Predict The Product For The Following Sn1 Reaction

Alright chemists, gather 'round! Or maybe just you curious folks who stumbled upon this page. Let's talk about something that might sound intimidating but is actually kinda fun, like trying to guess what's inside a mystery box. Today, we're playing a game: "Predict the Product!"
We've got a special recipe for you. It's called the SN1 reaction. Don't let the fancy name fool you. Think of it as a chemical dance. A rather chill dance, actually. It's not a high-energy, jump-around kind of dance. It's more of a "take your time, let's see what happens" kind of vibe.
So, what's the deal with this SN1 reaction? Imagine you have a molecule. It's got a group attached to it. This group is a bit of a diva. It's like, "You know what? I'm outta here!" And poof, it leaves. This is the first step. It's like when your favorite snack disappears from the pantry. Gone. Vanished. Poof!
Once that diva group is gone, the molecule is feeling a little… naked. It's got a spot where the diva used to be, and it's not too happy about it. This is what we call a carbocation. Think of it as a molecule having an existential crisis. "Who am I now that [diva group] is gone?" It's a bit sad, a bit lost. It's got this positive charge, like it’s feeling all the emotions at once.
Now, here comes the hero of our story. Or, you know, just another molecule minding its own business. Let's call it the nucleophile. This nucleophile is feeling generous. It's got extra electrons, like it just won the lottery of electrons. And it sees the sad, lost carbocation. It's like, "Hey, buddy, you look like you could use a friend. And I have plenty of electrons to share!"
So, the nucleophile, with all its electron love, swoops in and gives the carbocation a big, electron-y hug. This hug is what makes the magic happen. The carbocation feels better. The negative charge from the nucleophile balances out the positive charge of the carbocation. They stick together. They form a new bond. It’s a chemical love story!
And that, my friends, is basically how an SN1 reaction goes down. It's a two-step process. First, the leaving group makes a dramatic exit. Then, the nucleophile makes a grand entrance and saves the day. Simple, right? Well, maybe not that simple if you're trying to get an A in organic chemistry. But for us regular folks, it's pretty neat.
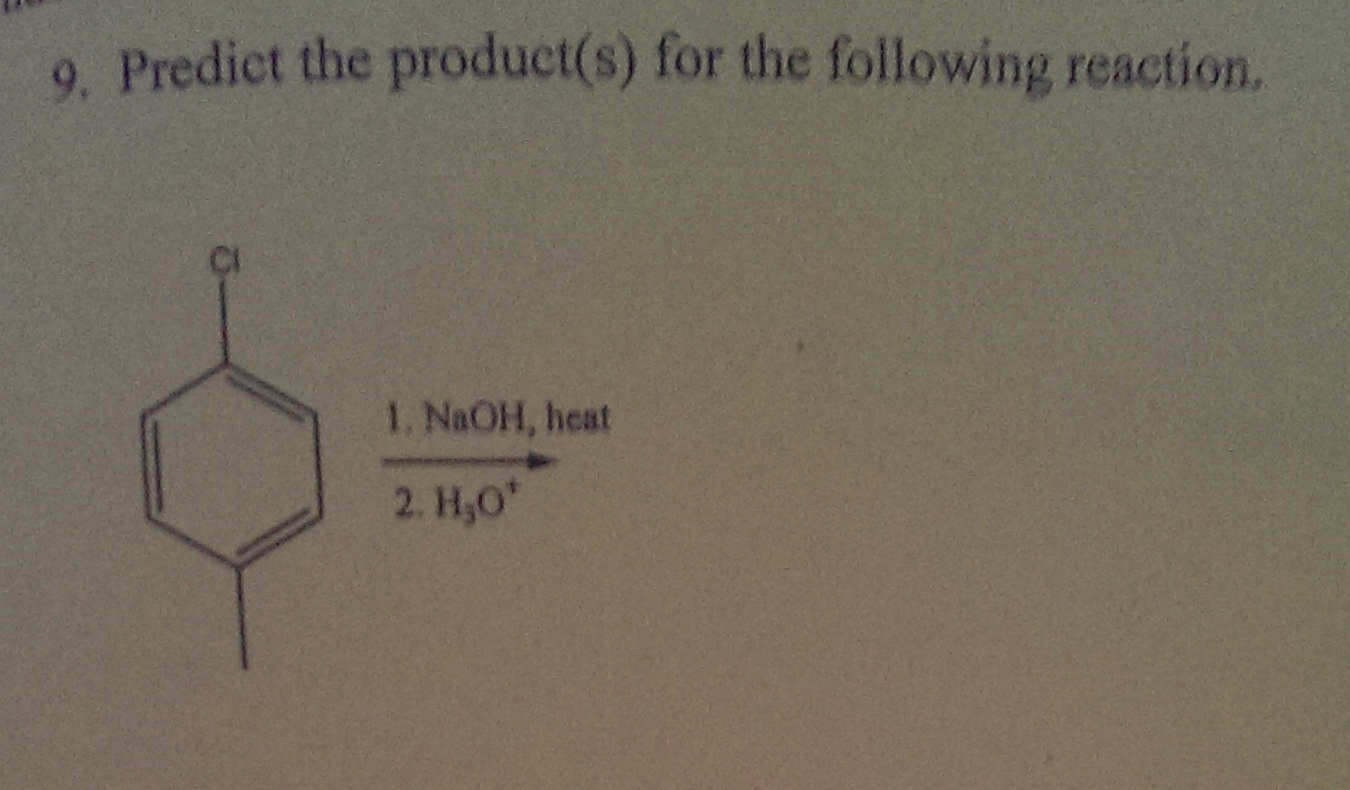
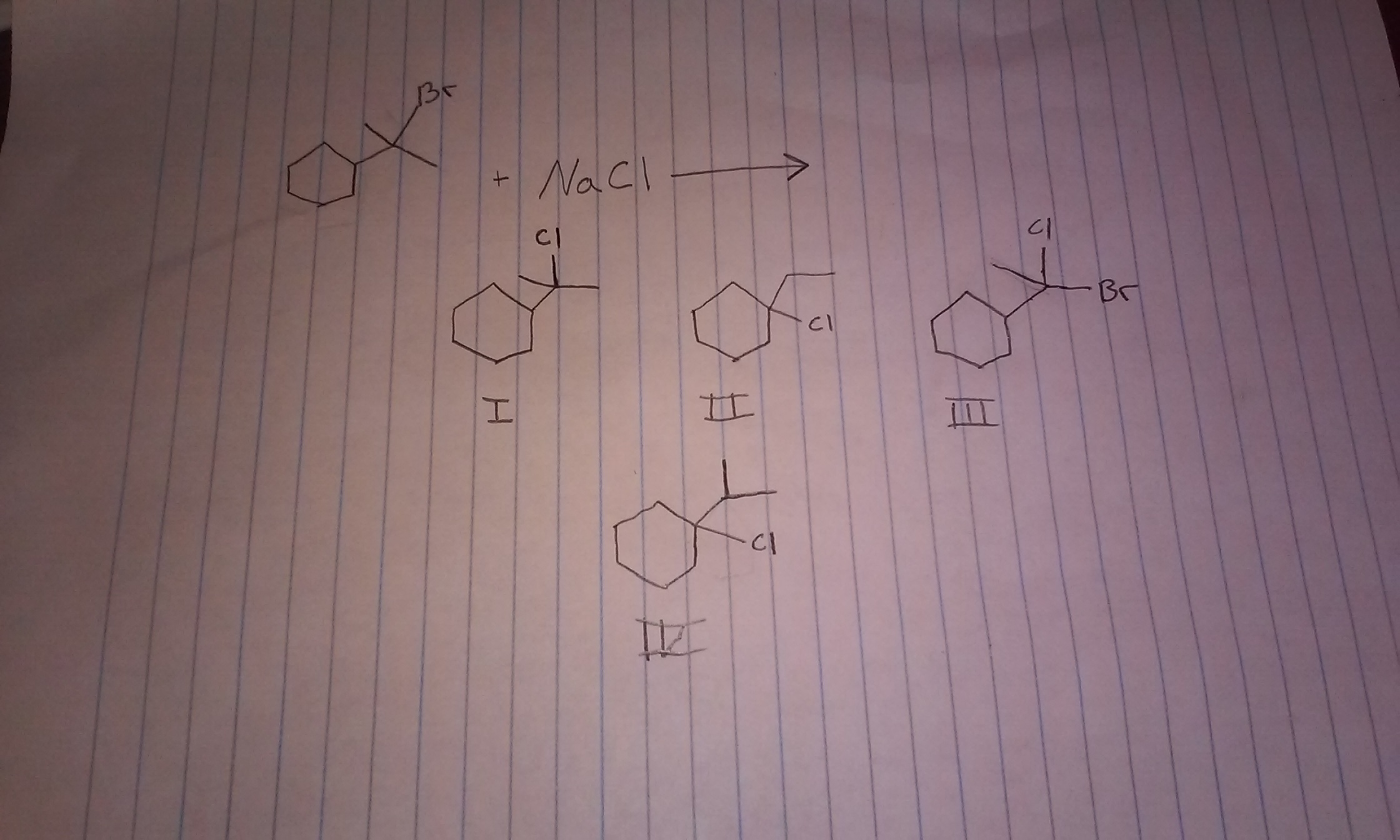
Now, let's get to the fun part: predicting the product. Imagine you're given a starting molecule. It's got that potential diva leaving group. And you're told that a nucleophile is hanging around. Your job is to figure out what the final product will be. It's like a chemical puzzle!
First, identify that leaving group. Is it ready to make its escape? Is it a good candidate for a dramatic exit? Some groups are just better at leaving. They're like those people who always have an excuse to leave a party early. They're just wired that way.
Once it's gone, picture that carbocation. Visualize its lonely, positive self. Then, look for your nucleophile. Where are those extra electrons? What kind of atom is it? Is it oxygen? Nitrogen? Sulfur? These are your electron donors, your potential romancers of the carbocation.

