Predict The Molecular Shape Of The Sulfite Ion.

Hey there, science enthusiasts and curious minds! Ever wonder about the tiny, invisible world that makes up everything around us? It’s a place full of incredible structures and fascinating behaviors. Today, we’re going to dive into the wonderfully weird world of chemistry and tackle a question that might sound a little intimidating at first: how do we predict the molecular shape of the sulfite ion? But trust me, by the end of this, you’ll see that understanding these molecular blueprints can be not just cool, but downright fun, and even a little bit inspiring!
Think of it like this: you’re an architect, but instead of designing buildings, you’re designing molecules. And the sulfite ion? Well, that’s a particularly interesting client with a specific request for its shape. Sounds like a puzzle, right? And who doesn't love a good puzzle?
Unraveling the Mystery of the Sulfite Ion
So, what exactly is this “sulfite ion”? It’s a little chemical critter with the formula SO₃²⁻. That “SO₃” part tells us it’s made of one sulfur atom (S) and three oxygen atoms (O). The “²⁻” signifies that it has a negative charge, meaning it’s gained two electrons. This is important because those extra electrons play a big role in how everything fits together!
Now, you might be thinking, “How can I possibly know what this invisible thing looks like?” That’s where the magic of chemistry comes in. We have tools, like a super-powered X-ray vision for molecules, called Valence Shell Electron Pair Repulsion theory, or VSEPR theory for short. Don't let the fancy name scare you – it's actually a pretty intuitive concept.
VSEPR: The Ultimate Molecular Matchmaker
VSEPR theory is built on a simple, yet profound, idea: electrons, whether they’re busily bonding atoms together or just hanging out in pairs, really don't like being too close to each other. They’re like tiny, energetic toddlers who need their personal space! So, what do they do? They spread out as much as possible around the central atom, minimizing those awkward, electron-to-electron bumps.
To predict the shape of the sulfite ion, we need to figure out how many "things" (which are either bonding pairs of electrons or lone pairs of electrons) are around the central sulfur atom. This will tell us how those electron groups will arrange themselves in space to get the most room.
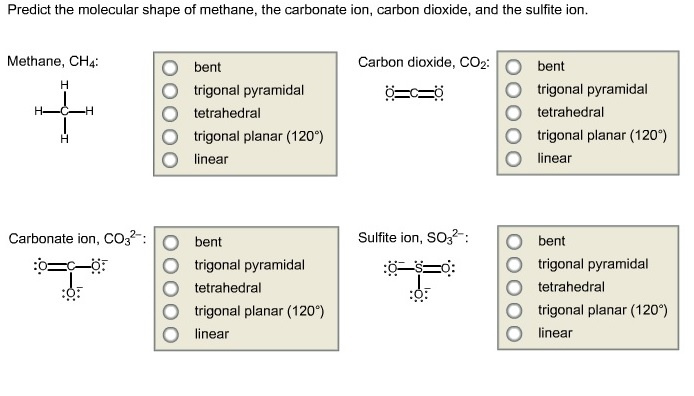
First, let's find our central atom. In SO₃²⁻, sulfur is the less electronegative atom, so it’s our star player in the middle. Now, we need to count up the valence electrons. Sulfur is in Group 16 of the periodic table, so it has 6 valence electrons. Each oxygen atom is also in Group 16, so each brings 6 valence electrons to the party. And don't forget that extra negative charge – those two added electrons are super important! So, our total valence electrons are: 6 (from S) + 3 * 6 (from O) + 2 (from the charge) = 26 valence electrons.
Next, we draw a Lewis structure, which is like a rough sketch of how the atoms and electrons are connected. We'll connect the sulfur to each of the three oxygens with single bonds. That uses up 3 * 2 = 6 electrons. We have 20 electrons left. Now we fill the outer shells of the oxygen atoms. Each oxygen needs 6 more electrons to be happy (have a full octet), so we add 6 electrons to each of the three oxygens. That’s 3 * 6 = 18 electrons. We have 2 electrons left. Where do they go? They go on the central sulfur atom as a lone pair.
But wait! If we look closely, the oxygen atoms only have 6 electrons around them (2 from the single bond and 4 lone pair electrons). To give them octets, we can move one lone pair from two of the oxygen atoms to form double bonds with the sulfur. Now, each oxygen atom has 8 electrons around it (2 from the double bond, 4 from lone pairs, and 2 from the single bond for the third oxygen, which means we would have two resonance structures where the single bond moves). Even better, the sulfur atom now has 8 electrons around it (2 from the single bond, 2 from the double bonds, and 2 from the lone pair).
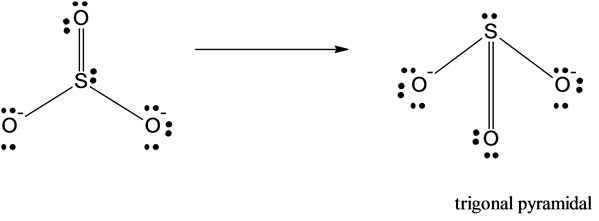
So, in our best Lewis structure for the sulfite ion, the central sulfur atom is bonded to three oxygen atoms (two with double bonds and one with a single bond) and has one lone pair of electrons. This means there are a total of four electron groups around the sulfur atom: three bonding groups and one lone pair.
The Shape Revelation: A Splash of Color!
Now, here’s where VSEPR theory truly shines! With four electron groups, the electrons will arrange themselves to be as far apart as possible. If they were all bonding pairs, they’d form a perfect tetrahedron. But we have a complication: a lone pair. Lone pairs take up more space than bonding pairs. Think of them as those boisterous cousins at a family gathering – they tend to hog a bit more room!
Because of that bulky lone pair, the three bonding pairs will be pushed slightly closer together. Imagine you’re trying to arrange three balloons around a central point, and then you add a fourth, slightly deflated balloon right in the middle. The three full balloons will shift to accommodate the fourth one, but they’ll still try to spread out as much as they can.

