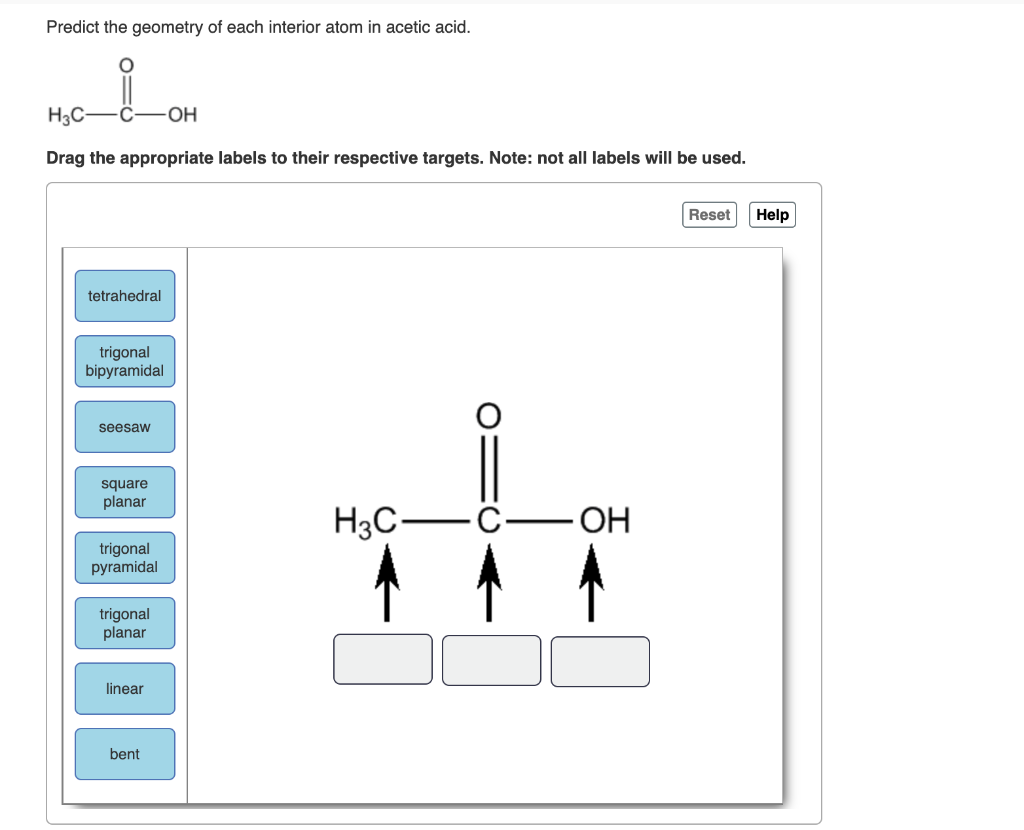
Predict The Geometry Of Each Interior Atom In Acetic Acid
Hey everyone! Ever wonder what makes molecules do their thing? Like, how they fold and bend and generally arrange themselves? Today, we're diving into the super cool world of acetic acid, that stuff in vinegar, and trying to predict the shape of each little atom inside it. Sounds a bit sci-fi, right? But it's actually pretty straightforward, and honestly, quite mind-blowing!
So, acetic acid. You know it, you've probably tasted it. It's got a bit of a kick. But behind that sour punch is a tiny, intricate structure. Imagine it like a microscopic LEGO set, where each brick is an atom, and they're all connected in a very specific way. We're not just talking about the overall shape of the molecule, but the geometry around each individual atom. It's like zooming in on each LEGO brick to see how it's oriented relative to its neighbors.
Why is this even a thing we care about? Well, the way atoms are arranged, their geometry, is super important. It dictates how the molecule interacts with everything else. Think of it like a handshake. The way your hand is shaped and positioned totally changes how you connect with someone else's. Molecules are the same way! Their shape determines if they'll stick to other molecules, react with them, or just float by like strangers.
