Predict The Major Product For The Sn1 Reaction Shown.

Hey there, fellow organic chemistry enthusiast! Grab your coffee, settle in, because we're about to dive into something super cool, a little bit like solving a mystery, but with molecules. We're gonna predict the major product of an SN1 reaction. Sounds fancy, right? But honestly, it's more like figuring out who's gonna win a popularity contest in the world of chemical reactions.
So, you've got this reaction staring you in the face, right? Maybe it looks a little intimidating at first, all those letters and arrows. But don't sweat it! We're gonna break it down, piece by piece, like a really engaging puzzle. Think of me as your friendly, slightly caffeinated guide through the wacky world of nucleophilic substitution, SN1 style.
First things first, what even is an SN1 reaction? Let's refresh our memories. SN stands for Substitution, Nucleophilic. And the '1' part? That means it's a unimolecular reaction. What does that even mean, you ask? It means the rate-determining step – the slowest, bottleneck step – only involves one molecule. Revolutionary, I know!
Imagine a party. The unimolecular step is like one person deciding to leave the dance floor all by themselves. Nobody else is involved in that specific decision. The other molecule, the nucleophile (more on that later!), can just chill out and wait for the drama to unfold. It's all about that one molecule doing its thing.
Now, what makes an SN1 reaction tick? It's all about the carbocation. Oh boy, carbocations! These guys are like the divas of the molecule world. They're positively charged, and they're usually formed when a good leaving group hops off. You know, like a tiny, spoiled celebrity ditching a bad gig.
And when I say 'good leaving group,' I mean it. Think of things like halides (bromine, chlorine, iodine – they're pretty decent at bouncing), or tosylates, or mesylates. These are the ones that are happy to go off on their own, leaving behind a positive charge. The weaker the bond holding them, the happier they are to leave, and the easier it is to form that carbocation. Easy peasy!
So, the general vibe of an SN1 reaction is a two-step process. Step one: the leaving group peaces out, forming a carbocation intermediate. This is the slow, lazy step. It takes its sweet time. Step two: the nucleophile, that eager beaver, swoops in and attacks the carbocation. This is the fast step, like a lightning-fast high five.
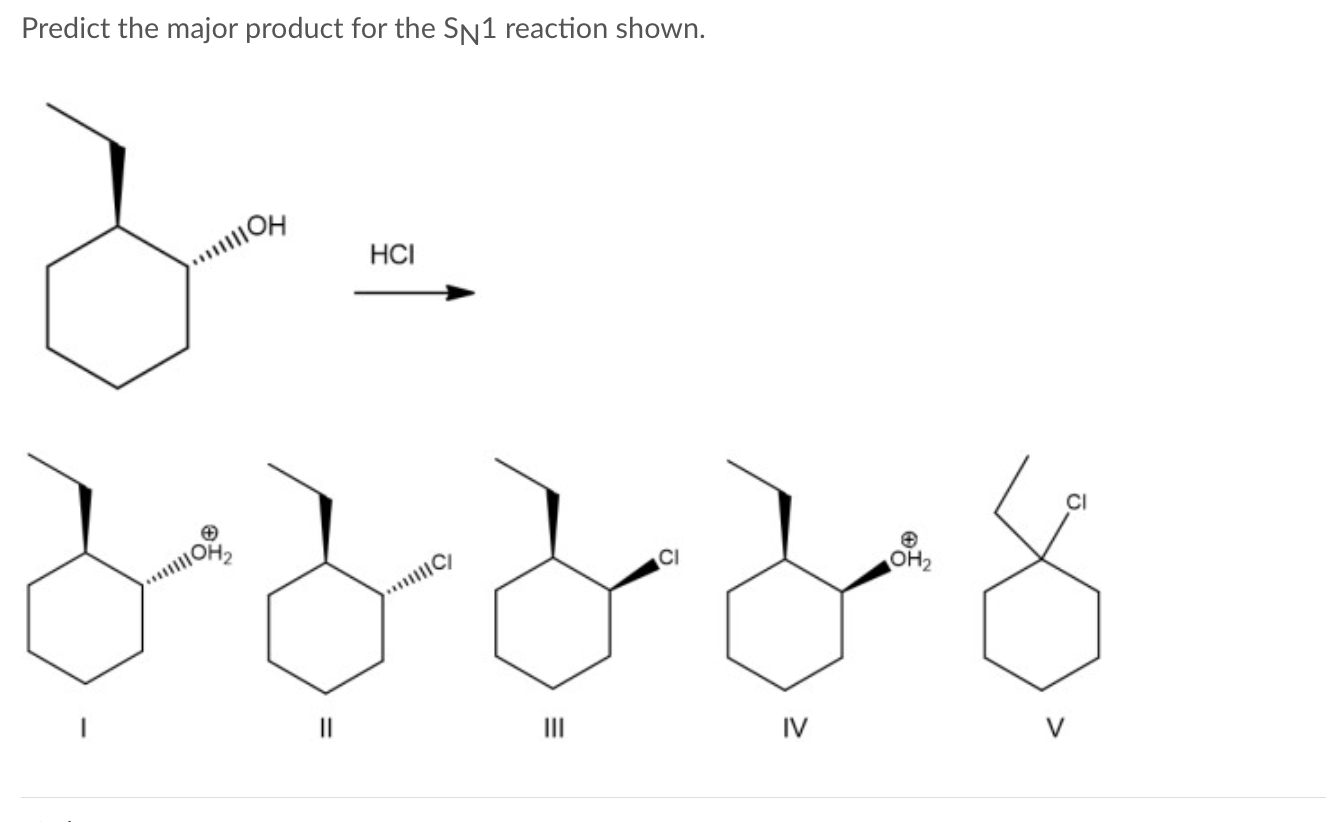
