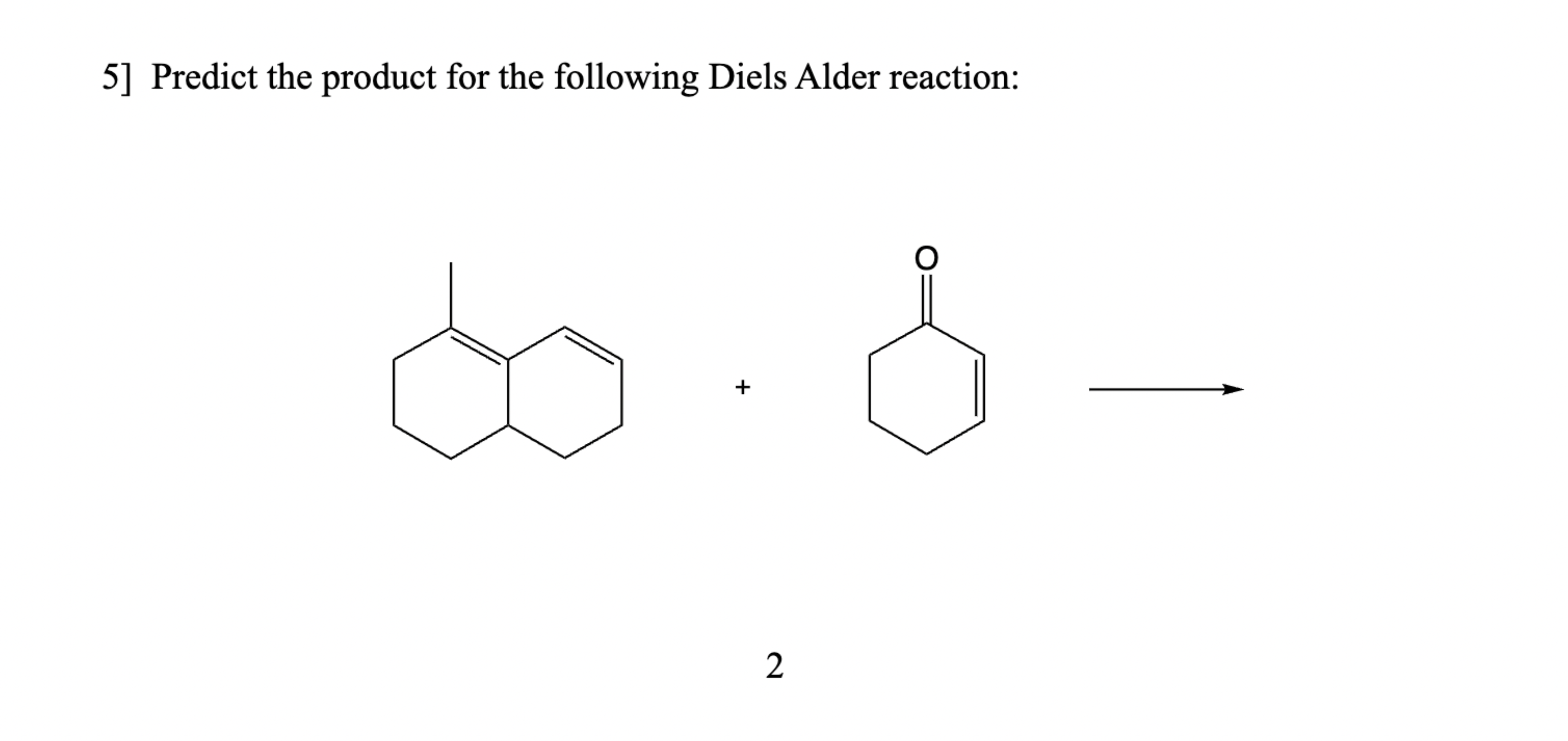
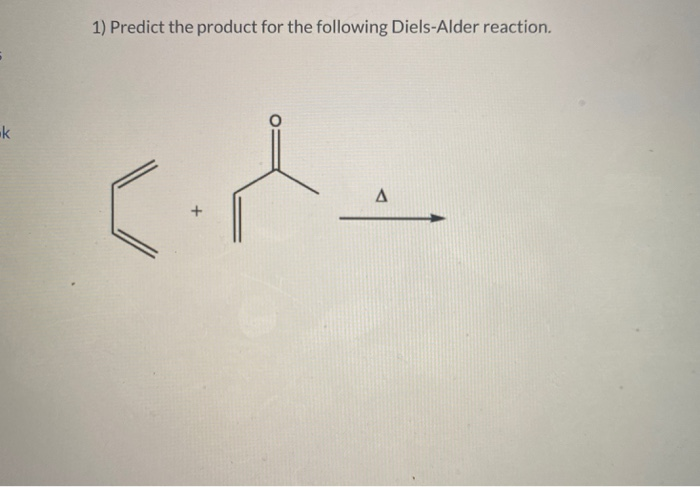
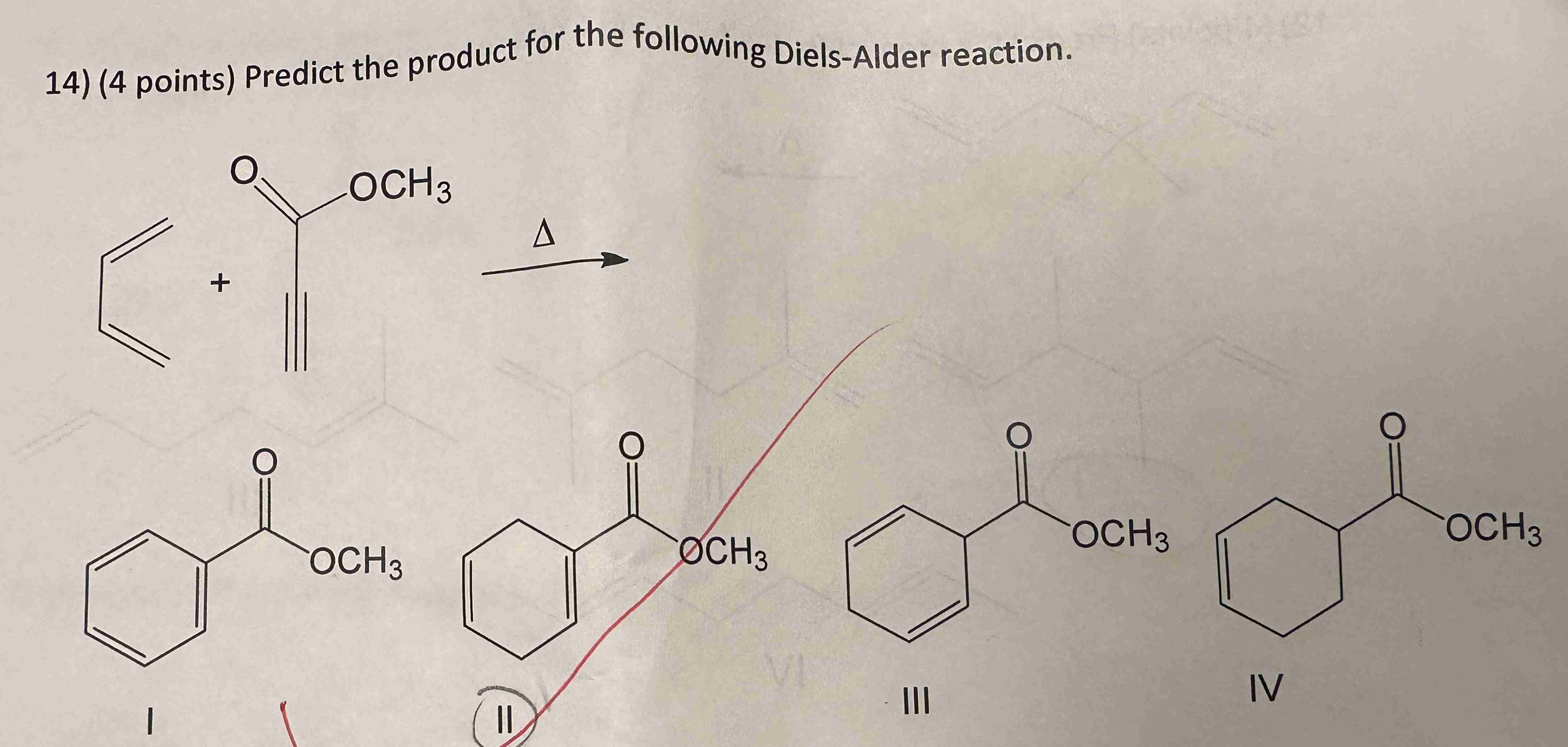
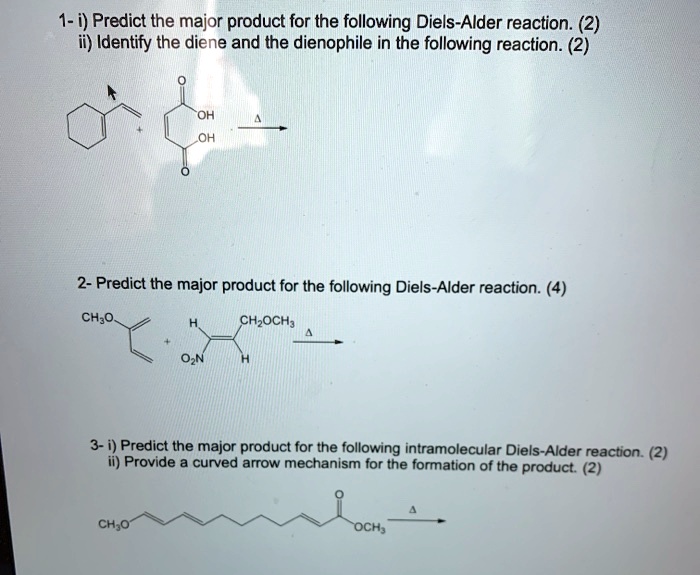
Predict The Product For The Following Diels Alder Reaction
Hey there, chemistry adventurers! Ever feel like you're staring at a puzzle with missing pieces? Well, get ready to feel like a super-sleuth because we're about to crack a really cool chemical mystery together! Today, we're diving headfirst into the dazzling world of the Diels-Alder reaction. Don't let the fancy name fool you – it's like a fantastic chemical dance, and we're going to predict the awesome dance partner that shows up at the end!
Imagine you've got two super-eager molecules, just itching to get together and form something new and exciting. In the Diels-Alder reaction, we have our star performers: a diene and a dienophile. They're like the perfectly matched couple ready for their big moment on the chemical dance floor.
