes. Now, they're all mixed up in the leafy goodness. Did the order increase or decrease? It definitely decreased. Things are now intermingled, less segregated. This is a big win for entropy. So, when you mix distinct substances together, especially if they don't form some kind of super-ordered crystalline structure, you're generally looking at a positive Delta S. It's the universe's way of saying, "Let's get this party started! Bring on the mingling!"
Imagine putting a drop of food coloring into a glass of clear water. At first, the color is concentrated in that one spot. But then, slowly but surely, it starts to spread. It diffuses throughout the entire glass, turning the whole thing a lovely shade of blue (or red, or green!). Did the dye molecules become more ordered or less ordered? They went from being all clustered together to being spread out in every nook and cranny of the water. That's a massive increase in disorder, a huge sigh of relief from the universe. So, Delta S is positive for the diffusion of a solute into a solvent.
It's like your favorite song playing. At first, it might be a single instrument, clean and clear. But then all the other instruments join in, creating a rich, layered sound. It’s a beautiful mingling, a harmonious spread of sound waves. Less predictability, more awesomeness. That’s a positive Delta S in action.
When Things Get Organized (Boo!)
Sometimes, however, things can become more ordered. This is where the universe might let out a little groan. Think about when you're making something very specific, like a perfectly cut diamond. You start with carbon atoms, which are usually bouncing around in a more disordered state (like in graphite, for example). To form a diamond, you have to force those carbon atoms into a super-tight, incredibly ordered crystalline lattice. This requires a lot of energy and effort, and the result is a structure that is much more ordered than the starting material. In this case, the change from disorder to extreme order means entropy has decreased. So, for the formation of something like a diamond from a less ordered precursor, Delta S is negative. It’s like tidying up your room so much that it looks like a museum exhibit – impressive, but a lot less fun to live in.
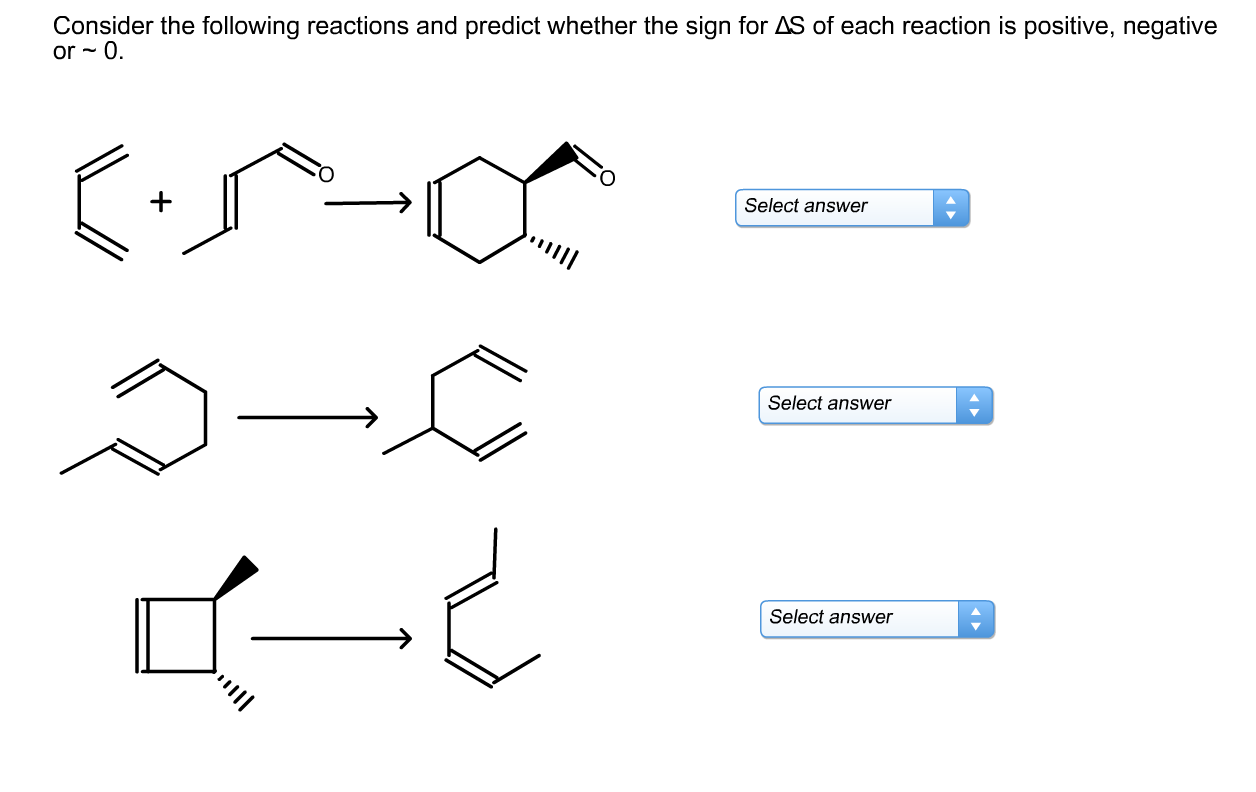
Another example is when a gas is compressed into a solid. Imagine a bunch of balloons floating freely in a room (that's your disordered gas). Now, imagine you somehow manage to squish them all together into a tiny, solid block. The molecules are now incredibly restricted in their movement. They're packed in tight, with very little freedom. This is a huge increase in order. Therefore, Delta S would be negative. The universe is not thrilled about this level of conformity.
