Predict The Product Of Each Monosaccharide Oxidation Reaction.

Hey there, science enthusiasts and curious cats! Ever wondered what happens when you give those sweet little monosaccharides a bit of a jolt? We're talking about the ultimate sugar makeover. Get ready for some fun chemistry. It’s not as scary as it sounds. In fact, it's pretty darn cool.
Monosaccharides. They're the building blocks of all things sweet and starchy. Think glucose, fructose, galactose. The usual suspects. These are the single, simple sugars. The VIPs of the carbohydrate world. They're what your body loves. They're what makes fruit, well, fruity. And candy, deliciously candied.
Now, imagine these sugars having a bit of an identity crisis. They’re chilling, being all sugary. Then, BAM! Oxidation happens. It’s like they’re getting a superhero power-up. Or maybe a mild electrical shock. Either way, things change. And we're here to predict what they become. It’s a bit like a chemistry guessing game. But with real, tangible results. Pretty neat, right?
The Mighty Aldehyde Group: Glucose's Big Moment
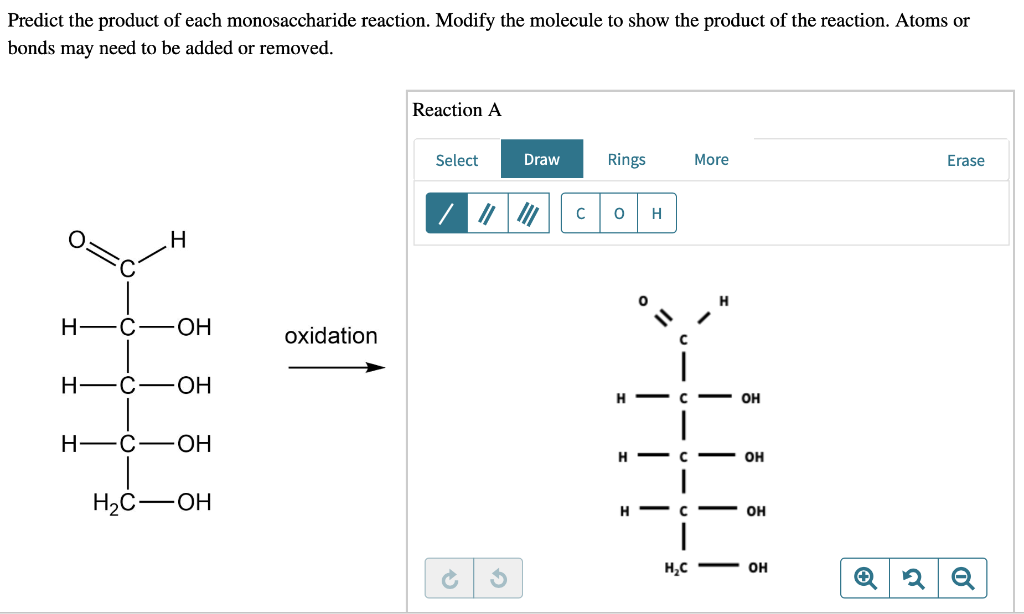
Let's start with our star player: glucose. Glucose is an aldose. That means it has an aldehyde group. Picture a little “CHO” hanging out at one end. This aldehyde group is the prime target. It’s like the weak spot. The first to get tweaked.
When you mildly oxidize glucose, something amazing happens. That aldehyde group gets a little makeover. It turns into a carboxylic acid group. Think “COOH”. It’s like the sugar grew a tiny, tangy tail. And what do we call this new, slightly sour creature? Gluconic acid. Ta-da!
Gluconic acid is actually a thing! It's found naturally. It's used in food. It gives certain vinegars their zing. And it’s a gentle chelating agent. Fancy word, huh? It means it can grab onto metal ions. Useful stuff. So, glucose, the sweet guy, becomes slightly tart and handy. Who knew?

