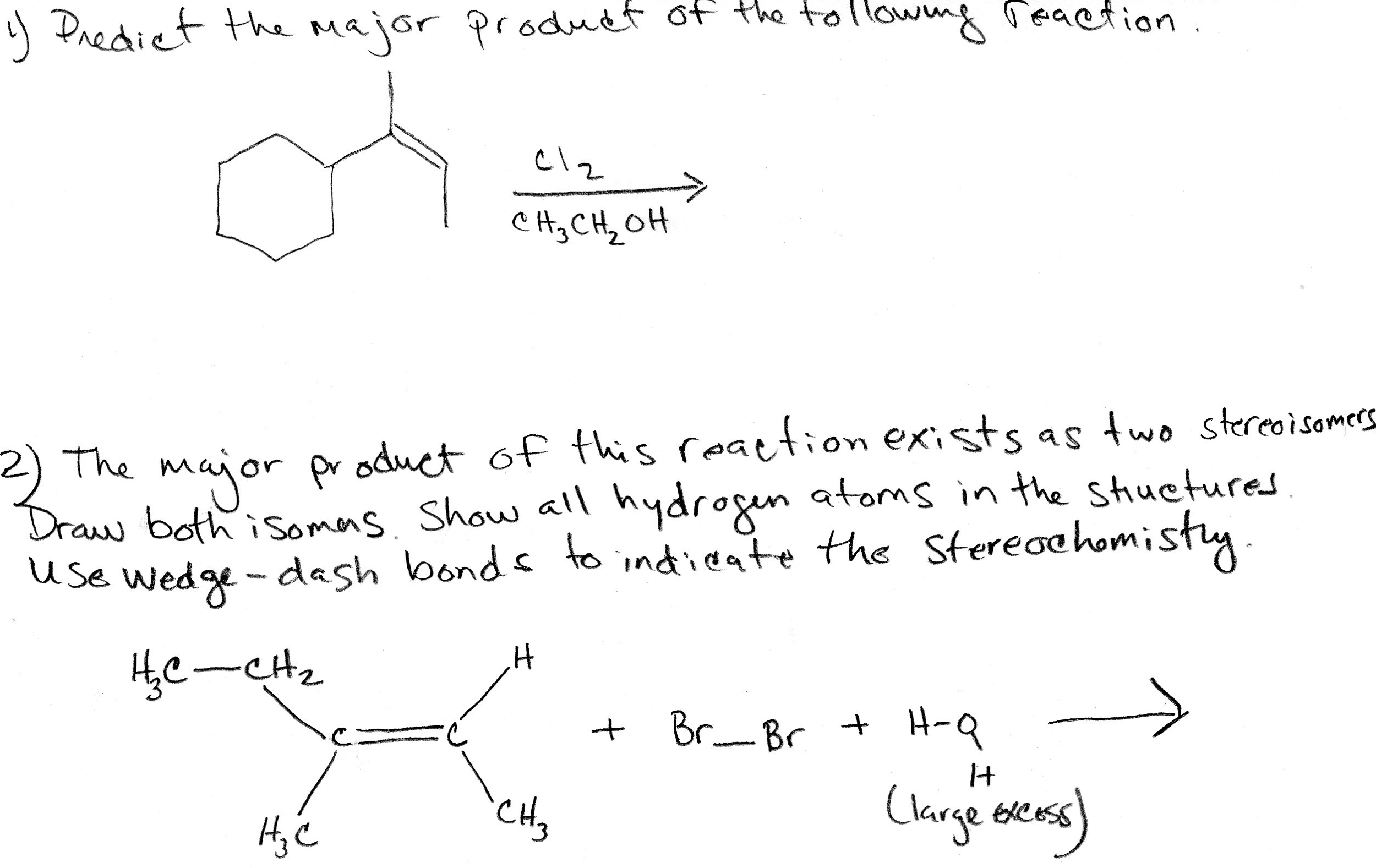
Predict The Product For The Sn2 Reaction Shown.
Ever feel like you're playing a guessing game in chemistry class? Well, get ready to level up your skills because we're diving into something super cool and incredibly useful: predicting the product of an SN2 reaction! Think of it like being a detective, but instead of solving mysteries, you're figuring out what new molecule will be cooked up. It's not just for super-chemists; understanding these reactions is a foundational skill that opens up a whole world of chemical possibilities. Whether you're a student wanting to ace your exams, a budding researcher, or just someone curious about how things are made, mastering this is your secret weapon.
Why is Predicting SN2 Products So Awesome?
