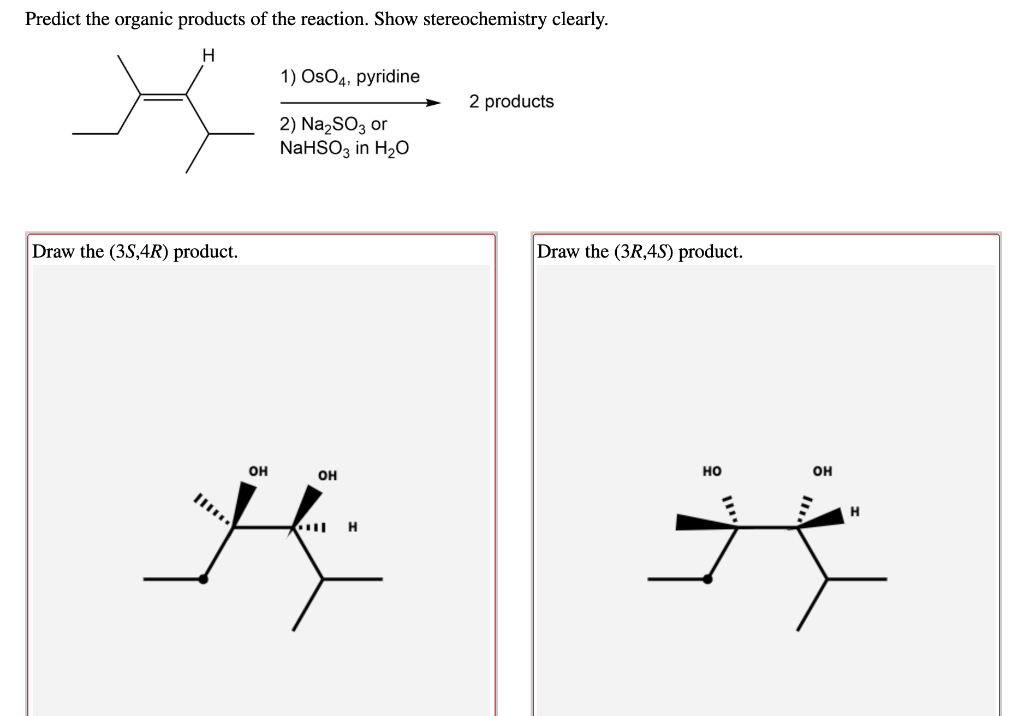
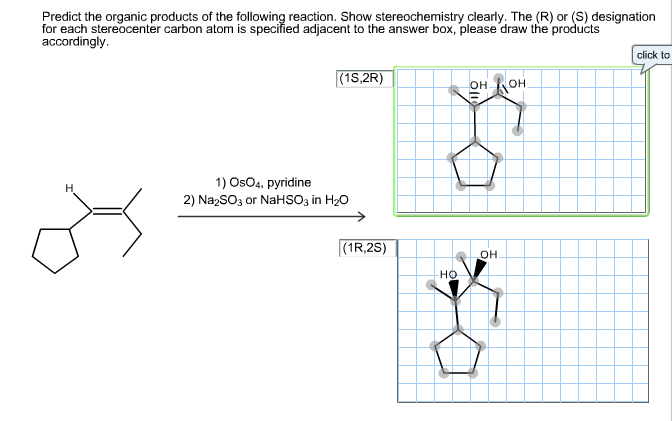
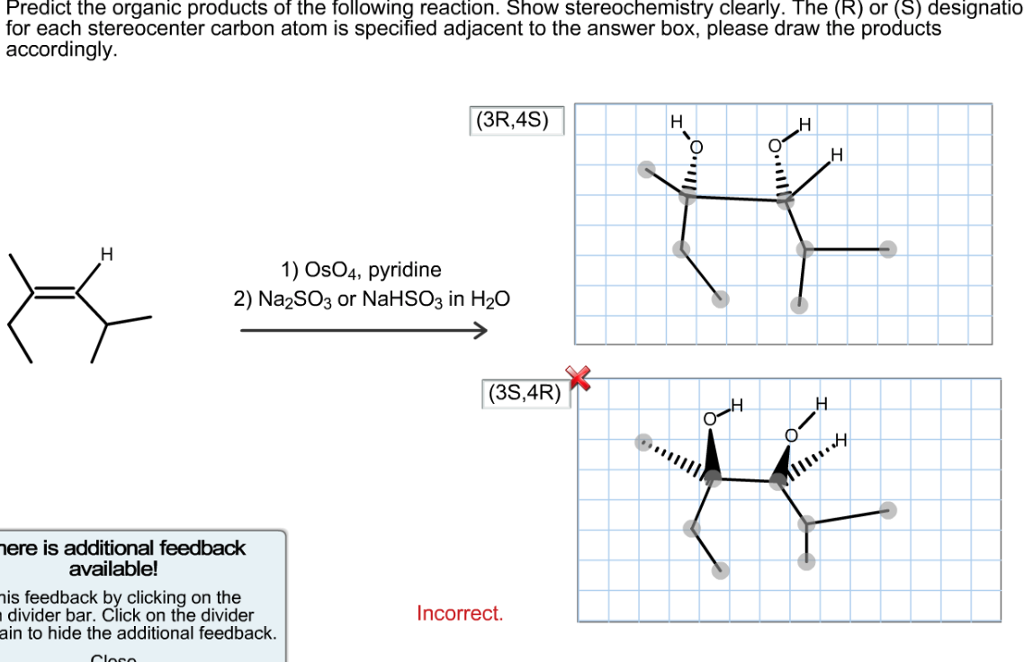
Predict The Organic Products Of The Reaction. Show Stereochemistry Clearly
Hey there, science enthusiasts (and even you curious folks who just stumbled upon this)! Ever looked at a recipe and wondered what magical combination of ingredients will end up on your plate? Or maybe you’ve seen a car engine hum and thought, “How do all those tiny parts work together to make it move?” Well, guess what? The world of chemistry has its own delicious recipes and intricate machinery, and today we’re going to peek behind the curtain to talk about predicting the organic products of a reaction. And the coolest part? We’ll even make sure to show you the stereochemistry clearly, which is like the fancy dance steps of molecules!
Now, before you click away thinking this sounds like a super-hard math problem, take a deep breath. We’re going to keep this as chill as a Sunday morning with a cup of coffee. Think of it like this: you’ve got a bunch of LEGO bricks, and you know how they fit together. Chemistry is kind of the same, but instead of plastic bricks, we’re dealing with atoms and molecules. And just like you can build a spaceship or a castle with LEGOs, chemists can predict what cool new molecules will pop out when you mix certain starting materials together.
So, why should you even care about predicting organic reaction products and stereochemistry? It’s not just for super-smart folks in lab coats. It’s actually pretty darn important for… well, pretty much everything you touch, use, and consume! Think about the medicines that keep us healthy, the clothes we wear, the fuel