Predict The Products Of The Following Ether Cleavage Reaction
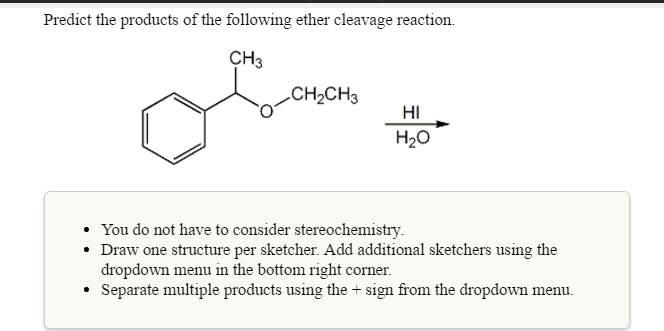
Hey there, science explorers and curious minds! Ever feel like you're watching a magic trick, but instead of rabbits and hats, it's about tiny little molecules doing amazing transformations? Well, get ready, because we're diving into a realm where everyday things get a chemical makeover, and it's surprisingly, wonderfully fun.
Imagine you have a special kind of molecule, something that holds together a bit like two friends holding hands. In the world of chemistry, we call these friends ethers. They're pretty neat, often showing up in things like solvents and even some flavors. But what happens when you decide to break up this little friendship? That's where the real excitement begins!
We're not talking about a sad breakup here, oh no. This is more like a friendly parting of ways, guided by a clever chemist with a special tool. Think of it like this: you have a candy bar with two delicious halves stuck together. A chemist comes along with a super-duper candy-splitter. They don't just break it randomly, though. They have a specific way of doing it, and that's what makes predicting the outcome so fascinating.
The star of our show today is a reaction called ether cleavage. It sounds a bit serious, doesn't it? Like something out of a dramatic movie. But trust me, it's more like a really well-choreographed dance. A molecule that looks a little like R-O-R' (where the 'R's are like different flavored gummy bears) gets an invitation to a party. And at this party, there's a special reagent, let's call it the 'Party Starter'.
This 'Party Starter' is usually something super reactive, like a strong acid. Think of it as the ultimate icebreaker. When it meets our ether, it doesn't just barge in. It politely nudges one of the 'friends' (the R groups) and whispers, "Hey, it's time for a change!"

Now, here's the cool part, the part that makes predicting the products so entertaining. The 'Party Starter' doesn't just pick a friend at random to break up with. It's a bit of a strategist. It looks at the two 'R' groups and makes a decision based on what's easiest or most stable for them to become once they're on their own.
"It's like guessing who will win a dance-off based on their style!"
So, what are these potential products? Well, once our ether friend R-O-R' is coaxed into separating, you're almost always guaranteed to get a couple of new pals. One of them will likely be an alcohol. You know, like the kind found in hand sanitizer or even your favorite beverage (though we're talking about the pure chemical kind here!). Alcohols have a structure that looks like R-OH. So, one of our gummy bear friends is now happily hanging out with an 'OH' group.
And what about the other friend? The remaining part of the ether molecule becomes something else entirely. Depending on the original 'R' groups and the strength of our 'Party Starter', it could become another alcohol, or it might become something a little different. The most common partner is often an alkyl halide, especially if a strong acid like hydrobromic acid (HBr) or hydroiodic acid (HI) is involved. This looks like R'-X, where 'X' is a halogen, like bromine or iodine.
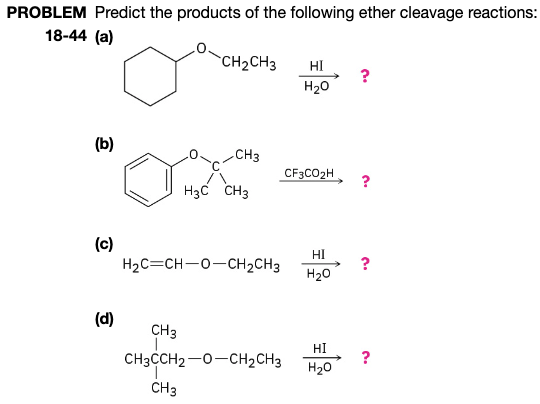
But here's where the fun really kicks in: predicting which R group goes with the OH and which R group becomes the alkyl halide. It’s not a coin toss! There are rules, and learning them feels like cracking a secret code. It’s all about stability. Some 'R' groups are happier on their own, or they're easier to kick out of the ether partnership. Chemists call these more stable or reactive groups.
Think about it: if one of your friends is really good at making new connections easily, they're likely to be the one that forms the new bond. In ether cleavage, the 'R' group that can form a more stable intermediate, like a carbocation (a positively charged carbon atom), is often the one that gets "chopped off" first by the acid and ends up as the alkyl halide. The other R group, often the one that's more resistant to forming that stable intermediate, tends to grab the 'OH' and become the alcohol.
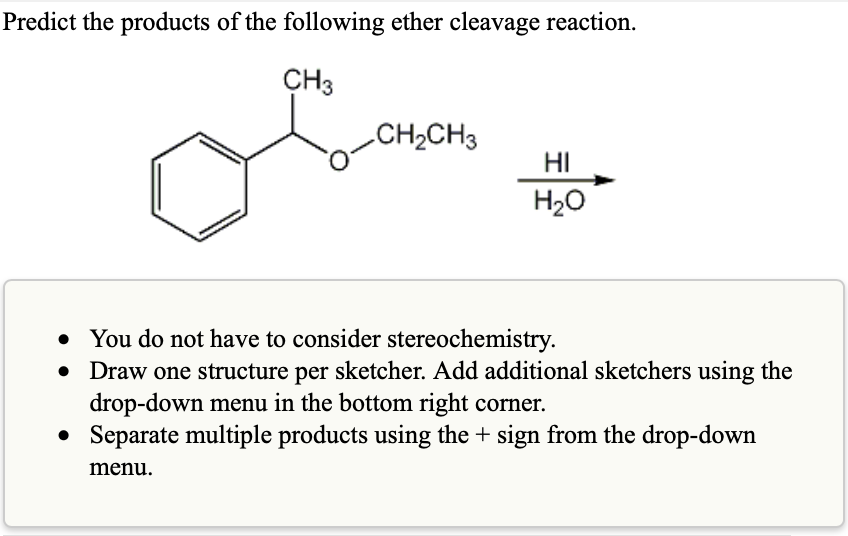
So, when you're presented with an ether molecule and told to predict the products of cleavage, it’s like being given a puzzle. You look at the two 'R' groups. You think about their personalities, their strengths. Are they simple like a plain gummy bear (a primary alkyl)? Or are they more complex and branching, like a fancy shaped gummy bear (a secondary or tertiary alkyl)? The more complex and branched they are, the more likely they are to be the ones that break free first and become the alkyl halide.
It’s this little bit of strategic thinking, this understanding of molecular behavior, that makes predicting the products of ether cleavage so satisfying. It’s not just memorization; it’s about applying logic and seeing the elegant patterns in chemistry.
So, next time you hear about ether cleavage, don't picture a boring lab experiment. Picture a molecular dance, a friendly parting of ways, and a chemical puzzle waiting to be solved. It’s a peek into the hidden world of molecules, where simple reactions can lead to predictable, and often surprisingly stable, new partners. Give it a try, and you might just find yourself enjoying the show!