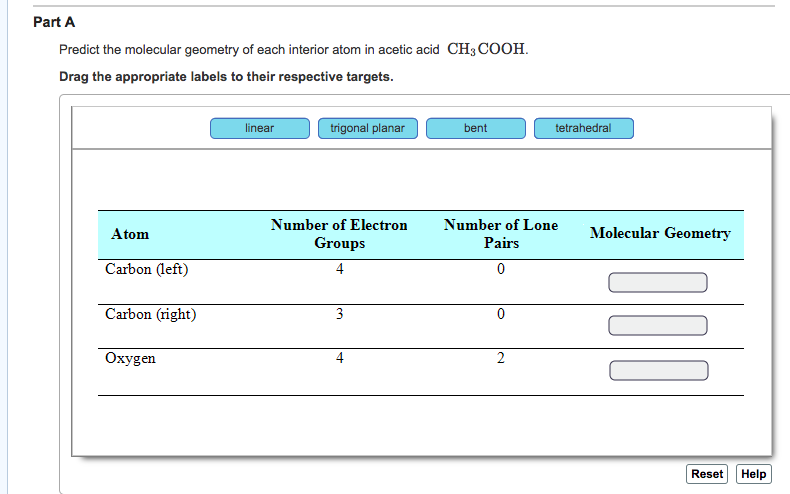
Let's break down acetic acid. Its chemical formula is CH₃COOH. This tells us we've got two carbon atoms (C), four hydrogen atoms (H), and two oxygen atoms (O). But that's just the ingredients list. We need to know how they're put together. We can visualize it as a central carbon atom attached to three hydrogens and another carbon atom. This second carbon atom is then attached to an oxygen atom with a double bond and another oxygen atom with a single bond, which is also attached to a hydrogen atom.
Okay, let's focus on predicting the geometry around each atom. This is where a handy concept called VSEPR theory (Valence Shell Electron Pair Repulsion theory) comes in. Don't let the fancy name scare you! It's basically a simple idea: electron pairs, whether they're in bonds or just hanging out as lone pairs, don't like to be too close to each other. They want to spread out as much as possible in 3D space to minimize repulsion. It's like a group of friends at a party – everyone tries to find their own personal space!
Geometry Around the First Carbon Atom (the CH₃ group)
Let's start with the carbon atom that's bonded to three hydrogens. We'll call this Carbon 1. This carbon atom is connected to four other atoms: three hydrogens and the second carbon atom (Carbon 2). So, we have four groups of electrons around Carbon 1. According to VSEPR theory, to get these four groups as far apart as possible, they'll arrange themselves in a tetrahedral shape. Imagine a pyramid with a triangular base, but with the apex pointing straight up. If you put the carbon atom at the center, the four surrounding atoms would be at the corners of this shape.
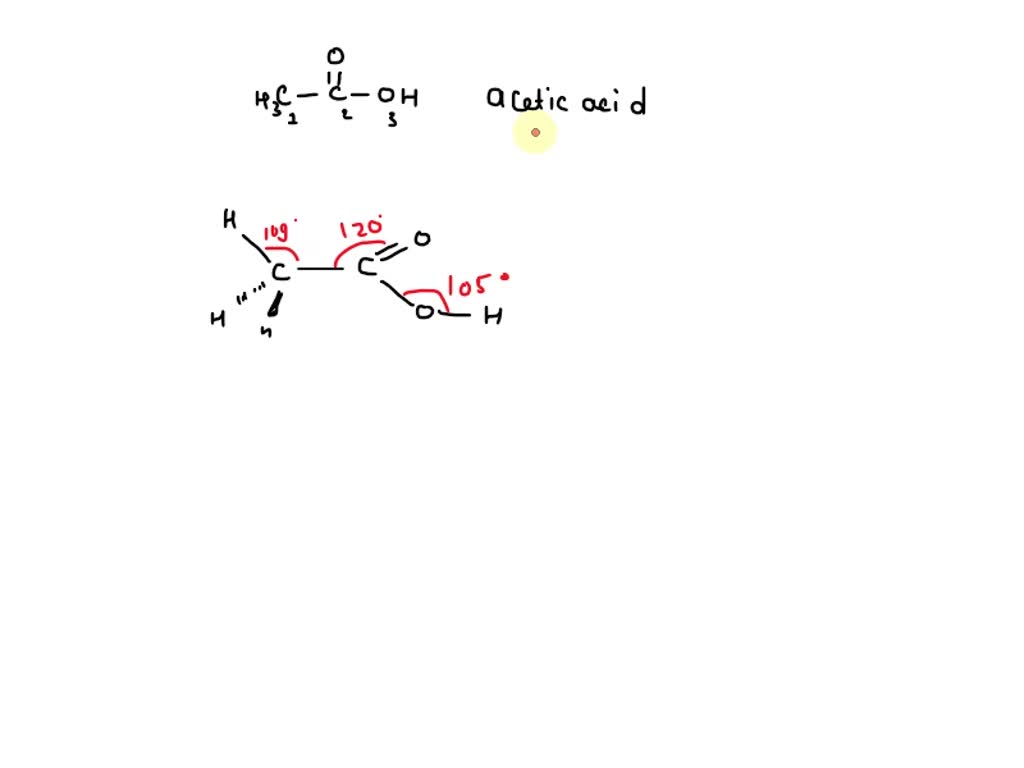
Each of these bonds – the C-H bonds and the C-C bond – will be roughly 109.5 degrees apart. It's a very stable arrangement. Think of it like a perfectly balanced tripod. Nothing's leaning too much in any direction. This is why the methyl group (CH₃) in acetic acid is often described as being tetrahedral. It's a neat and tidy arrangement, all spread out nicely.
