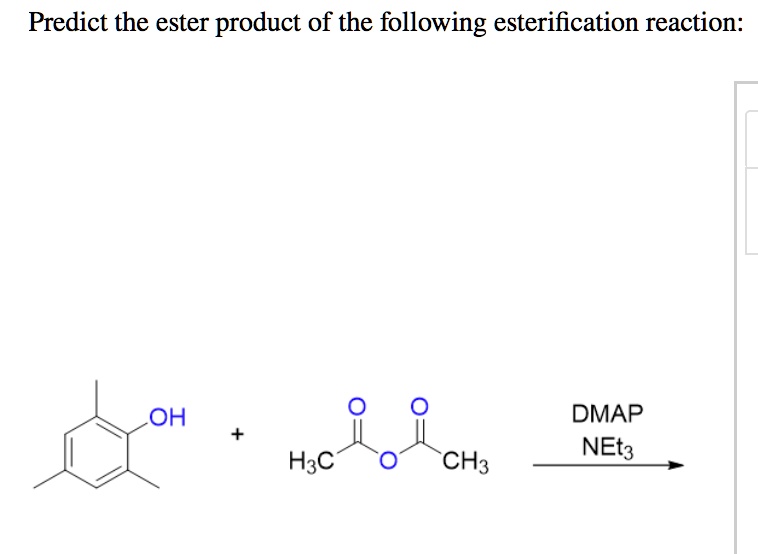
Predict The Ester Product Of The Following Reaction:
Ever feel like you're a secret chemist, concocting delicious flavors in your own kitchen? Well, get ready, because today we're diving into the super-cool world of making yummy-smelling stuff called esters! Think of those amazing smells that waft from a bakery, or the fresh scent of your favorite fruity shampoo. Yep, that’s often the magic of esters at play. And guess what? Predicting what kind of ester you’ll end up with is easier than you think, almost like a fun guessing game with a guaranteed sweet reward!
Imagine you’re a chef, and you have two key ingredients for your super-secret recipe. In our little chemical adventure, these ingredients are called an acid and an alcohol. Now, these aren't your everyday kitchen ingredients, but they’re the building blocks for all sorts of delightful smells and tastes. Think of the acid as the zesty, tangy part – maybe a bit like lemon juice, giving things a bit of a punch. And the alcohol? Well, it’s usually a bit smoother, a bit more… well, alcoholic in its pure form, but in this context, it’s like the carrier for our fragrant molecules. Together, they’re ready to dance and create something brand new!
Now, for the grand reveal! We’re going to look at a specific recipe, a little chemical dance floor, where our acid and our alcohol meet. The mission, should you choose to accept it (and trust me, you’ll want to!), is to predict the ester product. It’s like looking at your ingredients and knowing exactly what delicious cake you're going to bake. No mind-reading required, just a keen eye and a bit of chemical intuition.
Let’s say our star players are acetic acid and ethanol. Sounds fancy, right? But don’t let the names scare you! Acetic acid is basically the stuff that gives vinegar its zing. It’s that sharp, tangy note that makes your taste buds tingle. And ethanol? That’s the type of alcohol found in your favorite beverages, but here it's playing a starring role in creating wonderful aromas. When these two get together, it’s like they’re a match made in fragrance heaven!
So, how do we predict what magnificent ester they'll create? It’s all about a little bit of swapping. Imagine the acid is wearing a little hat, and the alcohol has a little hand. When they get close and cozy, the hat from the acid comes off, and the hand from the alcohol takes its place. It’s a simple swap, a chemical handshake, that leads to a whole new molecule with a brand new personality!
In our example of acetic acid and ethanol, the acid part, the "acetic" bit, will join up with the "ethyl" bit from the ethanol. And poof! You get ethyl acetate. Now, this isn’t just any old chemical compound. Oh no! Ethyl acetate is famous for its sweet, fruity smell. Think of the scent of nail polish remover – that's a good clue! Or, more pleasantly, it’s a key player in the aroma of certain fruits, giving them that lovely, inviting fragrance. It’s like the universe decided to bottle a little bit of fruity joy!
The beauty of this is that the structure of the ester is predictable based on the structure of the starting acid and alcohol. You just need to identify the "acyl" group from the acid (the part that's left after the hydrogen ion leaves) and the "alkyl" group from the alcohol (the part that's left after the hydroxyl group leaves). Then, you slap them together, and voilà! You’ve got yourself a brand new ester!

Let's try another one, just for kicks! Imagine we have benzoic acid and methanol. Benzoic acid has a lovely, slightly floral scent in its pure form. And methanol, well, it’s another type of alcohol. When these two get together, that "benzoyl" part from the benzoic acid is going to team up with the "methyl" part from the methanol. The result? Methyl benzoate! And what does methyl benzoate smell like? Get ready for this: it’s got a delightful, sweet aroma reminiscent of cherries! Can you believe it? Just two simple molecules, and you’ve conjured up the essence of cherry pie!
It’s like being a flavor detective, sniffing out clues and piecing together the puzzle to create delicious scents. The “-oic acid” ending on the acid usually tells you where to grab the "acyl" group, and the “-ol” ending on the alcohol is your signal for where to grab the "alkyl" group. Then, you just name the new creation by combining the alcohol’s name (minus the "-ol") with the acid’s name (minus the "-oic acid," but with "-ate" added). So, if you see propanoic acid and butanol, you know you're looking at butyl propanoate. Easy peasy, right?

So, the next time you encounter an acid and an alcohol in a reaction, don't just see them as boring chemical formulas. See them as ingredients ready to transform into something amazing! With a little practice, you’ll be predicting these ester products like a seasoned perfumer, all from the comfort of your own imagination. It’s a wonderful world of smells waiting to be discovered, and you, my friend, are the key!