Think of the diene as the groovy dancer with a whole lot of energy and two sets of partnered-up electrons, ready to swing around. It's got a special arrangement of double bonds, kind of like a synchronized dance routine with four atoms. This is where the magic really begins, folks!
Now, meet the dienophile! This is our other star player, and it's usually got a double or triple bond that's just begging for some attention. It's like the eager partner, ready to lock in with the diene and create an even bigger, more stable structure.
So, what happens when these two get together? It's a chemical hug of epic proportions! The electrons from the diene and the dienophile enthusiastically rearrange themselves, forming new bonds and creating a brand new, six-membered ring. It's like they do a perfectly choreographed move, and poof! A new molecule is born!
Our mission, should we choose to accept it (and we totally do!), is to figure out what this new, beautiful ring molecule will look like. It's all about predicting the product. And guess what? It's not as tricky as it sounds. We just need to pay attention to the atomic tango happening between our diene and dienophile.
Let's break it down with a super simple example. Imagine our diene is like a simple straight line with two double bonds in just the right spots. It’s got four carbons in a row, with double bonds separated by a single bond. Think of it as having four hands ready to link up!
And our dienophile? Let's say it's a smaller molecule with a single double bond. It’s like a little two-carbon piece with two hands ready to grab on. It’s got a thirst for connection!