Okay, so let's get to the juicy part: predicting the major product. This is where the real detective work comes in. We need to consider a few key players. First, the substrate – that's the molecule with the leaving group. Second, the nucleophile – the guy who's gonna do the attacking. And third, the solvent – where all this action is happening. It’s like choosing the right venue for your epic molecular party.
When we're talking SN1, we're usually dealing with tertiary substrates. Why tertiary? Because tertiary carbocations are the most stable. Think of it like this: a tertiary carbocation has three alkyl groups attached to the positively charged carbon. These alkyl groups are electron-donating, like little cheerleaders giving the positive charge a hug and saying, "We got your back!" This makes the carbocation much happier and more likely to form. Secondary substrates can do SN1, but they might be a bit more hesitant. Primary? Forget about it. Their carbocations are just too unstable. Like trying to build a house on sand, you know?
So, if you see a tertiary substrate with a good leaving group, you can pretty much bet your bottom dollar that SN1 is on the menu. Unless, of course, there are other, even crazier reactions trying to muscle in. But for now, SN1 is our main suspect.
Now, the nucleophile. For SN1, the nucleophile is typically weak. Think of water, or alcohols. They're not super aggressive attackers. They're more like, "Oh, is there a positive charge over there? I guess I could say hi." This is perfect for SN1 because if the nucleophile were super strong, it might try to attack the carbon before the leaving group even leaves. That would be an SN2 reaction, a whole different beast! We're not here to talk about SN2 today, so let's keep our focus on the chill, laid-back SN1 vibe.
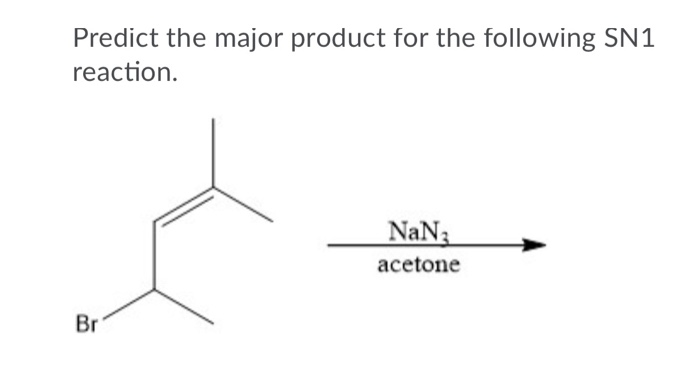
And then there's the solvent. For SN1, we love polar protic solvents. What's a polar protic solvent? It's a solvent that has a hydrogen atom bonded to an electronegative atom, like oxygen or nitrogen. Think water (H₂O), or methanol (CH₃OH), or ethanol (CH₃CH₂OH). These solvents are great because they can solvate both the carbocation and the leaving group. They surround them, stabilize them, and basically make them feel comfortable and less likely to bail. It's like having a whole support team for our molecules.
So, we've got our tertiary substrate, our weak nucleophile, and our polar protic solvent. The stage is set for SN1! The leaving group bails, creating that beautiful, albeit fleeting, carbocation. And then, the nucleophile, feeling emboldened by the solvent's support, waltzes in and attaches itself to the carbocation.
