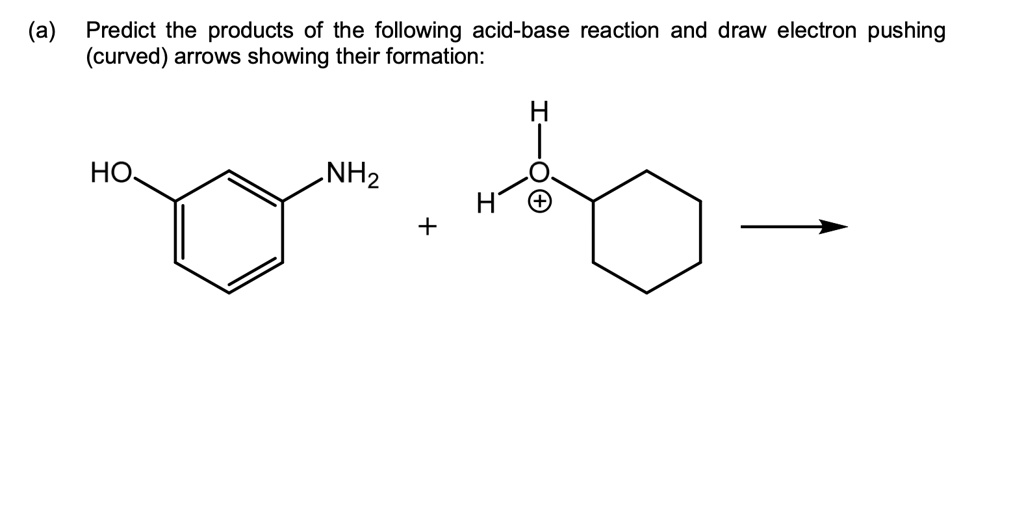
Predict The Products Of The Following Acid-base Reaction.
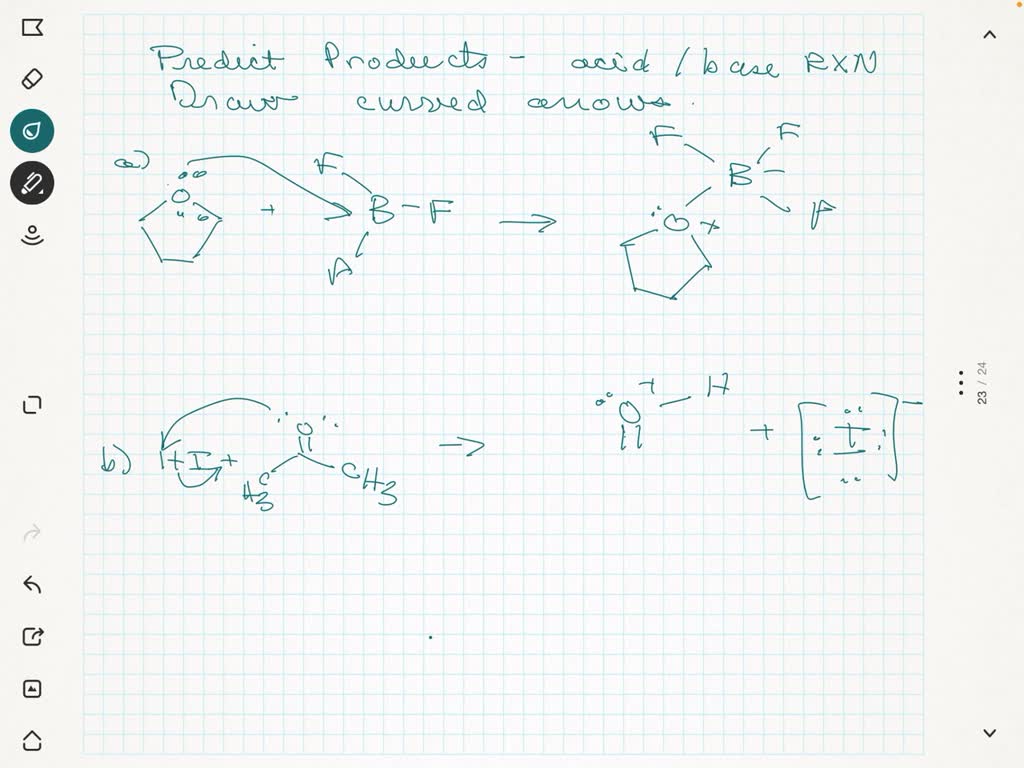
Ever wondered what happens when you mix certain kitchen ingredients, or why antacids work their magic? It all comes down to the fascinating world of acid-base reactions! These reactions are a fundamental concept in chemistry, but don't let that "chemistry" word scare you. Think of it like a playful dance between two types of molecules: acids and bases. They meet, they greet, and something new and often very useful is created. It's a bit like a chemical matchmaking service, where the "happily ever after" is a predictable outcome. This predictability is what makes predicting acid-base reaction products so incredibly satisfying and, dare we say, fun!