Geometry Around the Second Carbon Atom (the COOH group)
Now, let's move to the second carbon atom, let's call it Carbon 2. This one is a little more interesting. It's bonded to Carbon 1, and then it's involved in a double bond with one oxygen atom and a single bond with another oxygen atom, which is then bonded to a hydrogen. So, we have Carbon 2 attached to Carbon 1, one oxygen with a double bond, and one oxygen with a single bond.

Here's where it gets a bit different. VSEPR theory focuses on the number of electron domains around the central atom. A double bond counts as one electron domain, even though it has more electrons, because the electrons in the double bond are sharing the same general space. So, around Carbon 2, we have three electron domains: the bond to Carbon 1, the double bond to the oxygen, and the single bond to the other oxygen.
When you have three electron domains, the best way to spread them out is in a flat, triangular arrangement. This is called trigonal planar geometry. Imagine a frisbee. The carbon atom is at the center, and the three groups it's bonded to are spread out equally around it, like spokes on a wheel, with angles of 120 degrees between them. This makes the entire COOH part of the molecule relatively flat.
So, Carbon 2 isn't tetrahedral like Carbon 1. It's trigonal planar. This flatness is actually quite important for how acetic acid behaves. It means that these atoms are all in the same plane, which can make it easier for them to interact with other molecules.
Geometry Around the Oxygen Atoms
Let's not forget the oxygen atoms! They play a crucial role. We have two of them.
The Doubly Bonded Oxygen
First, the oxygen atom that's double-bonded to Carbon 2. This oxygen atom has its electrons involved in the double bond with carbon. Besides that, it also has two lone pairs of electrons. So, in total, we have four groups of electrons around this oxygen atom: the double bond (which counts as one electron domain for VSEPR purposes) and the two lone pairs. VSEPR theory tells us that four groups of electrons will arrange themselves in a tetrahedral electron geometry.
However, we're usually more interested in the molecular geometry, which describes the arrangement of atoms. Because two of these electron groups are lone pairs, which aren't visible as atoms, the shape we see is actually a bit different. It's like having three people sitting at a round table, but one of the chairs is empty. You'd still call it a table setting, but the arrangement of people is different. In this case, the two bonds (the double bond to carbon and a hypothetical lone pair arrangement) push the double bond atoms into a shape that's often described as bent, or more formally, angular. The bond angles here are typically less than the ideal tetrahedral angle because the lone pairs, being more repulsive, tend to push the bonding pairs closer together.
The Singly Bonded Oxygen (with Hydrogen)
Finally, the oxygen atom that's single-bonded to Carbon 2 and also bonded to a hydrogen. This oxygen atom has its single bond to carbon, a single bond to hydrogen, and two lone pairs of electrons. Just like the other oxygen, this gives us four groups of electrons around the oxygen atom. Again, the electron geometry is tetrahedral.
But again, when we look at the molecular geometry, with two lone pairs and two bonding pairs, the atoms arrange themselves in a bent or angular shape. The bond angles will be similar to the other oxygen, compressed by the lone pairs. This is the oxygen that makes acetic acid acidic – the hydrogen atom it's bonded to is a bit exposed and can be released as a proton.
Putting It All Together
So, to recap: we have two carbon atoms, four hydrogen atoms, and two oxygen atoms.
- The first carbon (CH₃ group) is surrounded by four electron domains, giving it a tetrahedral geometry.
- The second carbon (COOH group) is surrounded by three electron domains (counting the double bond as one), giving it a trigonal planar geometry.
- Both oxygen atoms have four electron domains (one or two bonds and two lone pairs), leading to a tetrahedral electron geometry, but their molecular geometry is bent or angular.
Isn't that fascinating? It’s like each atom has its own little dance floor and follows specific rules for how to arrange itself. The overall shape of acetic acid is a consequence of these individual geometries. It’s not just a random jumble of atoms; it’s a carefully orchestrated molecular structure. And knowing these shapes helps scientists understand everything from how drugs work to how materials behave. Pretty neat, huh?
