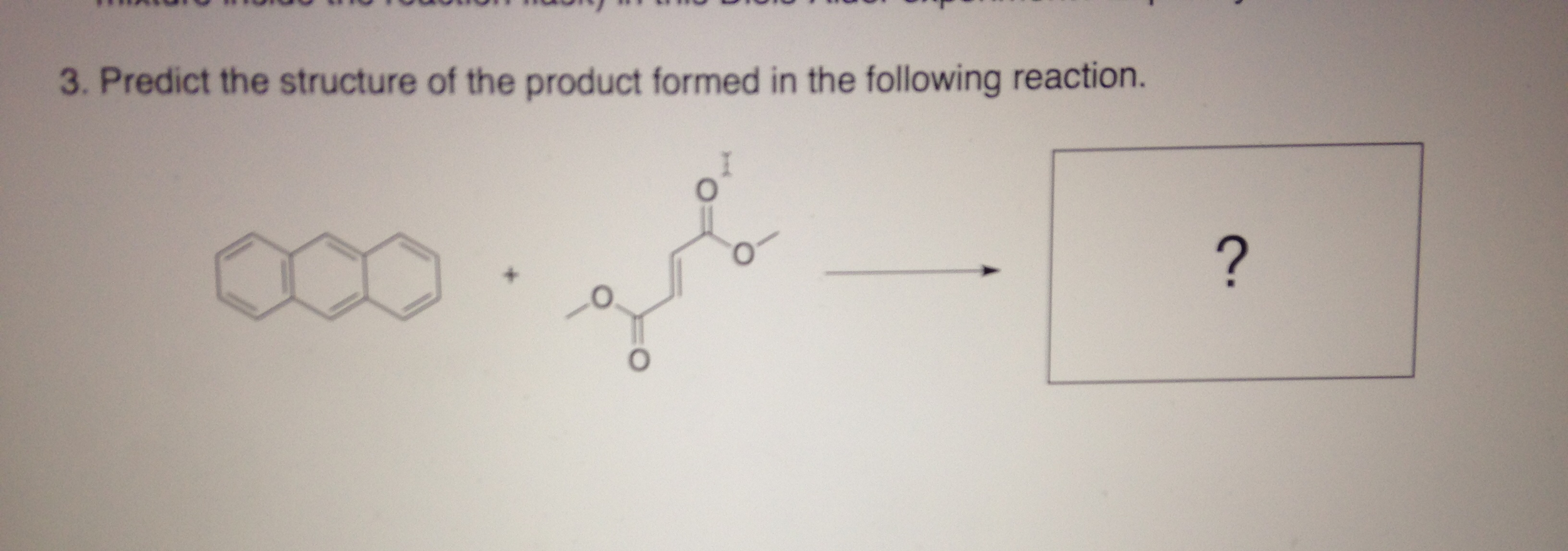
Predict The Products Formed In The Following Reactions.
Ever found yourself staring at a list of chemical reactions, feeling a little like you've stumbled into a secret code? You know, the kind where letters and numbers dance around, promising to reveal something new? Well, what if I told you that predicting the products of these reactions isn't some arcane wizardry reserved for lab coats and safety goggles, but rather a kind of intuitive art? Think of it less like complex math and more like putting together a killer playlist or assembling IKEA furniture – with a little understanding of the basic components and a dash of creative foresight, you can actually predict the outcome. Let's dive into the wonderfully chill world of chemical predictions, shall we?
Forget the sterile lab environment for a moment. Imagine yourself on a breezy patio, maybe with a cold brew in hand, surrounded by good company. That’s the vibe we’re going for here. Chemical reactions are just atoms and molecules having a party, rearranging themselves to form new, exciting combinations. Our job, as amateur chemists or simply curious minds, is to guess who’s going to end up dancing with whom.
The "Meet-Cute" of Molecules: Predicting Simple Reactions
Let’s start with the absolute basics, the molecular equivalent of a "meet-cute." We’re talking about reactions where things are pretty straightforward. For instance, consider a reaction where a metal reacts with oxygen. Think of iron rusting. That beautiful (though sometimes annoying) orange-brown coating? That's iron oxide, the result of iron atoms getting super friendly with oxygen molecules. The prediction here is simple: metal + oxygen → metal oxide. Easy peasy, right? It’s like knowing that if you put two people who really like each other in the same room, they’re probably going to hit it off.