Why is this so cool? Well, understanding these reactions is like having a secret decoder ring for the chemical world around you. From the way your stomach digests food to the creation of your favorite cleaning products, acid-base reactions are at play. Being able to predict the results of these interactions is not just a neat party trick; it's a powerful skill that opens doors to understanding and even controlling chemical processes.
The purpose of predicting acid-base reaction products is to understand what chemical transformations will occur when an acid and a base are combined. When we talk about acids, we often think of things that taste sour, like lemon juice (which contains citric acid) or vinegar (which contains acetic acid). They tend to release protons (a type of positively charged particle) when dissolved in water. Bases, on the other hand, often feel slippery, like soap, and taste bitter. They tend to accept protons or release hydroxide ions (a type of negatively charged particle) in water. The magic happens when an acid and a base meet – they essentially neutralize each other.
The benefits of mastering this skill are numerous. For students, it’s a cornerstone of chemistry education, helping them grasp more complex concepts. For aspiring scientists, it's a vital tool for designing experiments and synthesizing new materials. Even for everyday folks, it helps demystify common household reactions. Think about it: you've got a spill that needs neutralizing, or you want to understand why baking soda and vinegar create that fizzy eruption. Knowing the principles of acid-base reactions gives you that insight.
So, how do we predict these products? It’s all about the transfer of a proton. The acid is the "giver" of the proton, and the base is the "taker." When they react, the proton moves from the acid to the base. This usually results in the formation of a salt and water. Water is formed when the proton from the acid combines with the hydroxide ion (or a similar species) from the base. A salt is an ionic compound, typically formed from the remaining parts of the acid and base molecules after the proton has been exchanged.

Let's take a classic example that's easy to visualize. Imagine we have hydrochloric acid (HCl), a strong acid, and sodium hydroxide (NaOH), a strong base. In this scenario:
Hydrochloric acid (HCl) is like the energetic friend who's eager to give away a proton (H+).