When our straight-line diene meets our little dienophile, they don't just randomly stick together. Oh no! The Diels-Alder reaction is incredibly specific, like a perfect matchmaker. The double bonds in the diene and the double bond in the dienophile will hook up in a very particular way.
The electrons from the diene’s double bonds will reach out and grab the electrons from the dienophile’s double bond. Simultaneously, the electrons from the dienophile will also reach out to the diene. It’s a beautiful exchange!
The end result? They form a six-membered ring. That’s right, six atoms joining hands to create a lovely circular structure. It’s like turning our straight line and little piece into a perfectly formed bracelet!
Now, where do the parts of the original diene and dienophile end up in this new ring? That's the fun part of prediction! Think about the atoms in the diene. The two outer carbons of the diene will become part of the new ring, and the two inner carbons of the diene will also be incorporated.
The carbons that were part of the dienophile's double bond? They become the remaining two carbons in our six-membered ring. So, the entire structure of the dienophile, including anything hanging off of its double bond, gets neatly attached to the new ring.
Let's make it even more visual. Imagine the diene is like a springy, flexible ruler. It has four connection points. The dienophile is like a small, rigid connector with two connection points.
When they come together, the ruler bends and the connector fits perfectly into specific spots. The ends of the ruler connect to the connector, and the middle of the ruler closes up the loop. It’s a seamless integration!
So, if our diene has some cool decorations (or substituents, as chemists like to call them) on its outer carbons, those decorations will end up on the new ring in specific places. And if our dienophile has decorations on its double bond carbons, those decorations will also be part of the final ring structure.
The key to predicting the product is to visualize how the diene "opens up" and how the dienophile "inserts" itself. It’s like fitting puzzle pieces together in a very organized and predictable way.
The atoms from the diene form the "backbone" of the new ring, and the atoms from the dienophile become the "joining" parts that complete the ring.
Let's say our diene is a simple butadiene. It's the simplest diene, like the foundation of our chemical house. And our dienophile is something like maleic anhydride. This little guy is super reactive and has a double bond all ready to go.
When butadiene and maleic anhydride meet, they create a beautiful six-membered ring. The butadiene carbons form the basic ring structure. The maleic anhydride's double bond carbons become the other two carbons, and its anhydride part gets attached to the ring.
The amazing thing is, there are rules! We don't just guess. We understand how the electrons move. It’s like a secret handshake that chemistry follows.
One of the most important rules is about stereochemistry. That’s a fancy word for the 3D arrangement of atoms. But for now, just know that the "up" and "down" of the dienophile often stays "up" and "down" in the product.
Think of it this way: if the dienophile is like a flat little cracker, and it approaches the diene from the top, its own shape will remain the same in the new ring, just attached in a new place. It's not going to flip over unexpectedly!
So, if you see a dienophile with two groups sticking out, say, pointing upwards, those groups will likely still be pointing upwards in the final product, relative to the ring they’re attached to.
This predictability is what makes the Diels-Alder reaction so incredibly powerful. Scientists can use it to build complex molecules with great precision. It’s like having a super-powered Lego set for chemists!
We can design our diene and dienophile with specific "decorations" to make exactly the molecule we want. It’s like choosing the perfect toppings for your pizza, but for chemical structures!
Let’s try another mental picture. Imagine the diene is like a stretchy rubber band that can be pulled into a certain shape. The dienophile is like a rigid clamp.
When the rubber band (diene) is in the right configuration, and the clamp (dienophile) comes along, they snap together. The clamp attaches to the ends of the rubber band, and the rubber band closes the loop, forming a nice, stable circle.