But wait, there's a twist! Remember that carbocation we formed? It's planar. Like a flat pizza. And the nucleophile can attack it from either side. This is super important, especially if we started with a chiral center. If the leaving group was attached to a chiral carbon, and the nucleophile attacks from the opposite side of where the leaving group was, we get inversion of configuration. Like flipping a pancake. If it attacks from the same side, we get retention of configuration. Like… well, like the pancake stayed put. In an SN1 reaction, you usually get a racemic mixture, a 50/50 mix of both, because both sides are equally accessible.
However, there's a subtle little detail that can sometimes tip the scales. If the carbocation is particularly stable and lives for a tiny bit longer, the leaving group can sometimes hang around nearby, like a bodyguard. If the nucleophile attacks from the same side as this lingering leaving group, it can lead to a little more retention of configuration than you'd expect. It's a tiny effect, but it can make one product slightly more abundant. So, while we often say "racemic mixture," sometimes it's more like "mostly racemic mixture, with a hint of something else."
Now, let's talk about carbocation rearrangements. This is where things can get really interesting, and sometimes a little tricky. Carbocations, bless their hearts, are always looking to be more stable. If a carbocation can rearrange itself to become more stable, it will! This usually involves a hydride shift (a hydrogen atom with its electron pair moves) or an alkyl shift (an alkyl group moves). The goal is to move the positive charge to a carbon that's bonded to more other carbons (tertiary is better than secondary, secondary is better than primary, duh).
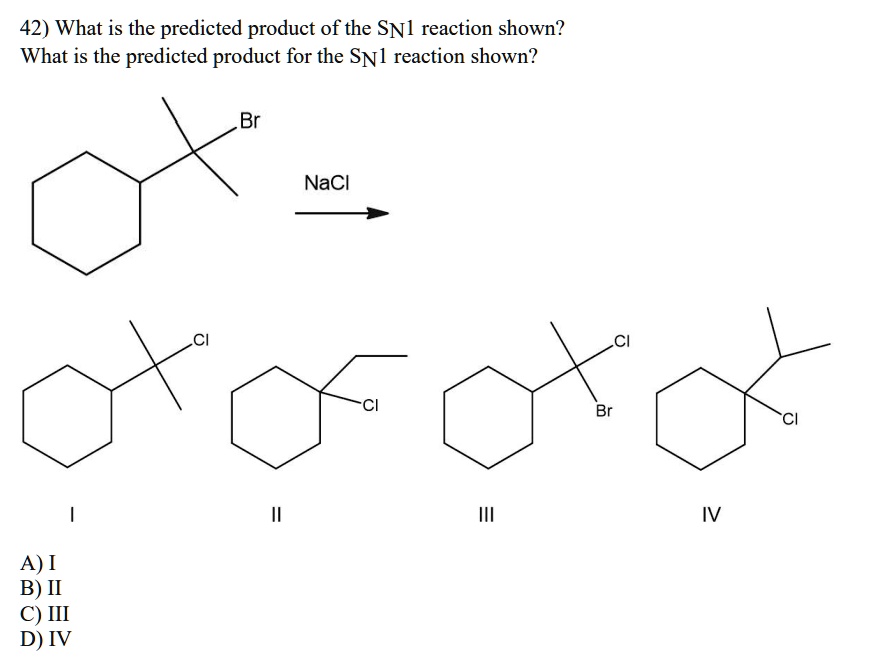
So, if you form a secondary carbocation, and there's a way for it to shift a hydride or an alkyl group to become a tertiary carbocation, it will do it. And then the nucleophile will attack the more stable tertiary carbocation. This means the product you get might not be where you initially expected the leaving group to be! It's like the molecule decided to take a scenic detour on its way to the final destination.
To predict the major product, you gotta be on the lookout for these potential rearrangements. Draw out the initial carbocation. Then, see if you can move any groups (hydrides or alkyls) to a neighboring carbon to make the carbocation more stable. If you can, that's where the nucleophile is likely to end up. It's all about chasing that stability!
Let's recap the strategy for predicting the major SN1 product. It’s like a checklist, a mental flowchart.
- Identify the substrate: Is it tertiary? Secondary? Primary? Tertiary is prime for SN1.
- Identify the leaving group: Is it a good one? Halides, tosylates, mesylates – these are your friends.
- Identify the nucleophile: Is it weak? Water, alcohols – perfect for SN1.