that powers our cars, even the flavors in your favorite ice cream – all of these are the result of carefully orchestrated chemical reactions. Understanding these reactions means we can design new and better stuff, make processes more efficient, and even help protect our planet.
Let’s start with the basics. In organic chemistry, we’re usually talking about molecules that contain carbon. Carbon is like the ultimate social butterfly of the periodic table – it loves to bond with other atoms and form long chains and intricate structures. Think of a carbon atom as a person who loves to hold hands with their friends. It usually holds four hands (bonds). Organic reactions are basically when these carbon-containing molecules get together, rearrange their hand-holding partners, and form something new.
Predicting the products is like being a detective. You look at the suspects (the starting materials), you see what clues they have (their chemical structures and functional groups), and you use your knowledge of how they like to interact to figure out who will end up with whom in the new formation.
Now, about that “stereochemistry” thing. Imagine you’re making those cool little gummy bears. You have the gummy mix, right? That’s your starting material. When it sets, you get gummy bears. But what if you want them to be facing left instead of facing right? That’s stereochemistry! It’s all about the three-dimensional arrangement of atoms within a molecule. Sometimes, molecules can exist in different forms that are mirror images of each other, like your left and right hands. They look super similar, but they’re not quite the same. And this difference can be a huge deal, especially in biology.