This arrangement of four electron groups (three bonding pairs and one lone pair) leads to a molecular geometry called trigonal pyramidal. If you picture it, it looks like a little pyramid with a triangular base. The sulfur atom is at the apex (the pointy top), and the three oxygen atoms form the triangular base. The lone pair sits above or below the sulfur, influencing the positions of the oxygens.
It’s like the sulfite ion is saying, “I’m supposed to be flat and spread out, but I’ve got this extra little oomph from my lone pair, so I’m going to get a little cozy and form this neat, pyramid-like structure!” Pretty cool, right?
Why This Matters (Beyond Just Knowing Shapes!)
You might be thinking, “Okay, so it’s a pyramid. So what?” Well, the shape of a molecule is hugely important! It dictates how that molecule interacts with other molecules. Think about a lock and key – the shape of the key has to perfectly match the lock for it to work. The same goes for molecules.

The shape of the sulfite ion influences its reactivity, its solubility, and how it behaves in chemical reactions. It plays a role in everything from the formation of minerals to how our bodies process certain compounds. It’s these tiny, unseen shapes that orchestrate the grand symphony of chemistry all around us.
And understanding these shapes? It’s not just about memorizing facts. It's about developing a visual intuition for the molecular world. It’s about seeing the underlying order and elegance in what might initially seem like chaos. It’s a bit like learning a new language, but instead of words, you’re learning the grammar of molecules. And once you know the grammar, you can start to appreciate the poetry!
So, the next time you hear about an ion or a molecule, remember that it has a specific, predictable shape. And that shape isn't just a random arrangement; it's the result of fundamental principles of electron repulsion and the elegant dance of atoms.
Learning to predict these shapes is like unlocking a secret code to the universe. It’s empowering, it’s fascinating, and it’s a fantastic reminder that even in the smallest of things, there’s incredible structure and beauty waiting to be discovered. Keep exploring, keep questioning, and you’ll find that the world of chemistry is an endless source of wonder. Who knows what other molecular shapes you’ll predict next!
