Predict The Molecular Geometry And Polarity Of The So2 Molecule.

So, you've got this molecule, right? Let's call it SO2. It's like the quirky cousin of water, you know? Everyone talks about H2O, but SO2 has its own cool story to tell.
Now, predicting its shape can feel like trying to guess what your cat is thinking. Sometimes it's obvious, and sometimes it's a complete mystery. But with SO2, we can actually make some pretty good guesses. It's not quite as simple as "straight line" or "perfect triangle."
Think of it like arranging furniture in a small room. You want everything to fit nicely, but sometimes there's just one awkward chair that has to go somewhere. SO2 has a similar vibe. It’s trying to find its happy place.
The big players in this shape-shifting game are the atoms themselves. We've got one sulfur atom, which is like the host of the party. And then we have two oxygen atoms, who are the enthusiastic guests. They all want their own space.
Now, these atoms don't just float around randomly. They've got these things called electron pairs. These are like little invisible hands that push and pull on each other. It's a bit of a molecular wrestling match.
The sulfur atom is kind of the center of attention here. It's got these electron pairs hanging around it. Some of these pairs are busy holding hands with the oxygen atoms, forming bonds. Others are just chilling, they're called lone pairs.
And here's where it gets interesting. These electron pairs, whether they're bonding or chilling, all want to be as far away from each other as possible. It’s like when you’re on a crowded bus, you try to spread out, right? Same principle.
So, the sulfur atom, with its bonding pairs and its lone pair, ends up arranging itself in a specific way. It's not a perfect flat arrangement, not like a table. More like a … well, let's get to that!

When you factor in those lone pairs, things get a little bent out of shape. Literally. If SO2 were just a sulfur and two oxygens with no lone pairs, it would probably be a nice, straight line. Boring!
But that lone pair on the sulfur atom is a bit of a troublemaker. It pushes the bonding pairs closer together. It's like that one friend who always makes you huddle up for a group photo.
This pushing and shoving leads to a molecular shape that’s not linear. It’s got a bend in it. Imagine a seagull in flight, with its wings spread out. That's sort of the vibe we're going for.
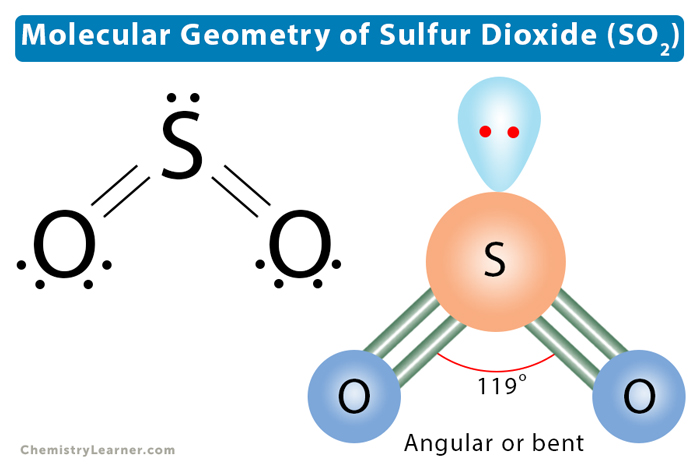
Scientists have a fancy word for this shape: bent. Or sometimes, they call it angular. It just means it’s not straight. It’s got a distinct angle.
So, the molecular geometry of SO2 is bent. It’s like a little V shape, with the sulfur atom at the point. The oxygen atoms are at the ends of the V.
Now, about this whole polarity thing. This is where things get even more fun. Polarity is all about how the electric charges are distributed within a molecule. Is it like a perfectly balanced scale, or is one side a little heavier?

With SO2, the oxygen atoms are a bit greedy when it comes to electrons. They pull harder on the shared electrons than the sulfur atom does. Think of them as having a stronger magnetic pull.
This means the oxygen end of the molecule ends up with a slightly negative charge. It's like a tiny bit of static electricity building up. Not enough to shock you, but it’s there.
The sulfur atom, on the other hand, ends up with a slightly positive charge. It’s lost a little bit of its electron-y goodness. It’s the opposite pole.
So, because the molecule is bent, and because the charges are not evenly distributed, SO2 is a polar molecule. It's got a distinct positive end and a distinct negative end.
It's not like water, which is also polar. But it's definitely not like, say, methane, which is nonpolar. SO2 has a personality when it comes to charge.
Think of it like a tiny magnet. It has a north pole and a south pole. SO2 has a positive pole and a negative pole.

Why does this matter? Well, this polarity affects how SO2 interacts with other molecules. Polar molecules tend to stick to each other. They're like tiny little sticky notes.
This is why SO2 can dissolve in water, for example. Water is also polar, and opposites attract, as they say.
It’s also important to note that even though the bonds between sulfur and oxygen are themselves polar, the overall polarity of the molecule depends on its shape. If SO2 were linear, the polarities of the two S-O bonds might cancel each other out. But it’s not linear, is it?
It’s that bend in its shape that really seals the deal for SO2 being a polar molecule. It's the combination of unequal electron pulling and a non-symmetrical arrangement.
So, there you have it. SO2. A bent molecule. A polar molecule. It’s like the understated but interesting character in a movie.
It’s not as universally loved as water, and it’s definitely not as simple as a straight-line molecule. But it's got its own charm and its own importance.
And the best part? We predicted this just by looking at the atoms and how their electron friends hang out. It's like molecular detective work, but way less messy.
So next time you hear about SO2, you can picture its little bent structure. And you can imagine its slight electrical tilt. It’s not rocket science, but it’s pretty neat science.
And who knows, maybe SO2 secretly wishes it were a straight line sometimes. Or maybe it loves its bent, polar personality. We’ll never truly know. But we can appreciate its molecular choices.
It’s a reminder that even the simplest-looking things can have complex and interesting structures. Just like people, I suppose. Some are straight shooters, and some have a bit of a curve to them.
So, embrace the bent. Embrace the polar. Embrace SO2. It’s got its own groove.
Sometimes, the most interesting things are a little bit bent out of shape.
And that's my not-so-unpopular opinion about SO2. It's cool. It's polar. It's bent. And that's just fine by me.
