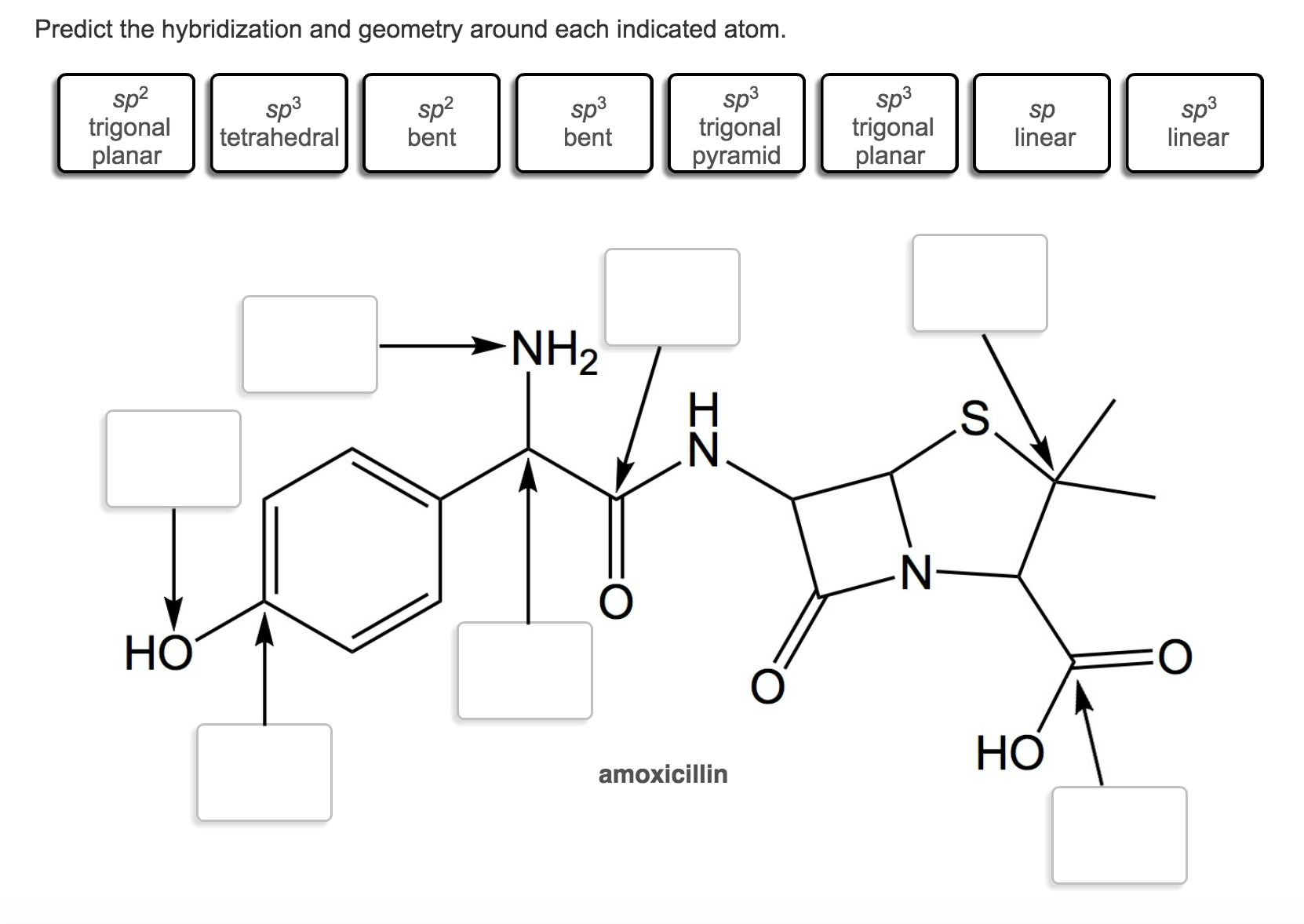
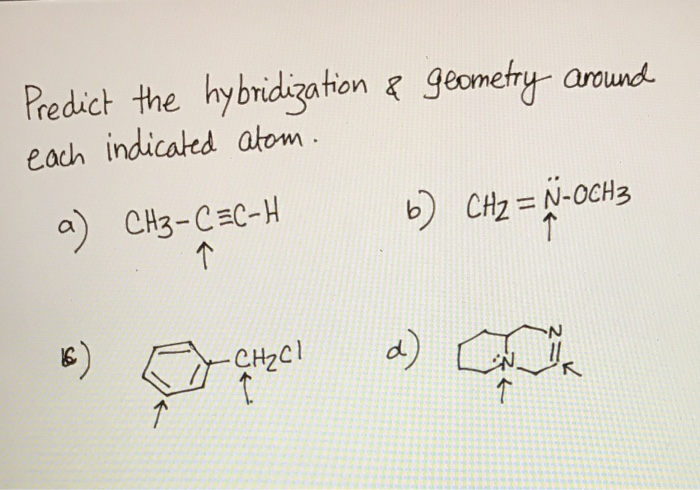
Predict The Hybridization And Geometry Around Each Indicated Atom

Let's dive into the wonderfully weird world of atoms, shall we? You know, those tiny building blocks that make up everything. Sometimes, they get together and decide to get cozy. And when they do, they arrange themselves in some rather predictable, yet oddly fascinating, ways. It’s like a cosmic dance, and we, the mere mortals, get to peek at the choreography. We're not going to get bogged down in the nitty-gritty, the “why” and the “how.” Nope, that's for the folks who enjoy wearing tweed and talking about quantum mechanics over lukewarm tea. We’re here for the fun part: predicting the atomic hodgepodge and the shapes they decide to form. It’s less about textbooks and more about trusting your gut. Or maybe just a hunch. My hunch usually involves snacks, but in this case, it’s about atoms.