It's like seeing a toddler's playroom, which is usually a delightful explosion of toys, suddenly become a pristine, military-style barracks. Everything is lined up, put away, and not a single toy is out of place. While it might look neat, it’s lost some of its spontaneous, joyful energy. That’s a negative Delta S – a decrease in the freedom to be messy and expressive.
The Subtle Art of Predicting Delta S
So, how do we predict the sign of Delta S without all the fancy equipment? It's mostly about looking at the number and state of the particles involved. Generally:
More Stuff = More Mess
If a reaction produces more molecules than it started with, it's usually a good bet that entropy has increased. Imagine you start with one thing and end up with three things. Those three things have more freedom to roam around and be independently chaotic. So, more products than reactants generally means positive Delta S.
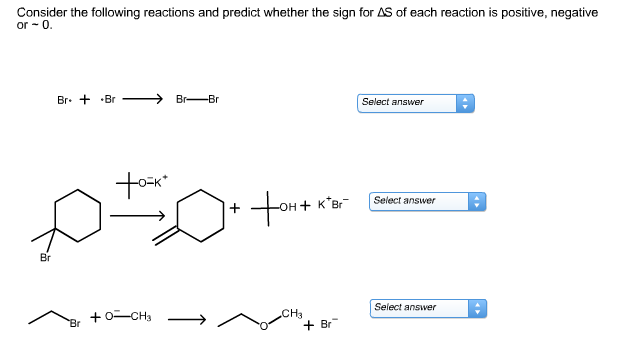
Example: 2H₂O₂(l) → 2H₂O(l) + O₂(g). Here, we start with two liquid molecules and end up with two liquid molecules and one gaseous molecule. The introduction of a gas, which is inherently more disordered, is a big clue. Plus, we've gained a molecule overall in terms of how spread out they can be.
Gas is King of Chaos
If a reaction produces a gas, especially from liquids or solids, that’s a huge entropy boost. Gases are the ultimate spreaders. They’re the life of the party, occupying every available space. So, the appearance of a gas in the products usually means positive Delta S.
Example: CaCO₃(s) → CaO(s) + CO₂(g). We're starting with a solid and ending with a solid and a gas. That CO₂ gas is going to zoom off and spread out, significantly increasing the disorder. Big time positive Delta S!
Fewer Bits = More Order
Conversely, if a reaction consumes more molecules than it produces, or if it converts gases into liquids or solids, you're usually looking at a decrease in entropy. It's like a big cleanup operation where everything gets put back into its designated place. So, fewer products than reactants, especially if gases are disappearing, generally means negative Delta S.

Example: N₂(g) + 3H₂(g) → 2NH₃(g). Here, we start with a total of four gas molecules (one nitrogen and three hydrogen) and end up with only two ammonia gas molecules. These two molecules have less room to roam independently than the original four. The particles are getting more "organized" into pairs, leading to a decrease in entropy. Hence, negative Delta S.
Phase Changes Are the Big Movers
As we discussed, solid to liquid to gas is always an increase in entropy (positive Delta S). Liquid to gas is a bigger jump than solid to liquid. Gas to solid is a huge decrease in entropy (negative Delta S).
Putting It All Together: The Universal Predictor
So, the next time you see a chemical reaction, just ask yourself: is this going to be more of a "let it all hang out" situation, or more of a "clean up your room, young man!" scenario? If it’s the former, you’re probably looking at a positive Delta S. If it’s the latter, it’s likely a negative Delta S.
It’s not about memorizing a million reactions. It’s about understanding the fundamental drive of the universe towards spreading out, towards mingling, towards that beautiful, inevitable mess. And Delta S is our handy little tool for measuring just how much that mingling is happening. So go forth, and predict the mess! It’s more fun than it sounds, and it’s probably happening in your kitchen right now, whether you’re baking, brewing, or just… existing. Because in the grand, messy scheme of things, entropy always wins, and Delta S is just its trusty sidekick, keeping score.
