Predict The Geometry And Polarity Of The Cs2 Molecule

Hey there, science curious folks! Ever stopped to think about those tiny, invisible building blocks that make up absolutely everything around us? We’re talking about atoms, and they’re like the ultimate Lego bricks of the universe. Sometimes, these atoms decide to team up, forming what we call molecules. And today, we’re going to peek into the life of a rather interesting, and perhaps a little bit shy, duo: the
Now, you might be thinking, "Why on earth should I care about two cesium atoms?" Well, imagine you’re at a party, and you see two people who are really, really different from each other. One is super outgoing and loves to share everything, while the other is a bit more reserved and likes to keep things to themselves. That’s kind of like how atoms behave when they meet! Their personalities, or rather, their properties, dictate how they interact and what kind of "friendship" they form.
And that’s where predicting the
So, What's the Deal with Geometry?
Think about how you arrange your furniture. You can have a couch against a wall, a coffee table in the middle, or maybe a comfy armchair in a corner. The arrangement matters, right? It affects how the room feels and how you can move around in it. Molecules are no different!
When atoms get together to form a molecule, they don't just randomly stick. They’re very particular about their personal space. They want to spread out as much as possible, like kids on a playground trying to get their own swing. This drive to spread out is all about minimizing those annoying repulsions between their electron clouds (which are like their tiny, buzzing energy fields).
For our Cs₂ molecule, we have just two cesium atoms. Imagine two balloons tied together. How can they be arranged? There’s really only one way for them to sit next to each other: in a straight line. They’re linked, one after the other. So, the

There are no other atoms to push them around, no complex angles to form. Just two buddies holding hands, and the only way they can do that is side-by-side, forming a straight connection. It’s the most straightforward arrangement imaginable. No drama, no complicated angles, just a simple connection.
Now, Let's Talk About Polarity (It's Not About Being Moody!)
Polarity is a super fun concept. Forget about human moods for a second; in chemistry, polarity is all about how evenly the "stuff" (electrons, specifically) is shared between atoms in a bond. Think of it like sharing a pizza. If everyone gets an equal slice, that’s like an even distribution. But if one person is a bit greedy and takes a bigger slice, that’s an uneven distribution.
Atoms have something called
If two atoms have a big difference in electronegativity, the electrons will spend more time hanging out around the more electronegative atom. This creates a slight "negative" charge on that atom and a slight "positive" charge on the other. This is what we call a
But here's the twist for our Cs₂ molecule. We have two cesium atoms. And guess what? They are exactly the same! They have the identical electronegativity. It's like two friends, both with the same love for pizza, deciding to share a pie. They're both equally good at grabbing slices, so the pizza gets divided perfectly equally.
Since both cesium atoms have the same pull on the electrons, the electrons are shared
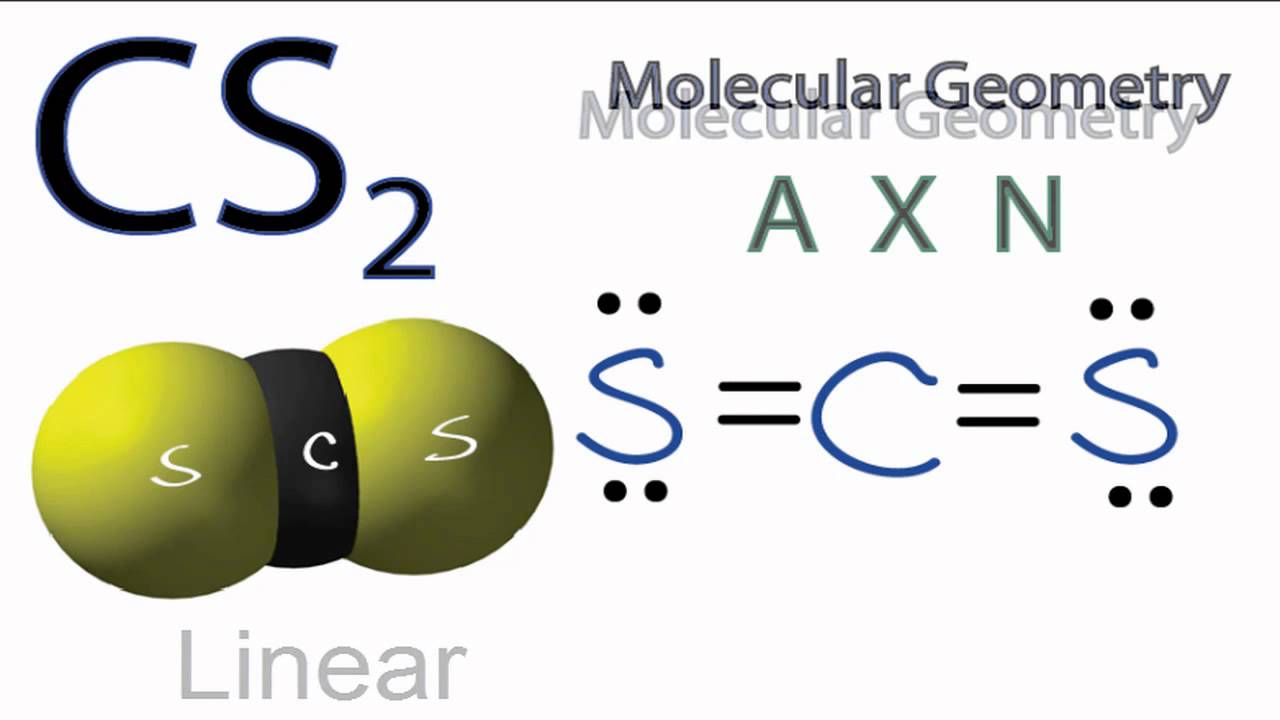
Why Should We Even Bother?
You might still be wondering, "Okay, so they're linear and nonpolar. Big deal!" Well, this simple knowledge is actually the key to unlocking a whole lot of fascinating properties and behaviors of matter. It's like knowing the basic ingredients to bake a cake – once you know them, you can create all sorts of deliciousness!
The polarity of a molecule dictates how it interacts with other molecules. Think about water (H₂O). It’s a polar molecule. That’s why water molecules like to stick together, forming surface tension (the reason you can sometimes float a paperclip on water) and why they're such a great solvent for many substances. They’re like little magnets, attracted to opposite charges in other polar molecules.
Nonpolar molecules, on the other hand, behave differently. They tend to mix better with other nonpolar substances. Imagine trying to mix oil and water. Oil is mostly nonpolar, and water is polar. They just don't get along, and they separate! If you had a molecule that was nonpolar like Cs₂, it would behave more like oil than like water when mixed with other things.
Understanding the geometry and polarity of molecules like Cs₂ is fundamental to fields like
It’s also crucial in
So, the next time you see something made of atoms, remember that their individual personalities – their geometry and their polarity – are working together to give that substance its unique characteristics. And for our little Cs₂ molecule, it’s a simple, straight handshake, with perfectly fair sharing. A humble but important piece of the grand puzzle of existence!