So, imagine you’ve got a molecule. It's like a little atomic party. We've got our indicated atoms, the VIPs of this particular shindig. Our mission, should we choose to accept it (and we do, because, well, it’s right here), is to predict their hybridization and geometry. Think of hybridization as the atom’s preferred way of mingling, and geometry as the resulting awkward or elegant pose it strikes. It’s not a precise science, at least not in the way we’re doing it. It’s more like guessing what your cat is going to do next. Usually, it involves a nap, but sometimes it’s a frantic dash up the curtains. Atoms are a bit like that, but with more bonds and less fur.
Let's start with something simple, shall we? Like, say, the carbon atom in methane. That little guy is surrounded by hydrogens, all holding hands. It’s like a perfectly harmonious, square-ish hug. For methane’s carbon, we’re going to predict it’s using its sp3 hybridization. And the geometry? Oh, that's a classic: tetrahedral. It’s like a perfectly balanced pyramid, but in 3D. If you were to draw it, it would look like it’s reaching out to hug everyone. Very friendly, this carbon.
Now, what if we switch gears? Let’s look at the carbon in ethene. This one’s a bit more sophisticated. It’s not just holding hands; it’s engaged in a double handshake with another carbon. This means it's got some extra orbitals to show off. For our ethene carbon, we're looking at sp2 hybridization. And the geometry? It’s flattened out, like it’s trying to be as efficient as possible. We call that trigonal planar. Think of a really neat pizza, all sliced into three perfect pieces. Each atom gets its own slice of space.
Moving on, we have the carbon in ethyne. This atom is even more streamlined. It's doing a triple handshake! That requires some serious atomic juggling. The hybridization here is a swift and efficient sp. And the geometry? It’s stretched out, like a supermodel on a runway. It's linear. Straight as an arrow. No fuss, no muss. Just pure, unadulterated straightness. It’s the minimalist of the molecular world, and I can respect that.
But it's not just carbon that gets to play this hybridization game. Oh no. Other atoms join the fun too. Take oxygen. In water, for instance. The oxygen atom is like the busy parent, holding onto two hydrogens and two lone pairs of electrons. It’s got a bit of a busy schedule. We predict its hybridization is sp3, just like the methane carbon. But its geometry? Because of those grumpy lone pairs pushing things around, it’s not a perfect tetrahedral. It's bent, like it's trying to peek around a corner. We call that bent or angular geometry. It’s a bit shy, this water oxygen.
Let's consider nitrogen. In ammonia, that nitrogen atom is surrounded by three hydrogens and has one lone pair of electrons. Similar to the water oxygen, it’s rocking the sp3 hybridization. And the geometry? Again, those lone pairs are doing their thing. It’s like a little pyramid with the nitrogen at the top, but one side is a bit squished. It’s trigonal pyramidal. Think of a tiny, pointy hat. Very distinguished.
And what about boron? In boron trifluoride, the boron atom is bonded to three fluorines. It’s quite spread out. For boron, we’re predicting sp2 hybridization. And the geometry? It’s all about creating space. It’s trigonal planar. Just like our ethene carbon. It’s like a perfectly arranged flower, with each fluorine petal in its own spot. Very organized.
Honestly, I think this whole hybridization and geometry thing is super underrated. It’s like knowing the secret handshake of molecules. It’s not about memorizing endless rules; it’s about recognizing patterns. And once you see them, you can’t unsee them. It’s like suddenly noticing all the different types of clouds, or the subtle variations in bird songs. You become a molecular detective, a shape-shifter predictor. And who doesn’t love a good prediction? Especially when it leads to a perfectly shaped atomic hug or a streamlined, linear stride. It’s the little things, you know? The atomic arrangements that bring a smile to our faces, even if we’re the only ones appreciating their geometric elegance. I, for one, am a big fan of the tetrahedral and the linear. They just seem so… confident. The bent ones are relatable, though. We all have those days, don’t we? Anyway, happy predicting!”