The nucleophile will then attach itself to the carbocation. And guess where it will attach? Usually, it attaches to the very spot where the leaving group used to be. It’s like replacing a broken part in your favorite gadget. You take out the old, put in the new. Ta-da! A fixed gadget. Or, in our case, a brand new molecule.
There are some little nuances, of course. Sometimes, the carbocation can rearrange itself to become more stable. It's like the molecule saying, "You know what? I don't really want to be here. I'd rather be over there because it's a better spot." It can move its parts around to get comfy. This can lead to unexpected products, which is where the real fun (and maybe a little bit of head-scratching) begins.

But generally, if you can identify the leaving group and the nucleophile, you're well on your way to predicting the product of an SN1 reaction. It's like knowing the two main characters in a play. You know they're going to interact, and you can guess the general plotline.
So, next time you see an SN1 reaction, don't panic. Don't run for the hills. Just imagine that diva leaving group making its exit, that carbocation having its moment, and the generous nucleophile swooping in to make things right. It’s a chemical soap opera, really. And you, my friend, get to predict the happy (or at least, new) ending!
It's an unpopular opinion, but I think predicting these products is way more satisfying than actually doing the reaction in a lab. Less mess, fewer explosions (hopefully), and you still get that "aha!" moment. So, there you have it. The mysterious world of SN1, demystified. Or at least, made a little less scary. Happy predicting!
