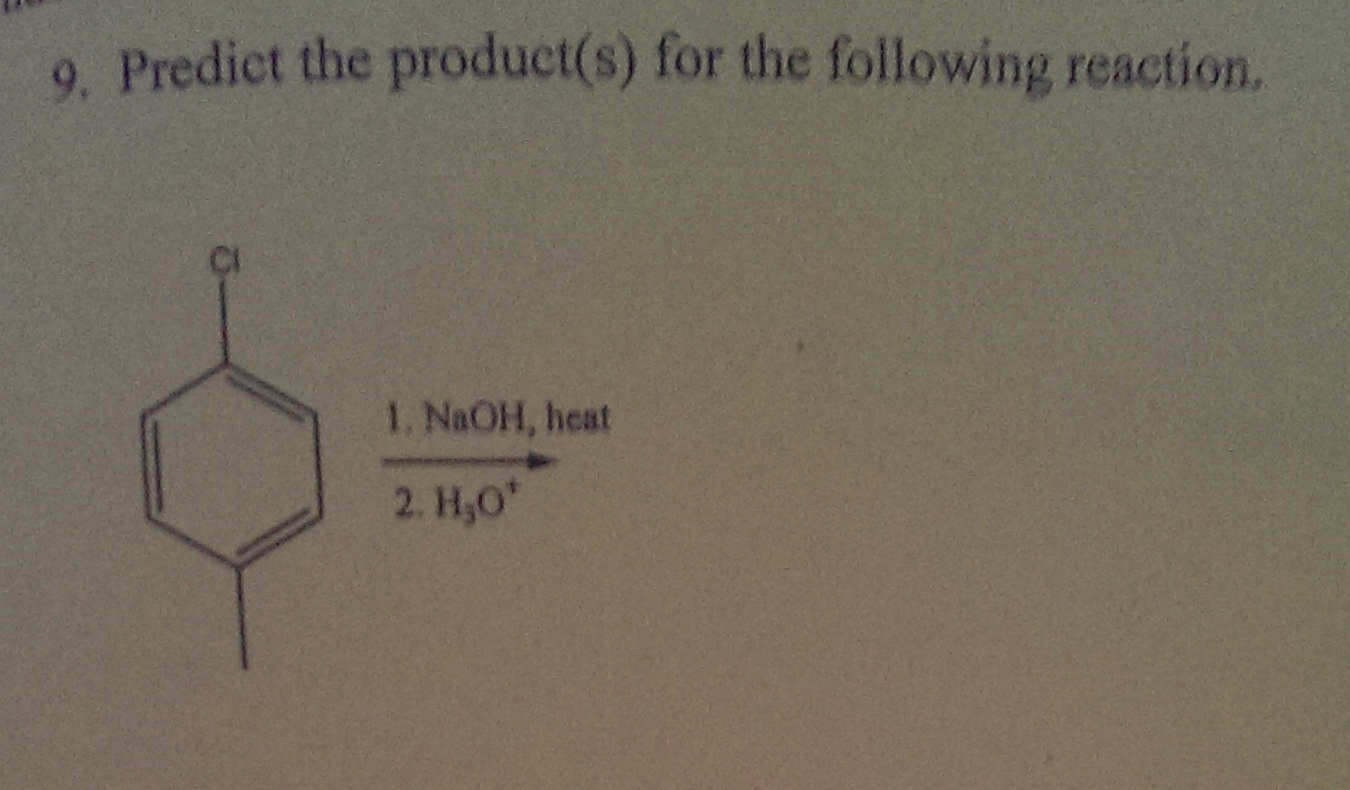
Predict The Product Of The Following Sn2 Reaction