- Identify the solvent: Is it polar protic? Water, alcohols – helps stabilize everything.
Now for the actual product prediction:
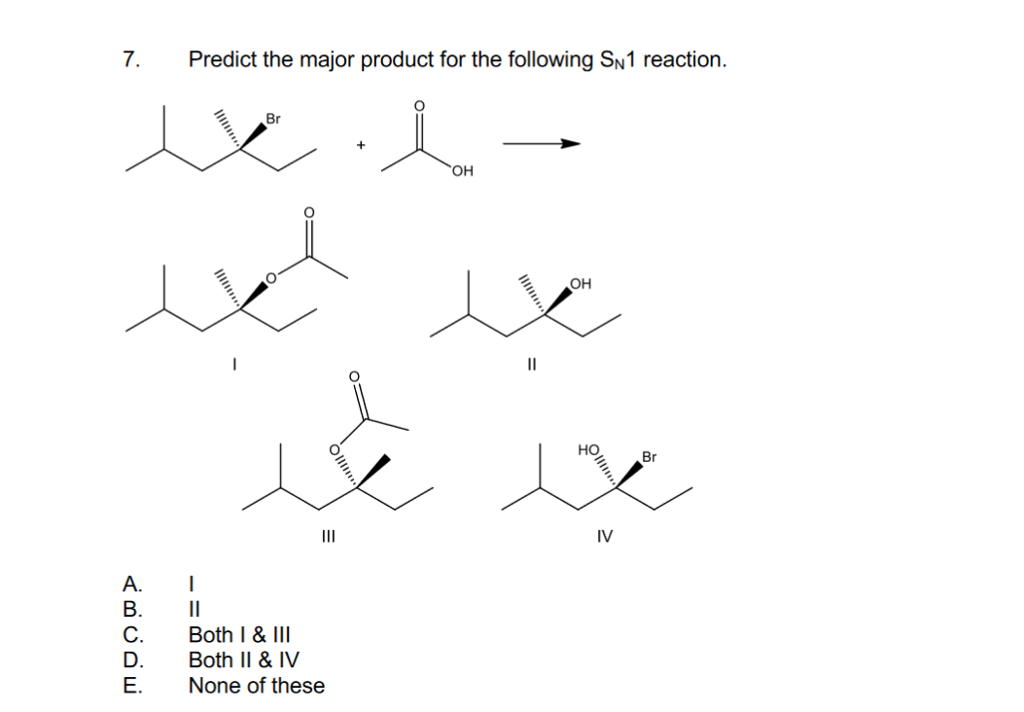
- Leaving group departs: Draw the carbocation intermediate.
- Check for rearrangements: Can the carbocation rearrange to become more stable? Look for hydride shifts and alkyl shifts to create a more substituted carbocation. If a more stable carbocation can form, that's the one the nucleophile will attack. This is CRUCIAL for predicting the major product.
- Nucleophile attacks: The nucleophile attacks the most stable carbocation (after any rearrangements). Remember, it can attack from either side, potentially leading to a racemic mixture (or a slight preference for one enantiomer if rearrangement leads to a new chiral center or affects a pre-existing one).
- Proton transfers (if applicable): If your nucleophile was neutral (like water or an alcohol), the initial product will be positively charged. You might need a final deprotonation step to get the neutral final product. Think of it as cleaning up the party afterwards.
So, when you're looking at that reaction, don't just see a bunch of atoms. See the potential for a carbocation. See the possibility of a rearrangement. See the dance between the leaving group, the carbocation, and the nucleophile. It's a dynamic process!
For example, imagine you have 2-bromo-2-methylbutane with water.
- It's a tertiary substrate. Bingo.
- Bromine is a good leaving group. Check.
- Water is a weak nucleophile. Check.
- Water is a polar protic solvent. Check.
But what if we had 2-bromobutane with water?
- Secondary substrate. Uh oh.
- Bromine is good. Check.
- Water is weak. Check.
- Water is polar protic. Check.
It's all about making those educated guesses, based on the stability of intermediates. Carbocations are like hyperactive toddlers – they want the most exciting (stable) spot. And the nucleophile just follows them there.
So next time you see an SN1 reaction, don't just panic. Take a deep breath, grab your mental toolkit, and start analyzing. Substrate, leaving group, nucleophile, solvent. Then, draw that carbocation. Then, hunt for rearrangements. That's your path to predicting the major product. Happy predicting, and may your reactions always be predictable (or at least, predictably interesting)!