Think about a drug. One form of a drug might be a fantastic medicine, while its mirror image could be completely useless, or even harmful! So, chemists need to be able to predict not just what molecule is formed, but also how it’s arranged in space. It's like making sure you're building a perfectly functioning robot, not one that's accidentally built backwards!
Let’s take a simple example. Imagine you have a molecule with a double bond, like the edge of a dance floor where two dancers are holding hands and facing each other. If you add something new, like a couple of new dancers wanting to join the party, they’ll break that hand-hold and find new partners on either side of where the double bond used to be. This process is called an addition reaction.
Now, the stereochemistry comes into play because those new dancers can join from the same side, or they can join from opposite sides. It’s like they can either step onto the dance floor together, or one can step from the left and the other from the right. The way they join will create a different arrangement of the atoms in the final molecule.
We have special ways of showing this on paper. Think of it like drawing a diagram for a dance move. We use wedges to show bonds coming out towards you, dashed lines for bonds going away from you, and regular lines for bonds in the plane of the paper. It’s our way of creating a 3D map on a 2D surface so everyone can see the molecular choreography.
For instance, if we’re adding hydrogen to a double bond using a catalyst like platinum or palladium (think of these as the enthusiastic dance instructors), the hydrogen atoms often like to add to the same side of the double bond. This is called a syn addition. It's like the two new dancers politely stepping onto the floor together, side-by-side.
On the other hand, sometimes the addition happens in a way where the new groups end up on opposite sides. This is called an anti addition. It’s like one dancer gracefully stepping in from stage left, and the other from stage right.

Predicting these things isn’t just about memorizing rules. It’s about understanding the mechanisms of the reactions. A mechanism is like the step-by-step play-by-play of how the reaction actually happens. It’s the story of how bonds break and form, which intermediates are involved (these are like temporary dancers on the sidelines), and how the final product is assembled.
Chemists use a variety of tools and knowledge to predict these products. They look at the functional groups present – these are like the specific features of our molecules that dictate their reactivity. For example, a molecule with a double bond behaves differently than one with a triple bond or one with an alcohol group (like in your hand sanitizer!). They also consider the reaction conditions – temperature, pressure, the presence of solvents or catalysts. These are like the music tempo and the size of the dance floor!
One really common type of reaction we predict is electrophilic addition to alkenes (those molecules with double bonds). Imagine our alkene is a hungry monster with a double mouth. An electrophile is something that’s electron-deficient, like a lost traveler looking for a place to stay. This electrophile will be attracted to the electron-rich double bond and will attack it, breaking one of the bonds in the double bond. Then, other parts of the molecule will jump in to fill the remaining spots.
The stereochemistry in these reactions is often determined by how the electrophile attacks and how the subsequent steps occur. Sometimes, it's a concerted process (like a perfectly choreographed ballet move where everything happens at once), and other times it involves intermediates that can lead to different stereochemical outcomes. We often talk about racemic mixtures in these cases, which are 50/50 mixtures of both mirror images. It’s like having an equal number of left-footed and right-footed dancers showing up!
So, why is this really so cool and relevant? Because every time a new drug is developed, a new polymer for your phone case is designed, or a more sustainable fuel is created, chemists have been doing this predictive work. They’ve been playing with molecular LEGOs, understanding the dance steps, and ensuring the final product is exactly what they intended.
It’s like being a chef. You don't just throw ingredients in a pot and hope for the best. You understand how heat affects proteins, how acids react with bases, and how different spices combine to create a symphony of flavors. Predicting organic products and stereochemistry is the chef’s understanding of chemical cooking. It’s what allows us to create all the amazing things that make our modern lives possible.
So, next time you’re enjoying a medicine, wearing a synthetic fabric, or even just marveling at the complex world around you, remember the invisible dance of molecules and the brilliant minds that can predict its steps and outcomes. It’s a fundamental part of the world we live in, and understanding it, even a little bit, opens up a whole new way of seeing things!