Imagine a tiny, bustling chemical party happening right under our noses! It's not the kind of party with loud music and disco balls, but more like a secret handshake among microscopic friends. Today, we're going to peek at one of these delightful shindigs and guess who the star guest will be at the end.
Our main characters are called nucleophiles and substrates. Think of the nucleophile as the energetic dancer, always eager to join the fun. The substrate is like the dance floor, a bit more reserved but ready to host the celebration.
Now, this particular dance move has a special name: the SN2 reaction. It's like a perfectly choreographed two-step. The nucleophile doesn't hesitate; it sees an opportunity and goes for it!
On the dance floor (our substrate), there's a special spot that the nucleophile is really interested in. It's like a prime piece of real estate, just waiting for the right partner. And our nucleophile is definitely the right partner!
The really cool thing about the SN2 reaction is that it's a bit of a surprise party for the leaving group. You see, as the nucleophile waltzes in, it gently nudges someone else right off the dance floor. This is the leaving group, and it's like a guest who gets the memo to leave just as the main attraction arrives.
It’s not a violent eviction, mind you. It’s more of a polite, “Excuse me, but there’s a new star in town!” The leaving group gracefully exits, making way for the new arrival.
Let's give our nucleophile a name. How about Amelia the Amine? Amelia is super friendly and has a little extra spark of energy (we call this a lone pair of electrons). She's just the type to brighten up any room.
And our substrate, the dance floor? Let's imagine it's a molecule called methyl bromide. It has a carbon atom in the center, and attached to it are three little hydrogen atoms and one bromine atom. The bromine atom is a bit of a drag, always wanting to leave. It’s like that friend who’s always looking for an excuse to bail.

So, Amelia the Amine spots the methyl bromide. She sees that carbon atom looking a little lonely, and she knows that bromine is just waiting for a chance to escape. It’s a perfect match!
Amelia, with her energetic spirit, approaches the carbon atom from the opposite side of where the bromine is sitting. It's like sneaking up from behind for a friendly hug. This is a key move in the SN2 dance – it’s a backside attack.
As Amelia gets closer and closer, the carbon atom starts to feel a bit of a squeeze. It’s like when you try to hold hands with two people at the same time; it gets a little awkward!
In this awkward in-between moment, the carbon is temporarily holding onto both Amelia and the bromine. It's a fleeting moment of chemical juggling. This is called the transition state, and it’s the peak of the SN2 performance.
Then, just as Amelia makes her final connection, the bromine atom decides it's had enough. "Adios!" it says, and floats away, leaving the carbon atom free.
And what do we have left on the dance floor? Well, it's our original carbon atom, but now it's proudly bonded to Amelia the Amine! The three little hydrogen atoms are still there, but their positions have subtly shifted. It's like they did a little spin as Amelia joined the party.
This is where the magic happens. The SN2 reaction has a neat trick: it causes something called inversion of configuration. Imagine you have a beautiful, delicate flower. If you were to flip it inside out, that's kind of what happens to the arrangement of atoms around the carbon.
So, if our methyl bromide had a specific three-dimensional shape, the final product, with Amelia attached, will have that shape flipped. It’s like looking in a mirror, but for molecules!
Let's think about a simpler analogy. Imagine you're holding a glove. The opening where your hand goes in is like the leaving group, the bromine. The rest of the glove is like the carbon and its attachments.
When you push your hand (Amelia) into the glove, you push the opening out the other side. The glove stays the same, but the "opening" has moved. In our chemical world, the carbon atom is like the glove, and the attachment points are where the "opening" (bromine) was.

The final product is a brand-new molecule where Amelia the Amine is now happily attached to the carbon. It’s a successful pairing, all thanks to the SN2 dance.
We started with methyl bromide and Amelia the Amine. We witnessed the SN2 reaction, a swift and direct exchange.
And the product? It’s a molecule where Amelia has replaced bromine! We can call it N-methylamine. It’s a simple molecule, but a testament to the power of these tiny chemical interactions.
This process is happening all around us, all the time, in our bodies and in nature. It's the unseen engine that keeps things going.
So, next time you think about chemistry, remember these little parties and dances. They might seem small, but they’re building blocks of everything we see and experience.

The SN2 reaction is a beautiful example of how molecules can interact in a predictable, yet wonderfully dynamic way. It's like a perfectly timed waltz, where one partner steps in as another gracefully exits.
And the outcome? A new, often more stable or useful molecule, ready to play its part in the grand chemical symphony.
It’s a reminder that even in the most minuscule of worlds, there’s order, intention, and a touch of delightful surprise.
So, we predict that the product of this particular SN2 reaction will be our happy couple: N-methylamine, with Amelia the Amine now securely bonded to the carbon, and the bromide ion having made its graceful exit.
It's a successful chemical union, all thanks to the efficient and elegant SN2 mechanism!
Who knew such a tiny dance could create such significant results? It's a heartwarming thought, indeed!