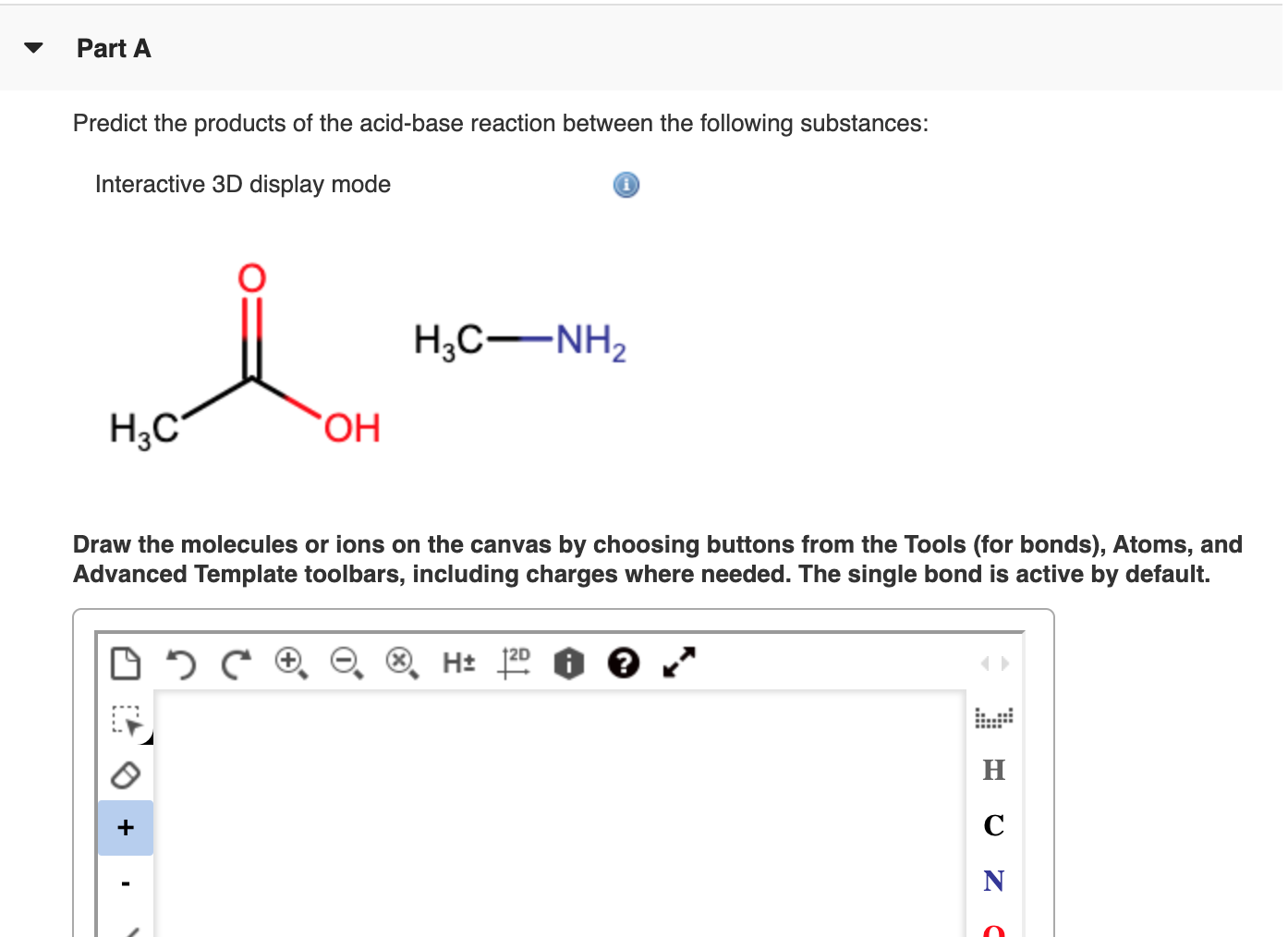
SOLVED: (a) Predict the products of the following acid-base reactionSodium hydroxide (NaOH) is like the welcoming friend ready to receive that proton, and it also has a hydroxide group (OH-).
When these two meet:
- The proton (H+) from HCl moves over to the hydroxide ion (OH-) from NaOH.
- Together, the H+ and OH- form a molecule of water (H2O).
- What's left of the HCl is the chloride ion (Cl-).
- What's left of the NaOH is the sodium ion (Na+).
These leftover ions, the sodium ion (Na+) and the chloride ion (Cl-), are attracted to each other because of their opposite charges. They come together to form an ionic compound called sodium chloride (NaCl), which is, you guessed it, common table salt!
So, the reaction between hydrochloric acid and sodium hydroxide produces sodium chloride and water. This general pattern – acid + base → salt + water – is a fantastic starting point for predicting many acid-base reactions. It’s like a fundamental rule of chemical interaction!
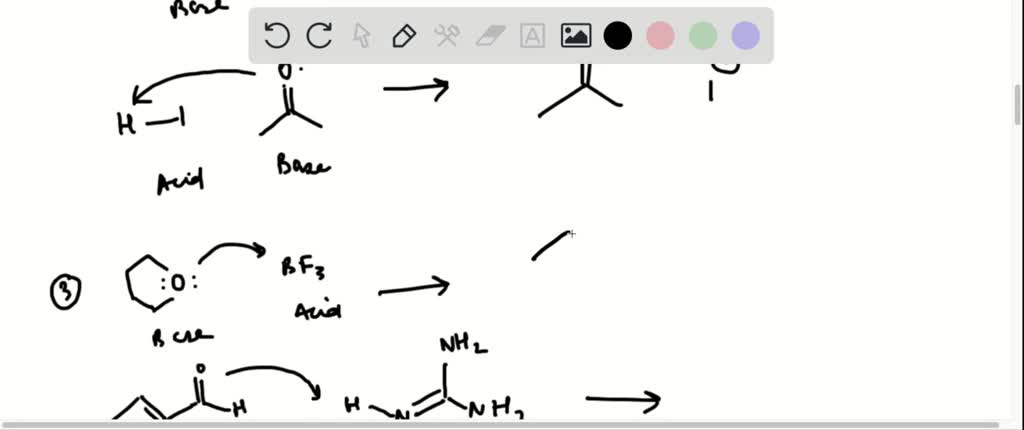
Another common scenario involves acids reacting with carbonates, like baking soda (sodium bicarbonate, NaHCO3) or washing soda (sodium carbonate, Na2CO3). When an acid, like our friend acetic acid (CH3COOH) from vinegar, meets a carbonate, things get a little more exciting. You get the salt and water, but also a third product: carbon dioxide gas (CO2). This is the gas responsible for the fizzing and bubbling you see when you mix vinegar and baking soda!
Predicting these products isn't just about memorizing equations; it's about understanding the why. It's about recognizing the proton-donating and proton-accepting tendencies of different substances. As you become more familiar with common acids and bases, you'll start to see patterns emerge. You'll learn to identify the "acidic part" and the "basic part" of molecules and how they'll rearrange themselves after the proton transfer.
The beauty of predicting acid-base reactions lies in their relative simplicity once you grasp the core concept of proton transfer. It's a gateway to understanding more complex chemical processes and appreciating the subtle yet powerful transformations happening all around us, from the lab to our kitchens. So, next time you see something fizz or bubble, you'll have a better idea of the exciting chemical dance that’s taking place!