Another classic is the reaction between an acid and a base. This is the ultimate chemical bromance (or sisterhood!). Acids are like the energetic, slightly wild ones, and bases are the calm, grounding forces. When they meet, they neutralize each other, forming salt and water. Think of it like your friend who’s always on the go meeting their chill roommate. The result? A beautiful, balanced harmony. So, acid + base → salt + water. This is why antacids work – they’re bases neutralizing the excess acid in your stomach. Pretty cool, huh? It’s the science behind feeling better after that spicy meal!
Fun Fact: The "salt" formed in these reactions isn't always table salt (sodium chloride). It depends on the specific acid and base. It could be anything from potassium chloride to magnesium sulfate. Variety is the spice of life, even in chemistry!
The "Breaking Up and Making Up" Scenario: Double Displacement Reactions
Now, let's move to the slightly more dramatic, but still predictable, world of double displacement reactions. Imagine two couples at a party. Suddenly, there's a bit of mingling, some quick exchanges, and… bam! The partners have swapped. That’s essentially what happens here. We have two compounds, let’s call them AB and CD. In a double displacement reaction, A might pair up with D, and C might pair up with B. So, AB + CD → AD + CB.
Why do they swap? Usually, it's because one of the new pairings is more stable, or it forms something insoluble (a precipitate) that drops out of the solution, or it produces a gas that bubbles away, or it forms water. These are the "driving forces" that make the reaction happen. It's like a couple deciding to move in together because they just click better. You can often predict these reactions by looking at the solubility rules – basically, a cheat sheet for which ionic compounds dissolve in water and which ones prefer to stick together and form a solid.
Practical Tip: When you see two aqueous solutions (meaning they're dissolved in water) react, immediately think "double displacement!" It’s your most likely scenario. Look for the cations (positive ions) and anions (negative ions) and see which new combinations they can form. Are any of them listed as "insoluble" on your handy solubility chart? Bingo! That's your precipitate.
Think about when you mix two clear solutions of different salts and suddenly a cloudy solid forms. That’s a double displacement reaction in action, and the cloudiness is your solid precipitate – the happy (or maybe not so happy) result of the partner swap.
The "Passionate Embrace" or "Dramatic Breakup": Single Displacement Reactions
Moving on, we have single displacement reactions. These are a bit like one element being a little too popular. Imagine element A is feeling lonely, and it meets a compound BC. If A is "more reactive" than B, it will push B out of the compound and take its place, forming AC and leaving B all by itself. So, A + BC → AC + B.
Alternatively, if it's a non-metal like X reacting with a compound YZ, and X is more reactive than Z, then X will displace Z: X + YZ → YX + Z. The key here is reactivity. It's like a popularity contest among elements. The more popular (reactive) element gets the partner. You can predict these by looking at an activity series, which is basically a ranked list of elements from most reactive to least reactive. If the element you're adding is higher on the list than the one it's trying to displace, the reaction will happen.
Cultural Reference: Think of it like a dance competition. The most skilled dancer (most reactive element) can easily "steal" a partner from a less skilled dancer. The less skilled dancer is then left on the sidelines.
Fun Fact: The activity series is usually determined experimentally. Scientists have done the hard work of figuring out who's who in the elemental popularity contest!
So, if you see a clean metal piece added to a solution of a metal salt, and the metal you added is more reactive than the metal in the salt, you’ll likely see the more reactive metal deposit itself onto your piece. It’s like the more popular metal is saying, "Hey, I can do better!"
The "Combustion Carnival": When Things Get Fiery
Ah, combustion! This is where things get exciting, fiery, and usually produce a lot of heat and light. The most common type is the combustion of a hydrocarbon (a compound made of hydrogen and carbon, like methane or propane) with oxygen. Think of burning natural gas or a campfire. When these burn completely, the products are always the same: carbon dioxide (CO2) and water (H2O).
