Predict The Major Alkene Product Of The Following E1 Reaction
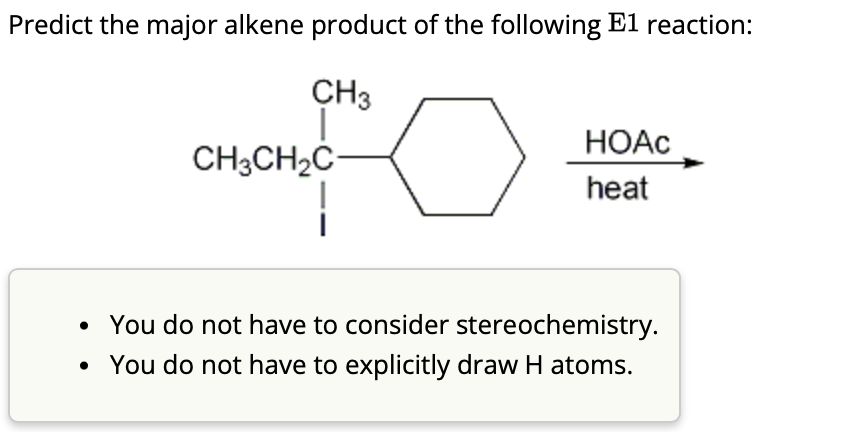
Imagine you're at a bustling party, and everyone's trying to get to the dance floor. Our special molecule is a bit like that. It's got this central character, let's call it the "leaving group," which is like that one friend who always announces they're "just popping out for a moment." This friend isn't just leaving; they're taking a piece of the molecule's structure with them, like a souvenir!
When this leaving group bails, it leaves behind a bit of a mess, a sort of emotional void where it used to be. Think of it like someone leaving a cozy spot on the couch. Suddenly, there's an empty space, and the molecule starts to feel a bit wobbly. This wobbly state is what chemists call a "carbocation." It's like the molecule's "uh-oh" moment. It's now feeling quite unstable, a bit like a toddler who's just discovered they can reach the cookie jar unattended – exciting but potentially chaotic.
Now, this carbocation doesn't like being wobbly for long. It's actively looking for something to make it feel more stable and grounded again. It's like that friend who, after their dramatic exit, is immediately scanning the room for a new conversation to jump into. The molecule, in its unstable state, wants to get back to feeling "normal," which in chemistry terms, means finding a way to share its electrons and make new, stronger bonds.
The interesting part is how it achieves this stability. It's not just one way; it's like there are multiple paths to happiness, or in this case, stability. The molecule has these things called "hydrogens" hanging around, which are like the friendly, eager bystanders at our party. These hydrogens are attached to neighboring parts of the molecule, and they're willing to lend a hand (or an electron, to be precise) to help the carbocation out of its predicament.
When a hydrogen decides to help, it basically hops over, bringing its electron with it, and forms a new connection, a "double bond." This double bond is like the molecule finding a new, exciting dance partner. It’s a more energetic and shared connection than the single bonds it had before. This is where things get really interesting, because there might be more than one hydrogen that could make this jump. Imagine our molecule has a few different dance-ready neighbors, each with a hydrogen willing to join the fun.

The question is, which hydrogen is the most likely to grab the dance floor with the carbocation? This is where the concept of "Zaitsev's Rule" pops up, which, despite its slightly intimidating name, is actually quite straightforward. Think of it as the molecule's internal popularity contest. The dance floor (the double bond) is most likely to form with the hydrogen that comes from the most crowded or "substituted" neighbor. It's like the molecule wants to form its new, exciting bond with the part of itself that already has more "friends" or attachments.
Why? Because forming a double bond in a more crowded area actually makes the resulting molecule more stable. It's like building a stronger, more robust structure. The more "stuff" already attached to the carbon atoms involved in the new double bond, the more stable the final product will be. It’s like choosing to build your new house on a well-established street rather than in a completely undeveloped area – there's more infrastructure, more support.
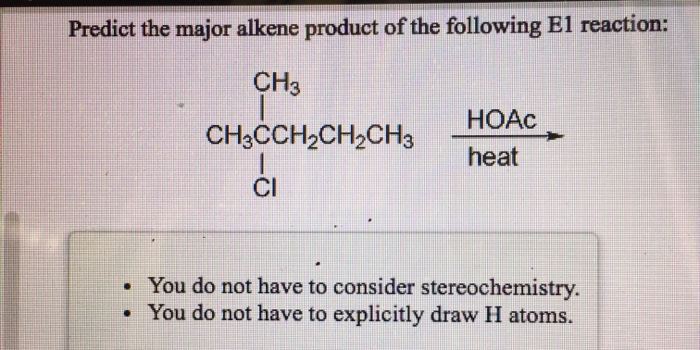
So, when we're trying to predict the major alkene product – that's our dancing, double-bonded molecule – we're essentially looking for the most stable dance floor. We're watching where those willing hydrogens will jump. Most of the time, the molecule will go for the option that creates the most connections around the new double bond. It's the most popular kid in school getting the most attention, but in a good way, making things more stable for everyone involved.
Sometimes, there might be a slightly less popular option, a hydrogen from a less crowded neighbor that also wants to join the dance. This will lead to a different alkene, a slightly less stable, perhaps more quirky dance move. But our molecule, being a creature of stability, usually gravitates towards the most popular, most substituted spot. It's like choosing the biggest, most vibrant party over the quieter gathering in the corner. It's the natural inclination for things to seek out the most comfortable and secure arrangements.
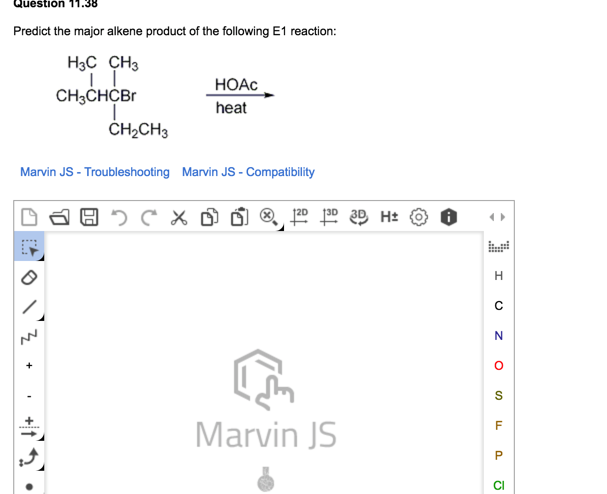
So, the next time you see a molecule going through this E1 process, imagine the drama, the leaving group's departure, the carbocation's wobble, and then the enthusiastic hydrogens stepping up. It’s a tiny, molecular soap opera, with the most stable alkene always aiming to be the star of the show, thanks to a little bit of chemistry and a lot of good old-fashioned seeking out the most crowded dance floor!