Now, what if you go a bit further? What if you really push the oxidation? Like, full-on superhero intensity. For glucose, that means attacking another part of the molecule. The primary alcohol at the other end of the sugar chain. That gets oxidized too. Into… you guessed it, another carboxylic acid group.
This dual oxidation is pretty intense. It breaks down the sugar quite a bit. The product here is called saccharic acid. Or, if you want to be super fancy, glucaric acid. It's a dicarboxylic acid. Two tangy tails! This stuff is stronger. More acidic. It’s like the supervillain version of gluconic acid. But still useful. It’s used in detergents and as a cleaning agent. Because sometimes, the sweetest things can clean your grubbiest messes.
Fructose: The Sweet Ketone with a Twist
Next up, the one and only fructose. Fructose is a ketose. Instead of an aldehyde group, it has a ketone group. Picture a “C=O” smack dab in the middle of the sugar chain. This makes fructose a bit different. And its oxidation story is also unique.
Mild oxidation on fructose is… complicated. Because that ketone group isn't as easily oxidized as an aldehyde. It's a bit more stubborn. So, mild oxidation on fructose doesn't produce a single, neat product like gluconic acid from glucose. Instead, it tends to break the molecule apart.

Think of it like a slightly less organized reaction. The fructose molecule might snap. It breaks into smaller pieces. These pieces are often short-chain organic acids. Like glyceric acid, formic acid, or glycolic acid. It’s a bit of a chaotic but predictable mess. The exact mix depends on the specific conditions. But the idea is fragmentation. Fructose, the simple, sweet fruit sugar, can get broken down. It’s like a sugar explosion. Controlled, of course.
If you go for stronger oxidation on fructose, it's even more dramatic. Similar to glucose, the alcohol groups can get oxidized. But the initial fragmentation is still a major player. The end result is a complex mixture of shorter organic acids. It’s the ultimate sugar deconstruction. Fructose goes from sweet to… very, very broken down. Not ideal for a snack. But fascinating chemistry!
Why is this fun? Because it shows how the structure of a molecule dictates its reactions. Glucose, with its aldehyde, has a predictable path. Fructose, with its ketone, has a more fragmented fate. It’s like playing with different Lego sets. Same basic bricks, but totally different creations.
Galactose: Another Aldehyde Adventure
Let's not forget galactose. Galactose is another aldose. Just like glucose! So, you might expect a similar story. And you'd be mostly right.

When galactose undergoes mild oxidation, its aldehyde group transforms into a carboxylic acid. What do you think that makes? You guessed it: galactonic acid. Pretty straightforward. It's the galactose version of gluconic acid. Same principle, different starting sugar.
Galactonic acid also has its uses. It's involved in biological processes. And it's studied for its potential health benefits. So, this sugar oxidation thing isn't just about making acids. It's about creating compounds with real-world implications. Cool, huh?
And if you push the oxidation on galactose, similar to glucose, you can get that di-carboxylic acid. The equivalent of saccharic acid. It'll be the oxidized form of the entire galactose chain. Again, strong acids. Used for industrial purposes. Proving that even our basic sugars can be transformed into potent chemical tools.
The "Why Bother?" Section: It's All About Function!
So, why do we care about oxidizing these simple sugars? Well, beyond the sheer joy of understanding chemical transformations, it’s actually super important.
In biology, enzymes do these kinds of oxidations all the time. They're crucial for metabolism. For energy production. For building and breaking down molecules. Understanding these reactions helps us understand life itself. Seriously!
In industry, these oxidized sugars have tons of applications. Think food additives. Pharmaceuticals. Cleaning products. Even biodegradable plastics. These simple sugars, with a little chemical nudge, become versatile ingredients for a modern world.
And for us geeks? It's a beautiful illustration of chemical principles. The reactivity of functional groups. How subtle structural differences lead to vastly different outcomes. It’s a puzzle. A fascinating, sweet-smelling puzzle.
So next time you enjoy a sweet treat, spare a thought for the potential chemical transformations happening. The aldehyde groups waiting to become acids. The ketones ready to fragment. It’s a wild world in there. A world of sugar science. And it’s way more fun than you might think. Keep exploring. Keep questioning. And keep that curiosity buzzing!