So, hydrocarbon + O2 → CO2 + H2O. It’s a reliable party trick of chemistry. The oxygen is the fuel for the fire, and the hydrocarbon is what burns. If there's not enough oxygen (incomplete combustion), you might also get carbon monoxide (CO), which is less fun and, well, poisonous. But for complete combustion, it’s carbon dioxide and water, every time.
Practical Tip: If you see a hydrocarbon reacting with oxygen, especially with a spark or flame involved, immediately jump to CO2 and H2O as your predicted products. This is the basis of how engines work and how we generate a lot of our energy.
Cultural Reference: Think of the classic "caveman discovering fire" trope. It’s the fundamental reaction that allowed humans to cook, stay warm, and develop civilization. A little bit of oxygen and a bit of fuel – and voilà, progress!
It's like baking a cake. You know the ingredients (hydrocarbon and oxygen), and you know the outcome (CO2 and H2O). It's a predictable transformation.
Decomposition: When Things Fall Apart (Nicely)
Sometimes, reactions are about breaking things down. These are called decomposition reactions. It's the opposite of synthesis (where things come together). Think of a complicated molecule that decides it's had enough and breaks into simpler components. For example, when you heat certain metal carbonates, they decompose into a metal oxide and carbon dioxide. Like calcium carbonate (limestone) heating up to form calcium oxide and carbon dioxide. So, AB → A + B.
The trick here is to know what kinds of bonds are weak or what compounds are prone to breaking down under certain conditions (like heat, light, or electricity). It’s like knowing which friendships are a bit fragile and might not last under pressure.
Fun Fact: Electrolysis, the process of using electricity to break down compounds (like water into hydrogen and oxygen), is a form of decomposition reaction.
Practical Tip: Look for single compounds on the reactant side that are being heated or exposed to electricity or light. Then, think about the simplest, most stable components that compound could break into. For instance, binary compounds (two elements) often break back into their constituent elements.
Synthesis: Building It Up
And finally, let's talk about synthesis reactions. This is where two or more simpler substances combine to form a more complex substance. It’s the inverse of decomposition. Think of simple elements coming together to form a compound. For example, hydrogen gas reacting with oxygen gas to form water. A + B → AB. It's like LEGO bricks clicking together to build something bigger.
Predicting these often involves recognizing common synthesis patterns. For instance, many non-metal oxides react with water to form acids, and metal oxides react with water to form bases. It’s about seeing the building blocks and knowing which ones fit together nicely.
Cultural Reference: Think of early alchemists trying to create gold. While their goals were often misguided, the underlying principle of combining substances to create something new is synthesis. Modern synthesis in pharmaceuticals and materials science is the evolution of that ancient curiosity.
Practical Tip: If you see two or more simple substances on the reactant side, especially elements or simple compounds, and no obvious indicators of other reaction types, synthesis is a strong possibility. Consider what stable compounds they might form together.
The Big Picture: It's All About Patterns
So, how do you get good at predicting these products? It’s not about memorizing every single reaction. It’s about recognizing the patterns. Most reactions fall into a few major categories, and within those categories, there are predictable outcomes. It’s like learning the basic chords in guitar – once you know them, you can play a thousand songs.
Start by identifying the types of reactants you have. Are they a metal and an acid? An acid and a base? A hydrocarbon and oxygen? Two ionic compounds? A single compound being heated? Once you’ve classified the players, you can often deduce the likely outcome based on established chemical principles and common reaction types.
Think of it as a conversation. The reactants are talking to each other, and their "personalities" (their chemical properties) dictate how they'll interact. Are they going to combine, swap partners, or break apart? The clues are usually right there in what you're given.
Final Reflection: In our daily lives, we're constantly predicting outcomes. Will that business meeting be productive? Will this new recipe turn out well? Will my friend be happy if I surprise them? We make these predictions based on past experiences, understanding of people and processes, and an intuitive sense of how things work. Chemistry is no different. By understanding the basic "personalities" and interaction styles of elements and compounds, we can start to predict the beautiful, sometimes surprising, and always fascinating ways they'll combine and transform. It’s a reminder that even in the seemingly complex world of science, there’s an underlying order and a predictable rhythm, much like the ebb and flow of our own lives.