So, why should you get excited about SN2 reactions? Because they're everywhere! From the way your body processes food to how pharmaceuticals are made, SN2 reactions are fundamental building blocks in organic chemistry. Being able to predict the outcome of these reactions means you can:
- Design New Molecules: Imagine creating new flavors for your favorite snacks or developing life-saving medicines. SN2 predictions are crucial for chemists designing new compounds with specific properties.
- Understand Existing Processes: Ever wondered how plastics are formed or how your dish soap works? Many industrial and biological processes rely on SN2 mechanisms.
- Troubleshoot Experiments: If an experiment doesn't go as planned, knowing the expected SN2 product helps pinpoint where things might have gone wrong.
- Save Time and Resources: Instead of trial and error, predicting the product allows for more efficient planning and execution of chemical synthesis.
The SN2 Showdown: What's Going On?
Let's break down what an SN2 reaction is all about. The name itself gives us a clue! 'SN' stands for Substitution Nucleophilic, and the '2' refers to the fact that two molecules are involved in the rate-determining step (the slowest step that dictates how fast the reaction happens). It's a one-step dance where a nucleophile (an electron-rich species that loves to donate electrons) attacks a substrate (the molecule being attacked), simultaneously displacing a leaving group (an atom or group that detaches from the substrate).
Think of it like this: You have a molecule, let's call it the substrate. It has a part that's a bit vulnerable, and attached to it is a leaving group, which is ready to make a quick exit. Then, along comes a friendly nucleophile, eager to form a new bond. In an SN2 reaction, the nucleophile doesn't wait around. It attacks the carbon atom that the leaving group is attached to, but here's the twist: it attacks from the opposite side of the leaving group. This is called inversion of configuration, and it's a hallmark of SN2 reactions. As the nucleophile forms a new bond, the leaving group simultaneously breaks away. It's a beautiful, coordinated maneuver!
The Key Players: What to Look For
To predict the product, you need to identify these crucial players in the reaction:

- The Substrate: In SN2 reactions, the substrate is typically an alkyl halide or a similar compound with a good leaving group. The steric hindrance around the carbon atom being attacked is super important. Primary (1°) and secondary (2°) substrates react readily via SN2. Tertiary (3°) substrates are too crowded for the nucleophile to get in, so they usually go via a different pathway (like SN1).
- The Nucleophile: This is your electron-rich attacker! Common nucleophiles include hydroxide ions (OH-), alkoxide ions (RO-), cyanide ions (CN-), and even some neutral molecules like ammonia (NH3) or amines when they have a lone pair of electrons. A strong nucleophile is essential for SN2 to occur.
- The Leaving Group: This is the part that departs. Good leaving groups are weak bases, meaning they are stable on their own. Examples include halides (like Cl-, Br-, I-) and tosylates. Poor leaving groups, like hydroxide (OH-) itself (unless protonated) or alkoxides (RO-), are less likely to participate in SN2 reactions.
- The Solvent: The reaction environment matters! SN2 reactions are favored by polar aprotic solvents like acetone, DMSO, or DMF. These solvents can solvate the cation accompanying the nucleophile without strongly solvating the nucleophile itself, making the nucleophile more reactive.
Let's Predict! A Simple Example
Imagine you see this reaction:
CH3CH2Br (bromoethane) + NaCN (sodium cyanide) in DMSO -> ?
Let's play detective!
- Substrate: CH3CH2Br. This is bromoethane. The carbon attached to the bromine is a primary (1°) carbon, which is great for SN2.
- Nucleophile: NaCN provides the cyanide ion (CN-). The carbon atom in CN- has a negative charge and lone pairs, making it a strong nucleophile.
- Leaving Group: Bromide (Br-) is a good leaving group.
- Solvent: DMSO is a polar aprotic solvent, ideal for SN2.
Since all the conditions are right for an SN2 reaction, the cyanide ion (CN-) will attack the carbon bonded to the bromine, from the opposite side. The bromine atom (Br-) will leave.
The result? The cyanide group replaces the bromine atom, giving you CH3CH2CN (propanenitrile). The configuration at the carbon atom will be inverted, but in this simple case, it doesn't lead to a stereochemical difference that's easily observable without more complex starting materials.
Practice Makes Perfect!
The more reactions you analyze, the better you'll become at predicting the products. Look at the substrate's structure, identify the nucleophile and leaving group, and consider the solvent. With a little practice and by remembering these key players, you'll be predicting SN2 products like a pro in no time!