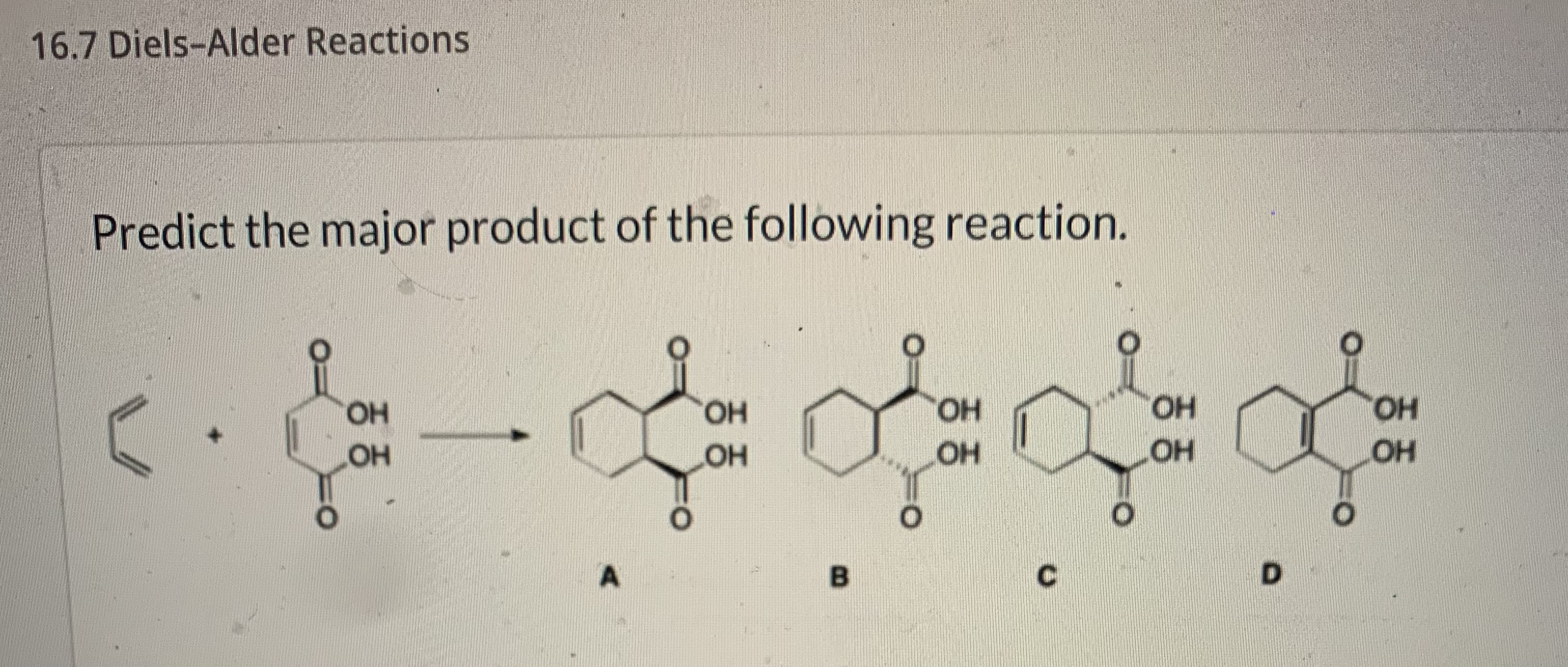
The carbons that were on the ends of the diene become part of the ring. The carbons that were in the middle of the diene also become part of the ring. And the two carbons from the dienophile connect everything and become part of the ring too.
It’s like a grand chemical reunion where everyone finds their perfect spot. The old double bonds break, and new single bonds form, creating a whole new molecular family.
So, when you see a Diels-Alder reaction, your brain should immediately start thinking about a six-membered ring. That's your new, exciting structure! Then, you just need to figure out where the pieces of your original diene and dienophile fit into that ring.
Look at the connectivity. The outer carbons of the diene are always the ones that form new bonds with the dienophile. And the dienophile's double bond carbons become part of the ring where those new bonds are formed.
It’s a beautiful dance of electrons and atoms, and the Diels-Alder reaction is one of its most elegant choreographies. By understanding the basic players and how they interact, you can totally predict the fantastic product they’ll create!
So next time you see a diene and a dienophile, don't be shy! Just imagine them doing their special chemical dance, and you’ll be able to predict the amazing ring they form. You've got this!
