Predict The Products When Cyclohexanol Is Dehydrated
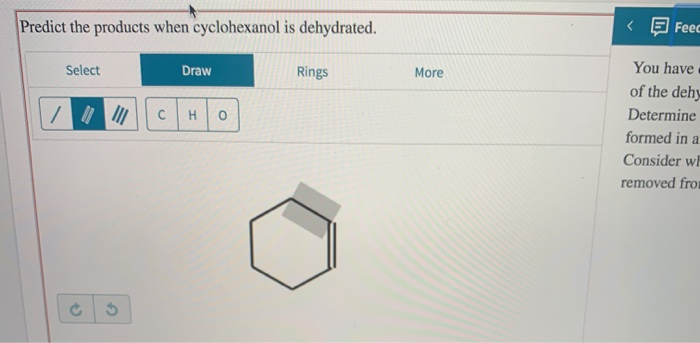
So, you’ve got this cool little molecule called cyclohexanol. Think of it like a tiny, six-sided ring with a party hat on top. That party hat is an -OH group, a little hydroxyl. And this hydroxyl, well, it’s a bit of a drama queen. It’s ready to leave the party. And when it leaves, it likes to take a friend with it.
This whole shindig is called dehydration. It’s like cyclohexanol is going on a diet, shedding a water molecule. And where does this water molecule go? It scoots off, taking a hydrogen from a neighboring carbon atom. Poof! A new double bond is born. It’s like a molecular breakup that leads to a brand new, exciting arrangement. And what’s so exciting about it? Well, that’s where things get interesting, and dare I say, a little bit predictable. But in a fun way!
When you put cyclohexanol through this dehydration process, usually with a little acid helping it along, it’s not just going to spit out one single, solitary product. Oh no. This isn’t a one-hit wonder. It’s more like a band with several chart-topping hits. And the biggest hit, the one everyone knows and sings along to, is cyclohexene. Think of it as the superstar of the dehydration show. It’s a six-sided ring, just like our friend cyclohexanol, but now with a double bond. It’s like the ring got a little more exciting, a little more ready to mingle.
But wait, there's more! While cyclohexene is busy taking all the applause, there are other possibilities. It’s like the opening acts, or the B-sides that are still pretty darn good. Because that hydroxyl group can leave from different sides of the ring, and the hydrogen it snags can also come from different neighbors. This means you can get slight variations on the theme. You might get a double bond right here, or a double bond a little further down the ring. It’s like choosing your favorite flavor of ice cream – there are options!
Now, I have a little, shall we say, unpopular opinion. While scientists might get all technical about this, talking about "major" and "minor" products, and carefully measuring yields, I like to think of it as cyclohexanol having a bit of a mischievous side. It’s not just going to give you one answer. It’s going to give you a whole menu of answers. And you, the intrepid chemist, get to decide which one you want to highlight. Or which one sneaks out the most.

Most of the time, the cyclohexene is the clear winner. It’s the Beyoncé of the reaction. Everyone loves it. It’s the most stable. It’s the most likely to form. But what about the other guys? Let’s call them the supporting cast. They’re still important! They’re still part of the story. They’re just… less famous.
Imagine you’re at a concert. The headliner comes out, everyone goes wild. But then there are those other bands that played earlier. They were great too, right? You enjoyed them. You might even have a favorite song from them. That’s kind of like the other potential dehydration products of cyclohexanol. They’re there. They’re formed. They’re just not the main event.
So, what are these other, less celebrated products? Well, if you get really picky, and if the conditions are just right, you might see some of these. One possibility is that the double bond doesn’t form between two adjacent carbons in the most obvious way. Instead, it might sneak a carbon further away. This can lead to something called a cycloheptene. Yes, a seven-sided ring! It’s like the molecule got so excited about leaving that it completely changed its shape. It's a bit of a wild card. It’s the unexpected encore.
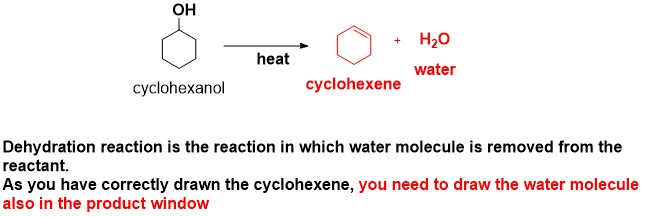
But honestly, most of the time, if you’re just doing a standard dehydration of cyclohexanol, you’re going to get a whole lot of cyclohexene. It’s the predictable one. The reliable one. The one that shows up on time and does its job. It’s the workhorse of the reaction. It’s the sensible choice.
And here’s the truly entertaining part. Even though we know what’s likely to happen, there’s always that tiny, thrilling possibility that something else might surprise you. It’s like baking a cake. You follow the recipe, but sometimes, just sometimes, it turns out a little different. A little more… interesting. A little more you.
So, when cyclohexanol decides to shed its water, it’s not a simple goodbye. It’s a transformation. It’s a chance for new bonds to form. It’s a journey from a simple alcohol to an alkene. And while cyclohexene is usually the star of the show, it’s fun to remember that there are other possibilities, other pathways, other little molecular adventures waiting to happen. It's chemistry, but with a hint of surprise. And who doesn't love a good surprise?
It’s almost like cyclohexanol is saying, "Sure, I'll give you the most obvious answer, but just in case, I’ve got a few other tricks up my sleeve." And that, my friends, is what makes this little dehydration reaction so much fun. It’s predictable, yes. But it also leaves a little room for wonder. A little room for a smile. And maybe, just maybe, a little room for an unpopular opinion that the other products are secretly cooler.
